Revolutionizing Ergothioneine Production: Advanced EanB Mutants for Commercial Scale-Up
Revolutionizing Ergothioneine Production: Advanced EanB Mutants for Commercial Scale-Up
The global demand for high-purity ergothioneine, a potent natural antioxidant with significant therapeutic potential, has driven intense research into more efficient manufacturing methodologies. A pivotal advancement in this field is detailed in patent CN114667346A, which discloses novel EanB enzyme mutants derived from Chlorobium limicola. This technology represents a paradigm shift from traditional fungal fermentation to precise enzymatic catalysis, offering a robust pathway for the reliable ergothioneine supplier market. The core innovation lies in the engineering of oxidative sulfurase variants that exhibit dramatically improved catalytic efficiency, enabling the direct conversion of N(alpha)-trimethylhistidine into ergothioneine with superior kinetics.
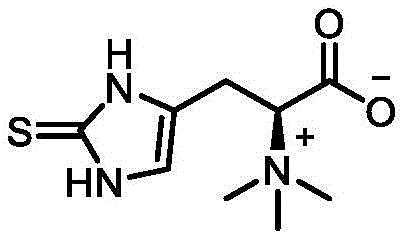
Understanding the molecular architecture of the target compound is essential for appreciating the synthetic challenge. As illustrated in the structural diagram, ergothioneine is a unique 2-thioimidazole derivative of histidine, characterized by a betaine structure that contributes to its stability and bioavailability. The synthesis of this specific zwitterionic structure requires the precise formation of a carbon-sulfur bond at the epsilon position of the imidazole ring. Conventional chemical synthesis often struggles with regioselectivity and the introduction of toxic heavy metal impurities, while biological extraction from mushrooms is limited by low yields and seasonal variability. The enzymatic approach described in the patent addresses these fundamental bottlenecks by leveraging the specificity of protein catalysts to achieve clean, high-yield transformations under mild conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of ergothioneine has relied heavily on the submerged fermentation of edible mushroom mycelia or extraction from natural sources. While biologically native, this approach suffers from inherent inefficiencies that hinder cost reduction in antioxidant manufacturing. The primary drawback is the extremely slow growth rate of mushroom mycelia, which typically requires a fermentation period of 7 to 10 days to accumulate sufficient biomass. Furthermore, the ergothioneine produced is largely intracellular, necessitating energy-intensive downstream processing steps such as mycelial crushing, solid-liquid separation, and complex extraction protocols to release the product from the cell wall matrix. These factors collectively result in high production costs, long lead times, and significant batch-to-batch variability, making it difficult to secure a consistent supply of high-purity ergothioneine for sensitive pharmaceutical applications.
The Novel Approach
In stark contrast, the novel approach utilizing EanB enzyme mutants offers a streamlined, single-step biocatalytic solution that bypasses the limitations of whole-organism fermentation. By isolating the specific oxidative sulfurase responsible for C-S bond formation and enhancing its activity through protein engineering, the process decouples product synthesis from cellular growth constraints. The patent describes a method where the mutant enzyme catalyzes the formation of ergothioneine from N(alpha)-trimethylhistidine in a reaction timeframe as short as 24 hours. This drastic reduction in processing time, combined with the ability to use recombinant E. coli as a highly efficient expression host, allows for a continuous or semi-continuous production model. The elimination of complex mycelial disruption steps significantly simplifies the purification workflow, thereby enhancing the overall economic viability and scalability of the process for commercial scale-up of complex amino acid derivatives.
Mechanistic Insights into EanB-Catalyzed C-S Bond Formation
The core of this technological breakthrough lies in the rational design and directed evolution of the EanB oxidative sulfurase. The wild-type enzyme, while functional, possesses kinetic limitations that restrict its utility in high-throughput industrial settings. The patent details a sophisticated protein engineering strategy involving error-prone PCR to generate a diverse mutation library, followed by high-throughput screening to identify variants with superior catalytic properties. Key mutations identified, such as the substitution of Isoleucine at position 75 with Arginine (I75R) and Glutamic Acid at position 369 with Proline (E369P), induce conformational changes in the enzyme's active site. These structural modifications likely optimize the binding affinity for the sulfur donor and the histidine substrate, stabilizing the transition state and lowering the activation energy required for the sulfur transfer reaction.
From a mechanistic perspective, the EanB mutant facilitates an anaerobic or micro-aerophilic sulfur insertion, distinguishing it from other oxygen-dependent sulfurases. This characteristic is crucial for maintaining process robustness, as it reduces the sensitivity of the reaction to dissolved oxygen levels, a common variable in large-scale fermenters. The enhanced relative activity of the EanB-888 mutant, reported to be 4.8 times higher than the wild type, translates directly to process intensification. Higher specific activity means that less enzyme protein is required per unit of substrate, reducing the burden on the expression host and minimizing the presence of host cell proteins in the final reaction mixture. This purity profile is critical for reducing lead time for high-purity ergothioneine, as it alleviates the pressure on downstream chromatographic purification steps to remove biocatalyst residues.
