Advanced Synthesis of Potent Antitumor Benzophenone Derivatives for Commercial Scale-up
Advanced Synthesis of Potent Antitumor Benzophenone Derivatives for Commercial Scale-up
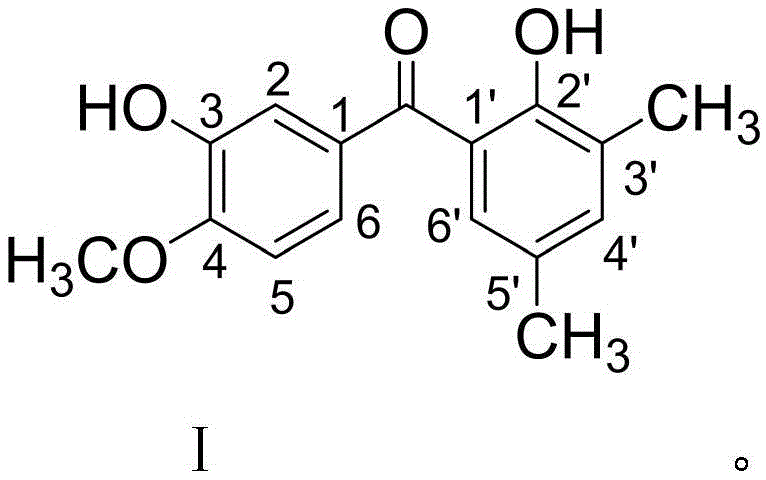
The pharmaceutical landscape is constantly evolving with the discovery of novel scaffolds that offer improved therapeutic indices against resistant malignancies. Patent CN111333495B introduces a significant breakthrough in this domain by disclosing a new benzophenone derivative, specifically (4-methoxy-3-hydroxyphenyl)(3,5-dimethyl-2-hydroxyphenyl)methanone. This compound represents a distinct structural class within the broader family of diphenyl ketones, characterized by specific substitution patterns that enhance its biological profile. Unlike conventional benzophenones which may exhibit limited solubility or metabolic instability, this novel molecule incorporates strategic hydroxyl and methoxy groups that optimize its interaction with cellular targets. The structural integrity and specific substitution pattern are critical for its observed broad-spectrum antitumor activity, ranging from leukemia to various solid tumors including lung and liver cancers.
For R&D directors evaluating new leads, the significance of this molecule lies not only in its potency but also in the robustness of its synthetic accessibility. The patent details a preparation method that circumvents many of the pitfalls associated with traditional Friedel-Crafts acylations. By leveraging a specific reagent system, the inventors have achieved a balance between reaction efficiency and product purity. This is particularly vital for a reliable pharmaceutical intermediates supplier, as the ability to consistently produce high-purity material is the cornerstone of drug development. The compound's demonstrated efficacy against taxol-resistant strains further underscores its potential as a next-generation therapeutic agent, offering a new avenue for overcoming multidrug resistance in clinical settings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted benzophenones has relied heavily on classical Friedel-Crafts acylation protocols utilizing strong Lewis acids such as aluminum chloride (AlCl3) or zinc chloride. While these methods are well-documented, they suffer from severe drawbacks that hinder their application in modern green chemistry and large-scale manufacturing. The primary issue is the stoichiometric requirement of the Lewis acid, which often exceeds one equivalent relative to the substrate, leading to the generation of massive quantities of hazardous waste upon hydrolysis. Furthermore, the workup procedures for these reactions are notoriously cumbersome, involving ice-water quenches that produce corrosive acidic sludge, posing significant environmental and safety challenges. Additionally, the harsh conditions often required can lead to side reactions, such as demethylation or polymerization, which complicate the impurity profile and necessitate extensive purification efforts that drive up costs in API manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in patent CN111333495B employs Eaton's reagent, a powerful yet manageable system composed of phosphorus pentoxide dissolved in methanesulfonic acid. This approach represents a paradigm shift towards more sustainable and efficient synthesis. The liquid nature of the reagent system allows for better heat transfer and mixing compared to heterogeneous Lewis acid slurries. Crucially, this method operates under milder thermal conditions, typically around 90°C, which minimizes thermal degradation of sensitive functional groups like phenols and methoxy ethers. For procurement managers focused on cost reduction in pharmaceutical intermediate manufacturing, this translates to a streamlined process that reduces the burden on waste treatment facilities and lowers the consumption of auxiliary chemicals. The simplicity of the workup, involving a straightforward precipitation and extraction, ensures a higher overall yield and a cleaner crude product, facilitating easier downstream processing.
Mechanistic Insights into Eaton Reagent-Catalyzed Acylation
The mechanistic pathway for this transformation is a sophisticated example of electrophilic aromatic substitution facilitated by superacidic conditions. In the Eaton's reagent system, phosphorus pentoxide acts as a potent dehydrating agent that activates the carboxylic acid moiety of the 4-methoxy-3-hydroxybenzoic acid. This activation leads to the formation of a highly reactive acylium ion species or a mixed anhydride intermediate stabilized within the methanesulfonic acid matrix. This electrophile is sufficiently potent to attack the electron-rich aromatic ring of the 2,4-dimethylphenol substrate. The presence of the hydroxyl group on the phenol ring directs the incoming acyl group to the ortho position relative to the hydroxyl, driven by strong electronic activation and hydrogen bonding interactions that pre-organize the transition state. This regioselectivity is paramount for ensuring the correct structural isomer is formed, as alternative substitution patterns would lack the desired biological activity.
