Advanced Synthesis of Potent Antitumor Benzophenone Intermediates for Commercial Scale-Up
Advanced Synthesis of Potent Antitumor Benzophenone Intermediates for Commercial Scale-Up
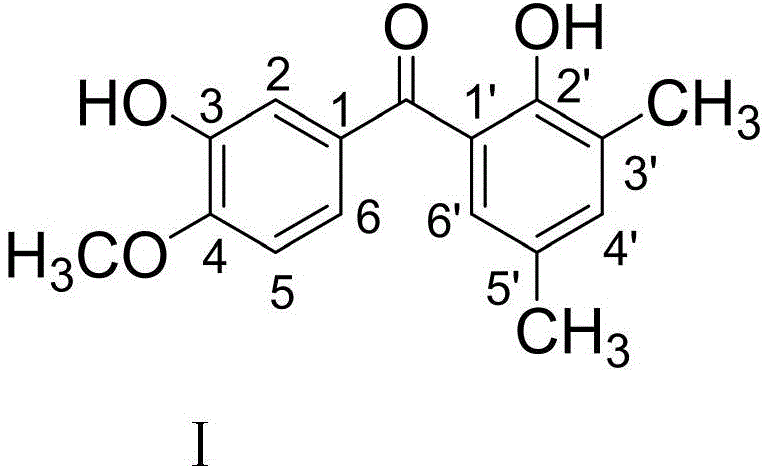
The pharmaceutical landscape is constantly evolving with the discovery of novel scaffolds that offer improved therapeutic indices against resistant malignancies. A significant breakthrough in this domain is documented in patent CN111333495A, which discloses a new benzophenone derivative, specifically (4-methoxy-3-hydroxyphenyl)(3,5-dimethyl-2-hydroxyphenyl)methanone. This compound represents a critical advancement in the field of biochemical medicine, exhibiting remarkable antitumor properties across a diverse panel of human cancer cell lines. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediates supplier partnerships, understanding the synthetic accessibility and biological potential of this molecule is paramount. The patent outlines a robust preparation method that leverages Eaton's reagent to achieve high selectivity and operational simplicity, addressing common bottlenecks in complex ketone synthesis.
The strategic value of this technology lies not only in the biological efficacy of the final product but also in the elegance of its manufacturing process. Unlike traditional methods that often rely on harsh conditions or expensive catalysts, this approach utilizes a modified Friedel-Crafts acylation strategy that is both cost-effective and environmentally considerate. By integrating this novel intermediate into your pipeline, organizations can achieve substantial cost reduction in pharmaceutical intermediates manufacturing while securing a supply chain for next-generation oncology therapeutics. The following analysis dissects the technical merits, mechanistic insights, and commercial viability of this invention, providing a comprehensive roadmap for its adoption in commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted benzophenones has been plagued by significant technical challenges that hinder efficient commercial scale-up. Conventional Friedel-Crafts acylation protocols frequently employ stoichiometric amounts of strong Lewis acids such as aluminum chloride (AlCl3) or zinc chloride. These reagents, while effective in promoting the reaction, generate substantial quantities of hazardous waste and require rigorous quenching procedures that complicate downstream processing. Furthermore, the heterogeneous nature of these reactions often leads to poor heat transfer, resulting in localized hot spots that can degrade sensitive functional groups like hydroxyls and methoxyls. The workup typically involves acidic hydrolysis of the Lewis acid complex, producing large volumes of aluminum sludge that pose severe environmental disposal issues and increase the overall E-factor of the process.
Additionally, achieving high regioselectivity in the acylation of polysubstituted phenols is notoriously difficult using standard conditions. The presence of multiple activating groups on the aromatic rings can lead to a mixture of ortho- and para-substituted isomers, necessitating complex and yield-loss-inducing purification steps such as repeated recrystallization or preparative HPLC. For a reliable pharmaceutical intermediates supplier, these inefficiencies translate directly into higher production costs and longer lead times. The inability to consistently produce high-purity material without extensive chromatographic intervention has traditionally limited the availability of such specialized benzophenone derivatives for preclinical and clinical evaluation.
