Advanced Spiro Nitrogen Compounds for High Efficiency OLED Electron Transport Layers
The rapid evolution of the organic optoelectronic industry demands materials that transcend the limitations of traditional phosphorescent and fluorescent emitters, particularly in the realm of electron transport layers where carrier balance is critical. Patent CN112500410B introduces a groundbreaking class of spiro nitrogen-containing organic light-emitting compounds that fundamentally address the stability and efficiency bottlenecks found in current OLED architectures. By utilizing a spiro nitrogen derivative as the parent nucleus, this technology ingeniously incorporates a nitrogen-containing heterocycle that disrupts molecular symmetry, thereby preventing the detrimental intermolecular aggregation and crystallization that often plague thin-film devices. This structural innovation results in exceptional film-forming properties and robust electron transport capabilities, directly translating to organic electroluminescent devices with significantly higher luminous efficiency and prolonged operational lifetimes. For R&D directors and procurement specialists seeking a reliable OLED material supplier, this patent represents a pivotal shift towards next-generation display and illumination technologies that promise both performance superiority and manufacturing viability.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the organic electroluminescence industry has relied heavily on materials like Tris(8-hydroxyquinoline) aluminum (Alq3), which, despite its three-decade dominance, suffers from inherent limitations in carrier mobility balance. In conventional OLED device structures, the hole mobility of transport materials typically far exceeds the electron mobility of electron transport materials, creating a significant imbalance in carrier transport rates that leads to premature device degradation and reduced efficiency. Furthermore, existing materials often exhibit strong selectivity issues where performance varies drastically across different device structures, complicating the standardization required for mass production. The tendency for conventional small molecule materials to crystallize over time under thermal stress also compromises the integrity of the thin film layers, resulting in dark spots and reduced service life. These technical deficiencies necessitate frequent material replacements and complex device engineering to mitigate efficiency roll-off, thereby increasing the overall cost reduction in electronic chemical manufacturing challenges for producers.
The Novel Approach
The novel approach detailed in this patent overcomes these historical constraints by engineering a spiro-structured core that inherently resists crystallization while maintaining high thermal stability. By integrating a nitrogen-containing heterocycle into the spiro framework, the material achieves a delicate balance of high electron affinity and ionization energy, facilitating efficient electron injection from the cathode while effectively blocking holes to confine excitons within the emission layer. This molecular design ensures that the electron transport layer can effectively transport electrons to recombination regions far away from the cathode, correcting the carrier imbalance that plagues traditional architectures. The resulting compounds demonstrate superior film-forming characteristics that remain stable under prolonged operation, directly addressing the industrial application requirements for high efficiency and long service life. This strategic molecular engineering allows for the commercial scale-up of complex spiro compounds without sacrificing the electronic properties required for high-performance displays.
