Advanced Synthesis of Chiral Alpha-Methyl Arylethylamine for Scalable API Manufacturing
Advanced Synthesis of Chiral Alpha-Methyl Arylethylamine for Scalable API Manufacturing
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective synthetic routes for critical chiral intermediates, particularly those serving as the backbone for major therapeutic classes such as respiratory agents and oncology drugs. Patent CN112920053B introduces a groundbreaking preparation method for chiral alpha-methyl arylethylamines, addressing long-standing inefficiencies in the production of key fragments like (R)-alpha-methyl-4-methoxyphenethylamine and (R)-3-(2-aminopropyl)-1H-indole. These compounds are indispensable precursors for blockbuster medications including Formoterol, Carmoterol, and the selective estrogen receptor downregulator AZD9496. By leveraging a novel sulfonamide intermediate strategy, this technology bypasses the hazardous high-pressure hydrogenation steps and wasteful chromatographic separations that have historically plagued this chemical space. For global procurement and R&D teams, this represents a pivotal shift towards greener, safer, and more economically viable manufacturing protocols that align with modern regulatory and sustainability standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral alpha-methyl arylethylamines has been fraught with significant technical and economic hurdles that impede efficient large-scale production. Traditional routes often rely heavily on palladium-catalyzed high-pressure hydrogenation, a process that not only incurs substantial capital expenditure for specialized reactor equipment but also introduces severe safety risks associated with handling hydrogen gas under pressure. Furthermore, alternative methods utilizing chiral induction or diastereomeric resolution frequently suffer from poor atom economy; for instance, processes requiring silica gel column chromatography to separate unwanted isomers result in the discard of nearly half the theoretical yield, generating massive amounts of solid waste and driving up disposal costs. Other reported strategies employ expensive chiral auxiliaries like tert-butyl sulfinamide, which drastically inflate raw material costs and often necessitate multiple recrystallization steps to achieve acceptable optical purity, thereby complicating the operational workflow and extending production lead times significantly.
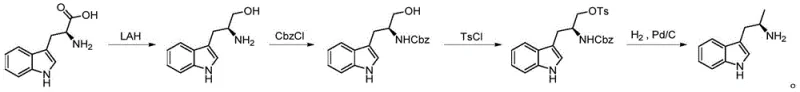
The Novel Approach
In stark contrast to these legacy methodologies, the innovative pathway disclosed in patent CN112920053B offers a streamlined, three-step sequence that fundamentally redefines the efficiency of chiral amine synthesis. This novel approach initiates with the reduction of readily available Boc-amino acid methyl esters, utilizing inexpensive sodium borohydride to generate Boc-amino alcohols with high fidelity. The core innovation lies in the subsequent conversion of these alcohols into sulfonamide compounds via thionyl chloride activation and ruthenium-catalyzed oxidation, a transformation that preserves the chiral integrity of the molecule while setting the stage for a highly selective reduction. By replacing dangerous high-pressure hydrogenation with a mild, Lewis acid-promoted borohydride reduction system, this method effectively eliminates safety hazards while ensuring exceptional stereochemical control. The result is a process that delivers single optical isomer products with superior purity and yield, completely obviating the need for resource-intensive column chromatography and making it ideally suited for continuous, large-scale industrial application.
Mechanistic Insights into Ruthenium-Catalyzed Oxidation and Lewis Acid Promotion
The mechanistic elegance of this synthesis lies in its strategic use of oxidation states and Lewis acid coordination to drive reactivity under mild conditions. In the second step, the conversion of the chloro-intermediate to the sulfonamide is facilitated by a ruthenium trichloride catalyst in the presence of sodium periodate. This oxidative system is crucial as it generates the sulfonyl functionality without racemizing the adjacent chiral center, a common pitfall in many amino acid derivatizations. The resulting sulfonamide acts as a robust activating group that significantly enhances the electrophilicity of the adjacent carbon center, preparing it for the final reductive cleavage. This specific activation allows for the use of sodium borohydride, typically a mild reducing agent, to perform a transformation that would otherwise require much harsher conditions. The preservation of the chiral pool information from the starting Boc-amino acid throughout this sequence ensures that the final amine retains the desired configuration without the need for downstream resolution.
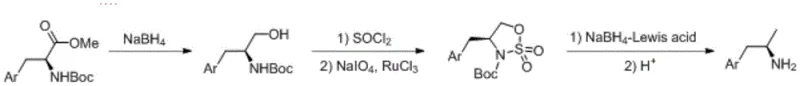
Furthermore, the final reduction step employs a sophisticated Lewis acid promotion mechanism that is critical for both reaction rate and selectivity. The addition of Lewis acids such as anhydrous lithium chloride or zinc chloride coordinates with the sulfonamide oxygen atoms, thereby lowering the energy barrier for the hydride attack by sodium borohydride. This coordination effect not only accelerates the reaction kinetics, allowing it to proceed at moderate temperatures between 50°C and 120°C, but also suppresses side reactions that could lead to impurity formation. Following the reduction, the acidic deprotection step cleanly removes the Boc group to release the free amine. This mechanistic pathway effectively decouples the reduction event from the safety risks of hydrogen gas, providing a chemically distinct advantage that translates directly into operational simplicity and enhanced process safety for manufacturing facilities.