The Novel Approach
The methodology described in patent CN111333495A offers a transformative solution to these longstanding problems by utilizing Eaton's reagent, a homogeneous mixture of phosphorus pentoxide (P2O5) and methanesulfonic acid (MSA). This system acts as a powerful yet controllable dehydrating and acylating agent that facilitates the direct condensation of 4-methoxy-3-hydroxybenzoic acid with 2,4-dimethylphenol. The reaction proceeds under remarkably mild thermal conditions, specifically at 90°C, which preserves the integrity of the sensitive hydroxyl and methoxy substituents. The homogeneity of the reaction medium ensures uniform heating and mixing, drastically reducing the formation of side products and improving the overall yield of the desired ketone.
Moreover, the workup procedure is significantly streamlined compared to traditional Lewis acid methods. Upon completion, the reaction mixture is simply poured into water, causing the product to precipitate as a viscous substance that can be easily extracted with organic solvents like ethyl acetate. This eliminates the need for complex neutralization steps and heavy metal waste treatment. The resulting crude product is of sufficient quality to be purified efficiently using medium pressure preparative chromatography, yielding the target compound as a dark green solid with a defined melting point of 101-104°C. This streamlined workflow exemplifies how modern process chemistry can deliver high-purity pharmaceutical intermediates with reduced operational complexity and environmental impact.
Mechanistic Insights into Eaton Reagent-Catalyzed Acylation
The success of this synthesis hinges on the unique mechanistic pathway facilitated by the P2O5/MSA system. In this environment, methanesulfonic acid serves as both the solvent and a proton source, while phosphorus pentoxide acts as a potent dehydrating agent. The reaction initiates with the protonation of the carboxylic acid group of 4-methoxy-3-hydroxybenzoic acid, followed by dehydration to form a highly reactive acylium ion intermediate. This electrophilic species is stabilized by the sulfonate anions in the medium, preventing premature decomposition. The acylium ion then undergoes electrophilic aromatic substitution with the electron-rich 2,4-dimethylphenol ring. The hydroxyl and methyl groups on the phenol ring act as strong ortho/para directors, guiding the attack of the acylium ion specifically to the position ortho to the hydroxyl group, thereby ensuring the formation of the desired 2-hydroxy-3,5-dimethyl substitution pattern.
From an impurity control perspective, the mild acidity of the Eaton's reagent system is crucial. Stronger Lewis acids often promote polyacylation or Fries rearrangement side reactions, especially at elevated temperatures. By maintaining the reaction at 90°C and utilizing a controlled acidic environment, the process minimizes these competing pathways. The specific molar ratio of 1:1 between the benzoic acid and the phenol further suppresses the formation of di-acylated byproducts. This precise control over the reaction kinetics allows for the generation of a clean crude profile, which is essential for meeting the stringent purity specifications required for commercial scale-up of complex pharmaceutical intermediates. The mechanism underscores the importance of reagent selection in optimizing both yield and selectivity in fine chemical synthesis.
How to Synthesize (4-Methoxy-3-hydroxyphenyl)(3,5-dimethyl-2-hydroxyphenyl)methanone Efficiently
Implementing this synthesis route requires careful attention to the preparation of the Eaton's reagent and the thermal profile of the reaction. The process begins with the dissolution of phosphorus pentoxide in methanesulfonic acid at 110°C to ensure complete homogenization before cooling to the reaction temperature. The subsequent addition of the substrates must be managed to maintain the exotherm within safe limits. Detailed standardized operating procedures regarding stoichiometry, agitation speeds, and quenching protocols are critical for reproducibility. For a comprehensive breakdown of the exact experimental parameters and safety considerations, please refer to the step-by-step guide below.
- Preparation of Eaton's Reagent by dissolving phosphorus pentoxide in methanesulfonic acid at 110°C with stirring until a homogeneous solution is obtained.