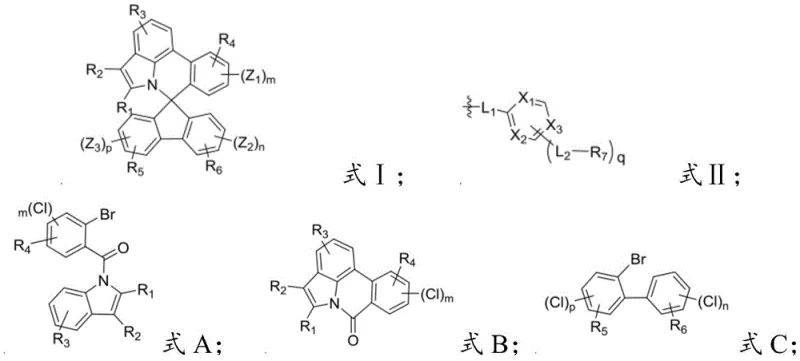
Mechanistic Insights into Spiro Nitrogen Compound Synthesis
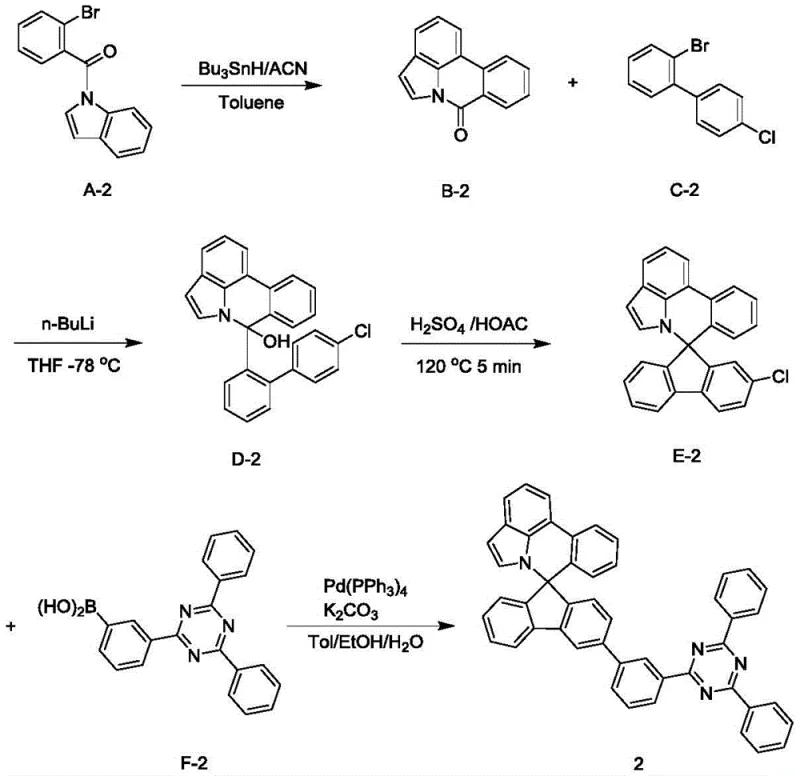
The synthesis of these advanced materials relies on a sophisticated multi-step sequence that begins with a radical cyclization to establish the core nitrogen-containing framework. In the initial stage, a precursor compound bearing a bromine substituent undergoes a reflux reaction with tributyltin hydride in the presence of acetonitrile and toluene, facilitating a radical-mediated intramolecular cyclization that constructs the fused ring system essential for the spiro architecture. This step is critical as it sets the stereochemical foundation for the subsequent spiro-center formation, ensuring that the nitrogen atom is correctly positioned to disrupt molecular symmetry in the final product. The reaction conditions are meticulously controlled under a nitrogen blanket to prevent oxidation of the sensitive radical intermediates, highlighting the need for precise atmospheric control during the manufacturing process to maintain high purity standards.
Following the core construction, the mechanism proceeds through a lithiation and acid-catalyzed spiro-formation sequence that locks the three-dimensional structure into place. The intermediate is treated with n-butyllithium at cryogenic temperatures of -78°C to generate a reactive organolithium species, which then attacks a ketone or electrophilic center to form a hydroxy-intermediate. This hydroxy group is subsequently subjected to acidic dehydration using concentrated sulfuric acid and glacial acetic acid at elevated temperatures of 120°C, driving the elimination of water and the formation of the quaternary spiro carbon center. This rigorous cyclization step is the defining moment of the synthesis, creating the rigid, non-planar geometry that prevents pi-stacking and aggregation in the solid state. The final step involves a palladium-catalyzed cross-coupling reaction, likely a Suzuki-Miyaura coupling, which attaches the functional electron-transporting side chains, completing the molecule with the precise electronic properties required for efficient device operation.
How to Synthesize Spiro Nitrogen Compound Efficiently
Implementing this synthesis route requires strict adherence to the patented four-step protocol to ensure the high purity and yield necessary for electronic grade applications. The process begins with the preparation of the spiro-intermediate through radical cyclization and subsequent spiro-ring closure, followed by a final coupling step to introduce the specific heteroaryl functionality. Each stage demands careful control of temperature and stoichiometry, particularly the cryogenic lithiation step and the high-temperature acid cyclization, to minimize byproduct formation. Detailed standardized synthesis steps are provided below to guide process engineers in replicating the high yields reported in the patent examples, such as the 84% isolated yield achieved for Compound 2.
- Reflux compound A with tributyltin hydride and acetonitrile in toluene to generate intermediate B via radical cyclization.
- React intermediate B with compound C and n-butyllithium in THF at -78°C to form hydroxy-intermediate D.
- Treat intermediate D with glacial acetic acid and concentrated sulfuric acid at 120°C to effect spiro-cyclization yielding intermediate E.
