Advanced Spiro Nitrogen-Containing Organic Luminescent Compounds for High-Efficiency OLED Manufacturing
The rapid evolution of display technology has necessitated the development of advanced organic photoelectric materials capable of meeting the rigorous demands of high-resolution, flexible, and energy-efficient screens. Patent CN112480113B introduces a groundbreaking class of spiro nitrogen-containing organic luminescent compounds designed specifically to address the limitations of traditional electron transport layers in Organic Light-Emitting Diodes (OLEDs). These compounds utilize a unique spiro nitrogen-containing derivative as a parent nucleus, where the integration of a nitrogen-containing heterocycle strategically breaks molecular symmetry. This structural innovation is critical as it effectively prevents intermolecular aggregation and crystallization, phenomena that often degrade device performance over time. Consequently, materials derived from this patent exhibit exceptional film-forming properties and superior electron transmission capabilities, directly translating to organic electroluminescent devices with markedly higher luminous efficiency and extended service life.
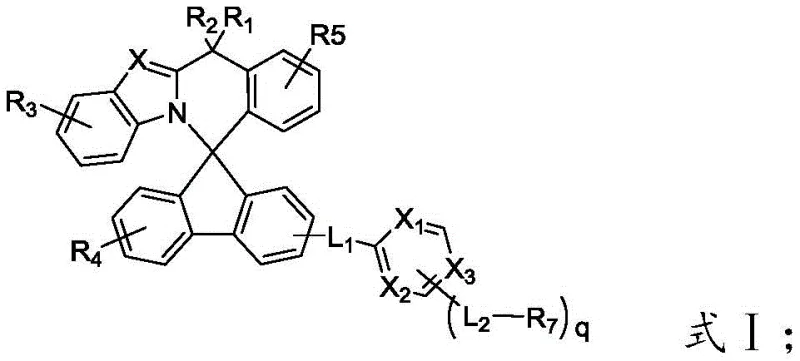
For R&D directors and technical leads, understanding the mechanistic advantages of this new architecture is paramount. The core innovation lies in the three-dimensional spiro configuration which disrupts the planar stacking typical of conventional aromatic systems. In traditional materials, planar molecules tend to pack closely, leading to concentration quenching and reduced charge mobility. By contrast, the spiro framework introduced in this patent creates steric hindrance that maintains molecular separation even in the solid state. This ensures that the electron transport layer remains amorphous and stable under thermal stress, a crucial factor for the longevity of commercial displays. The versatility of the structure is further highlighted by the variable substituents (R1-R7) and linkers (L1-L2), allowing for fine-tuning of energy levels to match specific host-guest systems in the light-emitting layer.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied heavily on materials like Tris(8-hydroxyquinoline) aluminum (Alq3) for electron transport applications. While Alq3 has served as a benchmark for nearly three decades, it suffers from inherent limitations when pushed to the performance boundaries required by modern high-end smartphones and large-area displays. One significant drawback is its tendency to crystallize over prolonged operation or under thermal load, which creates defects in the thin film and increases driving voltage. Furthermore, the electron mobility of conventional planar molecules is often insufficient to balance the injection rate of holes from the anode side. This imbalance leads to the formation of leakage currents where holes traverse the device without recombining with electrons, thereby reducing overall quantum efficiency and generating excess heat that accelerates device degradation.
The Novel Approach
The novel approach detailed in the patent overcomes these hurdles through a sophisticated molecular design centered on the spiro nitrogen scaffold. By incorporating nitrogen atoms directly into the conjugated system and enforcing a non-planar geometry, the new compounds achieve a delicate balance between high electron affinity and thermal stability. The synthesis strategy allows for the modular attachment of various aryl and heteroaryl groups, enabling chemists to optimize the Highest Occupied Molecular Orbital (HOMO) and Lowest Unoccupied Molecular Orbital (LUMO) levels for specific device architectures. This modularity ensures that the material can be adapted for different color emissions (red, green, blue) and device configurations without compromising the fundamental stability provided by the spiro core. The result is a material that not only transports electrons more efficiently but also forms robust, pinhole-free films essential for high-yield manufacturing.