- Cooling the reagent to 90°C and adding equimolar amounts of 4-methoxy-3-hydroxybenzoic acid and 2,4-dimethylphenol for the acylation reaction.
- Quenching the reaction mixture into water, extracting the viscous product with ethyl acetate, and purifying via medium pressure preparative chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that extend beyond mere technical feasibility. The elimination of expensive transition metal catalysts and the reduction in hazardous waste generation directly correlate to a leaner cost structure. By adopting a process that relies on commodity chemicals like methanesulfonic acid and phosphorus pentoxide, manufacturers can insulate themselves from the volatility of precious metal markets. Furthermore, the simplified workup reduces the consumption of solvents and water, aligning with global sustainability goals and reducing the burden on wastewater treatment facilities. These factors collectively contribute to a more resilient and cost-efficient supply chain for critical oncology ingredients.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of stoichiometric Lewis acids with a recyclable acidic medium. Traditional methods often require more than one equivalent of aluminum chloride, generating massive amounts of solid waste that incurs high disposal fees. In contrast, the Eaton's reagent method produces a liquid waste stream that is easier to manage and treat. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, effectively increasing the atom economy. This efficiency translates to a lower cost per kilogram of the active intermediate, allowing for more competitive pricing in the final drug formulation.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, 4-methoxy-3-hydroxybenzoic acid and 2,4-dimethylphenol, are commercially available bulk chemicals with stable supply chains. Unlike specialized organometallic reagents that may have long lead times or single-source dependencies, these precursors can be sourced from multiple global suppliers. This redundancy mitigates the risk of supply disruptions and ensures continuous production capability. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, further enhancing supply chain stability and reducing the frequency of batch failures.
- Scalability and Environmental Compliance: Scaling this process from laboratory to pilot and eventually to commercial production is straightforward due to the absence of extreme conditions. The reaction temperature of 90°C is well within the operating range of standard glass-lined or stainless steel reactors, eliminating the need for specialized cryogenic or high-pressure equipment. The mild nature of the reagents also simplifies compliance with increasingly stringent environmental regulations regarding heavy metal discharge. By minimizing the use of toxic metals and reducing solvent intensity through efficient extraction, this route supports the manufacture of high-purity pharmaceutical intermediates in an environmentally responsible manner, facilitating faster regulatory approval.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel benzophenone derivative. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the fit of this intermediate within your specific drug development portfolio.
Q: What are the key advantages of using Eaton's reagent over traditional Lewis acids for this benzophenone synthesis?
A: Eaton's reagent (P2O5/MSA) offers a homogeneous reaction medium that facilitates better heat transfer and regioselectivity compared to heterogeneous Lewis acids like AlCl3. It operates under milder conditions (90°C) and simplifies the workup process by avoiding the formation of difficult-to-remove aluminum sludge, thereby reducing waste treatment costs.
Q: What is the biological activity profile of this novel compound?
A: The compound demonstrates potent antitumor activity against a broad spectrum of human cancer cell lines, including leukemia (HL-60), liver cancer (SMMC-7721, HepG2), and breast cancer (MCF-7). Notably, its IC50 values against several lines are lower than those of cisplatin, indicating superior inhibitory potency.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the process utilizes readily available starting materials and avoids expensive transition metal catalysts. The reaction temperature is moderate (90-110°C), which is compatible with standard stainless steel reactors, and the purification via chromatography can be adapted to large-scale flash column techniques.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (4-Methoxy-3-hydroxyphenyl)(3,5-dimethyl-2-hydroxyphenyl)methanone Supplier
The synthesis of (4-methoxy-3-hydroxyphenyl)(3,5-dimethyl-2-hydroxyphenyl)methanone represents a significant opportunity for advancing antitumor drug candidates, and NINGBO INNO PHARMCHEM is uniquely positioned to support this journey. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from benchtop discovery to market supply. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for clinical trials and commercial manufacturing.
We invite you to engage with our technical team to explore how this optimized route can enhance your project's economics and timeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific financial benefits of adopting this methodology for your supply chain. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a partnership built on transparency and technical excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
