Advanced Manufacturing of Sulfadoxine: Optimizing Purity and Cost Efficiency for Global Supply Chains
Advanced Manufacturing of Sulfadoxine: Optimizing Purity and Cost Efficiency for Global Supply Chains
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with economic viability, particularly for established therapeutic classes like sulfonamides. Patent CN102304095B introduces a transformative preparation method for Sulfadoxine (4-(p-amino benzene sulfonyl)-5,6-dimethoxypyridin), addressing critical bottlenecks in the traditional five-step synthesis. This technology focuses on rigorous impurity control, the elimination of toxic catalysts, and the optimization of reagent states to enhance scalability. For R&D directors and procurement managers, this patent represents a significant opportunity to upgrade production lines, offering a pathway to reduce raw material consumption while simultaneously improving the environmental profile of the manufacturing process.
The core innovation lies in the precise manipulation of reaction conditions across all five stages, from the initial Claisen condensation to the final methoxylation. By shifting from liquid to solid reagents in key steps and avoiding acidic workups that cause equipment corrosion, the process ensures a more stable and continuous supply chain. This report analyzes the technical depth of this methodology, highlighting its potential for cost reduction in sulfa drug manufacturing and its alignment with modern green chemistry principles required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of Sulfadoxine has been plagued by inefficiencies that compromise both yield and equipment longevity. In the initial condensation step, conventional techniques often result in intermediates with purity levels below 92%, necessitating complex fractional distillation that increases energy consumption and lead time. Furthermore, the cyclization step traditionally isolates the cyclocompound under acidic conditions, leading to a product that exists in equilibrium between hydroxyl and hydroxyl sodium salt forms. This results in a hygroscopic material that is difficult to dry and requires extensive mother liquor recycling, which accumulates salts and degrades quality. Additionally, the reliance on N,N-dimethylaniline as a catalyst in the chlorination step introduces toxic residues that are difficult to remove, often imparting undesirable color to the final product and requiring expensive recovery systems.
The Novel Approach
The methodology outlined in CN102304095B fundamentally restructures these unit operations to maximize efficiency. The process utilizes solid sodium ethylate in the first step, maintaining temperatures below 65°C to prevent side reactions, followed by a streamlined vacuum fractionation that yields intermediates with purity ≥95%. Crucially, the cyclization step is modified to isolate the cyclocompound directly as an anhydrous hydroxy sodium salt by evaporating solvents under alkaline conditions, bypassing the corrosive acidic workup entirely. This shift not only protects reactor integrity but also simplifies drying. In the chlorination phase, the process remarkably eliminates the need for N,N-dimethylaniline, relying instead on optimized thermal conditions with phosphorus oxychloride to achieve high conversion rates. Finally, the substitution of sodium methoxide solution with solid sodium hydroxide in the methoxylation step prevents local superheating, ensuring a high-quality crude product with excellent color properties.
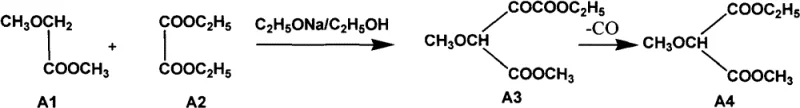
Mechanistic Insights into Optimized Heterocyclic Synthesis
The chemical elegance of this route is best understood through the lens of nucleophilic acyl substitution and heterocyclic ring formation. The initial step involves a Claisen condensation between methyl methoxyacetate and diethyl oxalate, facilitated by the strong base sodium ethylate. The resulting beta-keto ester undergoes decarbonylation, a critical transformation that establishes the carbon skeleton for the pyrimidine ring. The strict control of temperature and the use of anhydrous conditions are paramount here, as moisture can hydrolyze the esters prematurely, leading to yield loss. The subsequent cyclization with formamide creates the pyrimidine core. By maintaining an alkaline environment throughout the workup, the process stabilizes the intermediate as a sodium salt, preventing the tautomerization issues seen in acidic media which often trap impurities within the crystal lattice.
In the chlorination stage, the mechanism proceeds via nucleophilic attack of the chloride ion (generated from POCl3) on the hydroxyl groups of the pyrimidine ring. The absence of an amine catalyst suggests that the reaction kinetics are sufficiently driven by the high concentration of POCl3 and elevated temperatures (70°C–120°C), avoiding the formation of quaternary ammonium salts that complicate purification. The final methoxylation is a classic nucleophilic aromatic substitution where the methoxide ion displaces the chlorine atom at the 6-position. The use of solid NaOH in methanol generates the methoxide in situ but in a controlled manner that avoids the exothermic spikes associated with adding pre-formed sodium methoxide solutions, thereby preserving the integrity of the sensitive sulfonamide linkage.