Mechanistic Insights into Palladium-Catalyzed Spiro Cyclization
The synthesis of these high-performance materials relies on a robust sequence of transition metal-catalyzed reactions, primarily leveraging palladium chemistry to construct the complex carbon-nitrogen frameworks. A critical step in the pathway involves the formation of the spiro center itself, often achieved through an acid-mediated cyclization of a precursor alcohol. This step is pivotal as it locks the molecular conformation into the desired three-dimensional shape. Following this, the attachment of the electron-transporting side chains is typically accomplished via Suzuki-Miyaura cross-coupling reactions. This choice of chemistry is strategic; palladium-catalyzed couplings are renowned for their tolerance of various functional groups and their ability to proceed under relatively mild conditions, preserving the integrity of the sensitive spiro core. The use of ligands such as triphenylphosphine and bases like potassium carbonate facilitates the oxidative addition and reductive elimination cycles necessary to forge the carbon-carbon bonds linking the core to the peripheral aromatic systems.
Impurity control is another aspect where this mechanistic approach excels. The stepwise nature of the synthesis allows for the purification of key intermediates, such as the chlorinated spiro precursor, before the final coupling step. This is crucial for electronic grade materials where trace metal contaminants or unreacted halides can act as quenching sites for excitons. The patent specifies rigorous workup procedures, including multiple extractions, washing with saturated saline, and recrystallization from solvents like 1,4-dioxane. These purification protocols are designed to remove residual palladium catalysts and inorganic salts, ensuring that the final product meets the stringent purity specifications (>99.7% HPLC purity) required for commercial OLED fabrication. The high yields reported in the examples (often exceeding 80% for the final step) further indicate a clean reaction profile with minimal side-product formation.
How to Synthesize Spiro Nitrogen-Containing Organic Luminescent Compounds Efficiently
The preparation of these compounds follows a logical three-stage protocol that balances reaction efficiency with product purity. The process begins with the construction of the spiro skeleton, followed by functionalization and final assembly. Each stage is optimized for scalability, utilizing solvents and reagents that are readily available in the fine chemical supply chain. The detailed standardized synthesis steps below outline the specific conditions required to achieve the high yields and purities demonstrated in the patent examples, providing a clear roadmap for process chemists looking to implement this technology.
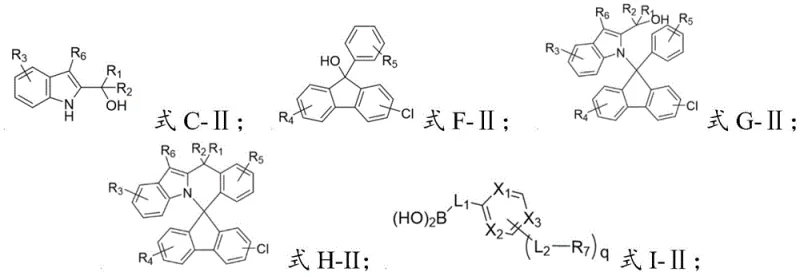
- React intermediate C-II with intermediate F-II using palladium hydroxide and sodium formate in p-xylene at 140°C to form intermediate G-II.
- Treat intermediate G-II with glacial acetic acid and concentrated sulfuric acid at 120°C to induce cyclization, yielding intermediate H-II.