From an impurity control perspective, the mechanism offers inherent advantages. The mild acidity of the system compared to molten Lewis acids prevents the cleavage of the methyl ether bond, a common side reaction in harsher environments. Furthermore, the solubility of the intermediates in the methanesulfonic acid phase helps to suppress intermolecular condensation reactions that could lead to oligomeric byproducts. For quality control teams, understanding this mechanism is essential for defining critical process parameters (CPPs). Maintaining the precise ratio of phosphorus pentoxide to methanesulfonic acid ensures that the concentration of the active acylium species remains optimal throughout the reaction, preventing the accumulation of unreacted starting materials which can be difficult to separate due to similar polarity. This deep mechanistic understanding allows for the design of robust control strategies that guarantee batch-to-batch consistency.
How to Synthesize (4-methoxy-3-hydroxyphenyl)(3,5-dimethyl-2-hydroxyphenyl)methanone Efficiently
The practical execution of this synthesis is designed for operational simplicity without compromising on yield or purity. The process begins with the careful preparation of the catalytic medium, where phosphorus pentoxide is dissolved in methanesulfonic acid under heating to ensure complete homogenization. Once the reagent is prepared, the temperature is carefully modulated to the optimal reaction window before the substrates are introduced. This sequential addition is critical to prevent exothermic spikes that could degrade the reagents. Following the reaction period, the quenching step utilizes water to precipitate the product, leveraging the poor solubility of the benzophenone derivative in aqueous media to achieve a preliminary separation from the acid matrix.
- Prepare Eaton's reagent by dissolving phosphorus pentoxide in methanesulfonic acid at 110°C with stirring until a homogeneous solution is obtained.
- Cool the reagent to 90°C and add equimolar amounts of 4-methoxy-3-hydroxybenzoic acid and 2,4-dimethylphenol, maintaining the temperature for reaction.
- Quench the reaction mixture into water, extract the viscous precipitate with ethyl acetate, and purify the crude product via medium-pressure preparative chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical curiosity. The shift away from traditional Lewis acid catalysis fundamentally alters the cost structure of production. By eliminating the need for stoichiometric amounts of metal salts, the process removes the necessity for expensive chelating resins or complex extraction protocols typically required to reduce metal residues to ppm levels acceptable for pharmaceutical use. This simplification directly correlates to substantial cost savings in raw material consumption and waste disposal fees. Moreover, the use of methanesulfonic acid, a relatively benign and biodegradable organic acid, aligns with increasingly stringent environmental regulations, reducing the risk of compliance-related shutdowns or fines.
- Cost Reduction in Manufacturing: The elimination of heavy metal catalysts significantly reduces the complexity of the purification train. Traditional methods often require multiple washes and specialized scavengers to remove aluminum or zinc residues, which adds time and material costs. In this novel process, the absence of such metals means the purification can focus solely on organic impurities, allowing for more efficient chromatographic or crystallization steps. This streamlined workflow results in a lower cost of goods sold (COGS) and improves the overall margin profile for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, namely 4-methoxy-3-hydroxybenzoic acid and 2,4-dimethylphenol, are commercially available commodity chemicals with stable supply chains. Unlike exotic catalysts that may be sourced from single suppliers with long lead times, these precursors can be procured from multiple global vendors. This redundancy mitigates the risk of supply disruptions and provides procurement managers with greater leverage in negotiations. Furthermore, the robustness of the reaction conditions means that production schedules are less likely to be impacted by equipment failures related to corrosion or extreme temperature requirements.
- Scalability and Environmental Compliance: The process is inherently scalable due to the liquid-phase nature of the reaction medium, which facilitates efficient heat and mass transfer in large reactors. The moderate operating temperature of 90°C is easily achievable with standard steam heating systems found in most multipurpose chemical plants, removing the need for specialized cryogenic or high-pressure equipment. From an environmental standpoint, the reduction in hazardous waste generation simplifies the permitting process for new manufacturing lines. The ability to recycle or treat the methanesulfonic acid waste stream more easily than heavy metal sludge contributes to a smaller environmental footprint, supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel benzophenone derivative. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for partners looking to integrate this intermediate into their drug discovery pipelines or commercial manufacturing processes.
Q: What represents the primary advantage of using Eaton's reagent over traditional Lewis acids for this synthesis?
A: The use of Eaton's reagent (P2O5/MSA) eliminates the need for stoichiometric amounts of corrosive Lewis acids like aluminum chloride, thereby avoiding the generation of massive quantities of acidic sludge and simplifying the downstream purification process significantly.
Q: Which specific tumor cell lines show the highest sensitivity to this novel benzophenone compound?
A: Experimental data indicates exceptional potency against leukemia HL-60 and liver cancer SMMC-7721 cell lines, with IC50 values as low as 0.111 μM, demonstrating superior activity compared to standard chemotherapeutic agents like cisplatin in specific assays.
Q: Is the synthetic route described in patent CN111333495B suitable for industrial scale-up?
A: Yes, the process operates at moderate temperatures (90°C) without requiring cryogenic conditions or exotic catalysts, making it highly amenable to large-scale production while maintaining high purity specifications through standard chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (4-methoxy-3-hydroxyphenyl)(3,5-dimethyl-2-hydroxyphenyl)methanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of novel scaffolds like the benzophenone derivative described in CN111333495B. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for oncology drug candidates. We understand that consistency is key in pharmaceutical development, and our process engineering team is dedicated to optimizing every step of the synthesis to maximize yield and minimize impurities.
We invite you to collaborate with us to unlock the full commercial potential of this compound. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Whether you need specific COA data for your regulatory filings or detailed route feasibility assessments for process validation, we are committed to delivering the transparency and expertise you need. Contact us today to discuss how we can support your supply chain with high-quality, cost-effective pharmaceutical intermediates.