- Perform Suzuki coupling on intermediate E with boronic acid derivative F using Pd(PPh3)4 and K2CO3 to finalize the spiro nitrogen compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this spiro nitrogen technology offers substantial strategic benefits by simplifying the purification landscape and enhancing material reliability. The synthetic route utilizes widely available industrial reagents such as tributyltin hydride, n-butyllithium, and palladium catalysts, which ensures a stable supply chain不受 limited by exotic or scarce raw materials. The robustness of the reaction conditions, including standard solvent systems like toluene and THF, allows for seamless integration into existing chemical manufacturing infrastructure without requiring specialized equipment upgrades. This compatibility significantly reduces the barrier to entry for mass production, enabling suppliers to scale output rapidly to meet the growing demand for high-resolution OLED displays in consumer electronics and automotive sectors.
- Cost Reduction in Manufacturing: The elimination of complex purification steps typically required to remove crystalline impurities translates directly into lower processing costs and higher overall throughput. By preventing intermolecular aggregation through molecular design rather than post-synthesis purification, the process reduces the consumption of solvents and energy associated with repeated recrystallization cycles. Furthermore, the high yields reported in the patent examples, often exceeding 80% in key steps, minimize raw material waste and maximize the output per batch. This efficiency drives down the cost per gram of the final active material, offering a compelling economic advantage for procurement managers negotiating long-term supply contracts.
- Enhanced Supply Chain Reliability: The use of stable intermediates and robust reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining the yield of downstream OLED panel manufacturing. The synthetic pathway avoids highly unstable intermediates that require immediate consumption, allowing for the stocking of key precursors to buffer against supply fluctuations. This stability reduces lead time for high-purity electron transport materials, as production schedules can be forecasted with greater accuracy and less risk of batch failure. For supply chain heads, this reliability means fewer production stoppages and a more predictable inventory management strategy for critical display components.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction vessels and conditions that are easily transferable from laboratory to pilot and commercial scales. The waste streams generated, primarily consisting of tin salts and aqueous acidic waste, are well-understood in the chemical industry and can be managed through standard treatment protocols, ensuring compliance with environmental regulations. The high atom economy of the coupling steps and the ability to recycle solvents like toluene further enhance the environmental profile of the manufacturing process. This alignment with green chemistry principles supports corporate sustainability goals while maintaining the high performance required for advanced electronic applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this spiro nitrogen technology in OLED production lines. These answers are derived directly from the patent specifications and experimental data to provide accurate guidance for engineering and procurement teams. Understanding these details is essential for evaluating the feasibility of integrating this material into your current product roadmap.
Q: How does the spiro nitrogen structure improve OLED device performance?
A: The nitrogen-containing heterocycle within the spiro derivative disrupts molecular symmetry, which effectively prevents intermolecular aggregation and crystallization. This structural feature ensures superior film-forming properties and enhances electron mobility, leading to higher luminous efficiency and extended device lifetime compared to conventional materials like Alq3.
Q: What are the key reaction conditions for the spiro-cyclization step?
A: The critical spiro-cyclization step involves reacting the hydroxy-intermediate with glacial acetic acid and concentrated sulfuric acid. The process requires heating to 120°C for approximately 5 minutes under acidic conditions to facilitate the dehydration and ring-closure necessary to form the rigid spiro core structure.
Q: Can these materials be scaled for commercial OLED manufacturing?
A: Yes, the synthetic route utilizes standard industrial reagents such as tributyltin hydride, n-butyllithium, and palladium catalysts in common solvents like toluene and THF. The process avoids exotic conditions, making it highly amenable to commercial scale-up for the production of high-purity electron transport materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro Nitrogen Compound Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to bring this sophisticated spiro nitrogen synthesis from patent to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory grams to industrial tons is managed with precision and safety. We maintain stringent purity specifications and operate rigorous QC labs equipped to analyze electronic grade materials, guaranteeing that every batch meets the exacting standards required for OLED manufacturing. Our commitment to quality ensures that the superior electron transport properties described in the patent are consistently delivered in every shipment.
We invite you to collaborate with us to optimize your supply chain and reduce your overall material costs through advanced chemical engineering. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes and device architectures. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your next-generation display projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