- Perform a final Suzuki-type coupling between intermediate H-II and boronic acid derivative I-II using Pd(PPh3)4 and potassium carbonate in a toluene/ethanol/water system.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this new class of spiro nitrogen-containing compounds offers substantial strategic benefits beyond mere performance metrics. The synthesis route is designed with manufacturability in mind, avoiding exotic reagents or extreme conditions that would complicate sourcing or increase safety risks. The reliance on established palladium-catalyzed coupling chemistry means that existing infrastructure in many CDMO facilities can be utilized with minimal modification. This compatibility significantly reduces the barrier to entry for scaling production from laboratory grams to metric ton quantities. Furthermore, the high thermal stability of the final products implies a longer shelf life and reduced sensitivity to storage conditions, mitigating the risk of spoilage during transit and warehousing.
- Cost Reduction in Manufacturing: The streamlined synthetic pathway contributes to significant cost optimization in electronic chemical manufacturing. By achieving high yields in the final coupling steps and utilizing efficient purification methods like recrystallization rather than costly preparative chromatography, the overall cost of goods sold is drastically reduced. Additionally, the elimination of complex multi-step protection and deprotection sequences often found in older synthetic routes simplifies the process flow. This simplicity translates directly into lower labor costs, reduced solvent consumption, and decreased waste disposal expenses, providing a compelling economic argument for switching to this new material platform.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, such as substituted bromobenzenes, boronic acids, and common solvents like p-xylene and toluene, are commodity chemicals with robust global supply chains. This abundance ensures that production is not bottlenecked by the availability of niche precursors. For supply chain heads, this means greater predictability in lead times and reduced vulnerability to market fluctuations. The ability to source starting materials from multiple vendors further strengthens supply security, ensuring continuous production schedules even in the face of regional disruptions or logistical challenges.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, with reaction conditions that are easily transferable from flask to reactor. The use of aqueous workups and standard organic solvents aligns well with existing environmental, health, and safety (EHS) protocols in modern chemical plants. Moreover, the high atom economy of the coupling reactions minimizes the generation of hazardous byproducts. This environmental friendliness not only simplifies regulatory compliance but also aligns with the growing corporate sustainability goals of major electronics manufacturers, making these materials a preferred choice for green supply chain initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these spiro nitrogen-containing compounds in OLED device fabrication. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on performance expectations and integration requirements.
Q: How does the spiro structure improve OLED device performance?
A: The spiro nitrogen-containing derivative serves as a parent nucleus where the nitrogen-containing heterocycle breaks molecular symmetry. This structural feature effectively avoids intermolecular aggregation and crystallization, resulting in superior film-forming properties and enhanced electron transport performance compared to conventional planar molecules.
Q: What are the key advantages of this synthesis route for large-scale production?
A: The synthesis utilizes robust palladium-catalyzed coupling reactions and standard acid-mediated cyclization steps that are well-suited for industrial scale-up. The process employs commercially available solvents like p-xylene and toluene, and the intermediates can be purified via standard recrystallization techniques, ensuring high purity (>99.7%) suitable for electronic applications.
Q: Can these compounds replace traditional electron transport materials like Alq3?
A: Yes, experimental data indicates that devices utilizing these spiro nitrogen-containing compounds exhibit significantly reduced driving voltage (by approximately 1.2-1.8V) and improved luminous efficiency compared to devices using Tris(8-hydroxyquinoline) aluminum (Alq3). Furthermore, they demonstrate extended operational lifetimes, making them a superior alternative for next-generation displays.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro Nitrogen Compound Supplier
As the demand for high-performance OLED materials continues to surge, partnering with a technically proficient manufacturer is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this technological shift, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that ensure every batch of spiro nitrogen-containing compound meets the exacting standards of the optoelectronics industry. We understand that consistency is key in display manufacturing, and our advanced process control systems guarantee lot-to-lot reproducibility that your production lines depend on.
We invite you to engage with our technical procurement team to discuss how these advanced materials can optimize your device architecture. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of integrating our spiro nitrogen derivatives into your supply chain. We are prepared to provide specific COA data and route feasibility assessments tailored to your unique application needs, ensuring a seamless transition from evaluation to full-scale adoption. Let us collaborate to drive the next generation of display technology forward with materials that deliver both performance and value.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