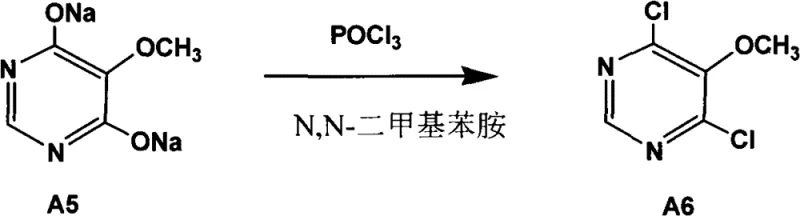
How to Synthesize Sulfadoxine Efficiently
Implementing this synthesis requires precise adherence to the stoichiometric ratios and thermal profiles defined in the patent embodiments. The process begins with the careful addition of solid sodium ethylate to a mixture of esters, followed by a controlled decarbonylation under vacuum. The subsequent cyclization demands efficient solvent removal to force the precipitation of the sodium salt. For the chlorination and condensation steps, moisture control is critical to prevent hydrolysis of the reactive chloro-intermediates. The detailed standardized operating procedures for scaling this route from laboratory to commercial production are provided in the technical guide below.
- Condense methyl methoxyacetate with excess diethyl oxalate using solid sodium ethylate, followed by decarbonylation to obtain high-purity 2-methoxy-methylethyl malonate.
- React the malonate derivative with formamide and sodium methylate to form the cyclocompound as an anhydrous hydroxy sodium salt.
- Perform chlorination using phosphorus oxychloride without N,N-dimethylaniline catalyst to generate 4,6-dichloro-5-methoxy pyrimidine.
- Condense the dichloro-pyrimidine with sulfanilamide sodium to introduce the sulfonamide group.
- Complete the synthesis via methoxylation using solid sodium hydroxide instead of sodium methoxide solution to ensure thermal stability and high yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible strategic benefits beyond mere technical superiority. The elimination of specific catalysts and the simplification of purification steps directly translate to a leaner manufacturing footprint. By removing the need for N,N-dimethylaniline recovery systems, facilities can reduce capital expenditure on specialized distillation columns and waste treatment units. Furthermore, the use of solid reagents like sodium ethylate and sodium hydroxide simplifies logistics and storage compared to handling large volumes of hazardous liquid alkoxide solutions, thereby enhancing operational safety and reducing insurance liabilities associated with chemical transport.
- Cost Reduction in Manufacturing: The process achieves significant cost savings primarily through the optimization of raw material utilization and the reduction of auxiliary chemical consumption. By avoiding the use of expensive amine catalysts and eliminating the complex recovery processes associated with them, the overall variable cost per kilogram of product is substantially lowered. Additionally, the higher purity of intermediates reduces the load on downstream purification, meaning less solvent and energy are required for recrystallization and washing, leading to a more economically efficient production cycle.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to greater supply continuity. Traditional methods often suffer from batch-to-batch variability due to the sensitivity of the acidic cyclization step; the new alkaline method produces a stable, free-flowing solid intermediate that is easier to store and transport between production stages. This stability minimizes the risk of production delays caused by off-spec materials, ensuring that delivery schedules for high-purity sulfadoxine can be met consistently, even during periods of high market demand.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns with increasingly stringent global regulations regarding waste discharge. The reduction in acidic wastewater from the cyclization step and the elimination of toxic amine residues simplify effluent treatment. The patent notes that phosphoric acid byproducts from the chlorination step can be converted into phosphate fertilizers, turning a waste stream into a value-added co-product. This circular approach facilitates easier permitting for commercial scale-up of complex pharmaceutical intermediates and strengthens the company's sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy technologies in terms of yield, purity, and operational safety.
Q: Why is the elimination of N,N-dimethylaniline significant in the chlorination step?
A: Traditional methods require N,N-dimethylaniline as a catalyst, which necessitates complex recovery processes and generates significant waste. The novel process achieves high conversion (>98.5% content) without this catalyst, drastically simplifying post-treatment and reducing environmental burden.
Q: How does the new method improve the purity of the intermediate in Step 1?
A: By utilizing solid sodium ethylate and controlling the reaction temperature below 65°C, followed by vacuum fractionation to remove low-boiling substances, the purity of 2-methoxy-methylethyl malonate is strictly controlled to be ≥95%, compared to <92% in traditional methods.
Q: What are the advantages of using solid sodium hydroxide in the final methoxylation step?
A: Replacing sodium methoxide solution with solid sodium hydroxide prevents local overheating and thermal decomposition during solvent recovery. This results in a crude product with superior color and luster, significantly increasing the melting point and overall quality of the final Sulfadoxine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfadoxine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN102304095B are fully realized in practice. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Sulfadoxine intermediate meets the exacting standards required by global regulatory agencies.
We invite pharmaceutical partners to collaborate with us to leverage this optimized technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project benefits from the highest standards of quality and efficiency in the industry.