How to Synthesize Ergothioneine Efficiently
The implementation of this enzymatic route requires a systematic approach to strain construction and bioprocess optimization. The patent outlines a clear workflow starting from gene synthesis to final product isolation, emphasizing the importance of selecting the right expression vector and host strain to maximize soluble protein yield. The process leverages standard molecular biology techniques that are well-understood in the industry, ensuring that the technology can be rapidly transferred from the laboratory to pilot and production scales. For detailed operational parameters, including specific buffer compositions, induction temperatures, and substrate feeding strategies, refer to the standardized synthesis guide below.
- Construct an EanB gene mutation library using error-prone PCR targeting the wild-type oxidative sulfurase sequence from Chlorobium limicola.
- Screen mutant libraries for enhanced catalytic activity, specifically identifying variants with mutations at positions 75 and 369 (e.g., I75R, E369P).
- Express the selected high-activity mutant in E. coli BL21(DE3) and catalyze the conversion of N(alpha)-trimethylhistidine to ergothioneine under optimized pH and temperature conditions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from mushroom fermentation to enzymatic synthesis offers profound strategic advantages beyond mere technical novelty. The shift fundamentally alters the cost structure and risk profile of ergothioneine sourcing. By moving to a recombinant enzyme system, manufacturers gain control over the production timeline, eliminating the biological unpredictability associated with fungal growth cycles. This reliability is paramount for securing long-term contracts with pharmaceutical clients who require guaranteed delivery schedules and consistent quality attributes. The ability to produce ergothioneine in a fraction of the time traditionally required allows for a more responsive supply chain capable of adapting to sudden spikes in market demand without the need for excessive inventory buffering.
- Cost Reduction in Manufacturing: The enzymatic process drives significant cost savings primarily through the intensification of production capacity and the simplification of downstream processing. By achieving high conversion rates within 24 hours, the facility throughput is drastically increased compared to the week-long cycles of mushroom fermentation. Furthermore, the use of soluble enzymes or permeabilized cells avoids the expensive and energy-consuming mechanical disruption of tough fungal cell walls. The elimination of heavy metal catalysts, often used in chemical synthesis, also removes the costly step of metal scavenging and validation, leading to substantial operational expenditure reductions while maintaining a green chemistry profile.
- Enhanced Supply Chain Reliability: Relying on agricultural products like mushrooms introduces vulnerabilities related to seasonality, climate change, and raw material quality fluctuations. In contrast, the enzymatic route utilizes defined chemical substrates and recombinant microbial strains that can be stored and propagated consistently year-round. This decoupling from agricultural supply chains ensures a stable and predictable flow of raw materials, mitigating the risk of production stoppages due to external environmental factors. The robustness of the E. coli expression system further guarantees that the biocatalyst itself can be produced reliably at scale, securing the continuity of supply for critical nutraceutical and pharmaceutical ingredients.
- Scalability and Environmental Compliance: The proposed biocatalytic method aligns perfectly with modern environmental, social, and governance (ESG) goals. The process operates under mild aqueous conditions, generating significantly less hazardous waste compared to traditional organic synthesis routes. The high specificity of the enzyme minimizes the formation of by-products, resulting in a cleaner crude product that requires fewer solvent-intensive purification steps. This reduction in solvent usage and waste generation not only lowers disposal costs but also simplifies regulatory compliance regarding environmental emissions. The scalability of bacterial fermentation is well-proven in the fine chemical industry, allowing for seamless expansion from pilot batches to multi-ton annual production capacities without fundamental process redesign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of EanB mutant technology for ergothioneine production. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route. Understanding these nuances is essential for R&D teams planning process validation and procurement teams assessing supplier capabilities.
Q: How does the EanB mutant improve upon wild-type enzyme performance?
A: The engineered EanB mutants, particularly those with I75R and E369P substitutions, demonstrate significantly higher relative enzymatic activity compared to the wild type. Patent data indicates up to a 4.8-fold increase in catalytic efficiency, allowing for faster conversion rates and reduced enzyme loading requirements in industrial reactors.
Q: What are the primary advantages of this enzymatic route over mushroom fermentation?
A: Traditional mushroom fermentation requires 7 to 10 days for mycelial growth and involves complex downstream processing to extract ergothioneine from cell walls. The enzymatic method described in CN114667346A utilizes soluble enzymes or whole-cell biocatalysts in a much shorter timeframe (approximately 24 hours for reaction), drastically simplifying purification and reducing overall production cycles.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process utilizes standard Escherichia coli expression systems (such as BL21(DE3)) which are well-established for industrial scalability. The use of defined substrates like N(alpha)-trimethylhistidine and robust enzyme mutants ensures consistent batch-to-batch quality, meeting the stringent purity specifications required for pharmaceutical and nutraceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ergothioneine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the EanB mutant technology in reshaping the ergothioneine market landscape. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial realities. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ergothioneine delivered meets the highest standards required for pharmaceutical and high-end nutraceutical applications. We are committed to leveraging advanced biocatalytic solutions to drive value for our global partners.
We invite you to collaborate with us to optimize your supply chain for this critical antioxidant. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to deep process expertise and the flexibility to adapt to evolving market needs. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments for your next project.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