How to Synthesize Chiral Alpha-Methyl Arylethylamine Efficiently
Implementing this advanced synthesis protocol requires precise control over reaction parameters to maximize yield and purity, particularly during the oxidation and reduction phases. The process begins with the careful reduction of the starting ester, followed by the critical formation of the sulfonamide intermediate which dictates the success of the final step. Operators must maintain strict temperature controls, especially during the exothermic addition of thionyl chloride and the subsequent oxidation, to prevent degradation of the sensitive intermediates. The detailed standardized operating procedures for executing this high-efficiency route, including specific molar ratios, solvent choices, and workup protocols, are outlined in the comprehensive guide below.
- Reduce Boc-amino acid methyl ester with sodium borohydride to obtain Boc-amino alcohol.
- React the alcohol with thionyl chloride followed by ruthenium-catalyzed oxidation with sodium periodate to form a sulfonamide compound.
- Perform Lewis acid-promoted reduction with sodium borohydride followed by acidic deprotection to yield the target chiral amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers transformative benefits that extend far beyond simple chemical yield improvements. By shifting away from high-pressure hydrogenation and chromatographic purification, manufacturers can significantly reduce the total cost of ownership associated with producing these critical intermediates. The elimination of expensive palladium catalysts and the avoidance of high-pressure reactor vessels lower both capital expenditure and ongoing maintenance costs, while the use of commodity chemicals like sodium borohydride and thionyl chloride ensures a stable and predictable raw material supply chain. Moreover, the drastic reduction in solid waste generation, specifically the removal of silica gel from the process flow, simplifies environmental compliance and waste disposal logistics, contributing to a more sustainable and resilient manufacturing operation that is less vulnerable to regulatory shifts.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the substitution of costly noble metal catalysts and high-energy unit operations with inexpensive reagents and ambient pressure conditions. By removing the need for palladium-on-carbon and high-pressure hydrogen gas, the direct material costs are substantially lowered, and the safety infrastructure requirements are minimized. Additionally, the high atom economy achieved by avoiding the discard of unwanted isomers means that more of the input raw material is converted into saleable product, effectively spreading the fixed costs over a larger output volume and improving the overall margin profile for the intermediate.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly enhanced by the reliance on widely available, commodity-grade starting materials such as Boc-amino acid methyl esters, which are produced globally in large volumes. Unlike specialized chiral auxiliaries or custom catalysts that may have long lead times or single-source dependencies, the reagents used in this novel route are standard inventory items for most fine chemical suppliers. This ubiquity reduces the risk of supply disruptions and allows for more flexible sourcing strategies, ensuring that production schedules can be maintained even during periods of market volatility or logistical constraints.
- Scalability and Environmental Compliance: From a scale-up perspective, the simplicity of the operation—characterized by the absence of complex filtration steps like column chromatography—makes this process inherently easier to transfer from the laboratory to multi-ton production scales. The reduction in hazardous waste streams, particularly the elimination of spent silica gel and heavy metal residues, aligns perfectly with increasingly stringent environmental regulations and corporate sustainability goals. This cleaner profile not only reduces the burden on waste treatment facilities but also streamlines the regulatory approval process for new drug filings, accelerating the time to market for downstream pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, drawing directly from the experimental data and beneficial effects described in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages related to safety, purity, and operational efficiency that distinguish this method from conventional alternatives.
Q: How does this new method improve safety compared to traditional hydrogenation?
A: The patented process eliminates the need for high-pressure hydrogenation reactions catalyzed by palladium, which are associated with significant safety hazards and high equipment costs. Instead, it utilizes a mild chemical reduction system at atmospheric pressure.
Q: What are the purity levels achievable with this synthesis route?
A: Experimental data from the patent indicates that the final products consistently achieve purity levels exceeding 99%, with high optical purity derived directly from the chiral starting materials without the need for column chromatography resolution.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is specifically designed for scalability. It avoids complex separation techniques like silica gel column chromatography and uses cheap, commercially available raw materials, making it highly suitable for ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alpha-Methyl Arylethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality chiral intermediates for the development of life-saving medications. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of volume. Our state-of-the-art facilities are equipped to handle the specific requirements of this novel sulfonamide-based synthesis, maintaining stringent purity specifications through our rigorous QC labs to guarantee that every batch meets the exacting standards required for API manufacturing.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your supply chain with reliable, cost-effective solutions.
