Advanced Synthetic Route for Ergothioneine: Enhancing Purity and Commercial Scalability
The pharmaceutical and nutraceutical industries are constantly seeking robust, scalable, and environmentally sustainable pathways for producing high-value antioxidants like ergothioneine. Patent CN102686568A introduces a groundbreaking synthetic method that addresses the critical limitations of prior art by utilizing a unique cleavage reaction of betaine-type compounds. This innovation shifts the paradigm from complex, multi-step protections involving hazardous reagents to a more direct, aqueous-based approach that maintains high stereochemical integrity. By leveraging a thiol-mediated scission at elevated temperatures, specifically greater than or equal to 60°C, this technology enables the efficient construction of the 2-thiocarbamoyl imidazole ring system found in ergothioneine and its analogs. For R&D directors and procurement specialists, this represents a significant opportunity to secure a reliable ergothioneine supplier capable of delivering material with superior purity profiles and reduced environmental footprint. The following analysis details how this proprietary chemistry translates into tangible commercial and technical advantages.
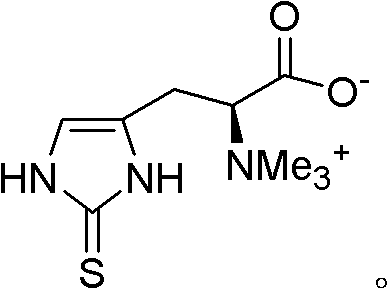
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of L-ergothioneine has been plagued by inefficient strategies that rely heavily on histidine or its N-dimethyl derivatives as starting materials. Prominent prior art, such as International Patent Application WO 95/00494, outlines a cumbersome five-step sequence that necessitates the protection of sulfur functionalities to prevent unwanted side reactions during methylation. This approach requires the use of thiophosgene, a reagent notorious for its extreme toxicity and difficulty in handling on a technical scale, posing severe safety risks and regulatory hurdles for cost reduction in antioxidant manufacturing. Furthermore, alternative routes described in documents like US 2009/093642 involve up to nine steps, utilizing highly toxic potassium thiocyanate in acidic media and large volumes of hydrochloric acid. These legacy methods accumulate significant defects, including the generation of hazardous waste streams, complex purification requirements, and low overall yields due to the attrition inherent in multi-step protection and deprotection cycles.
The Novel Approach
In stark contrast, the method disclosed in CN102686568A employs a biomimetic 'reverse' synthesis strategy that constructs the sulfur-containing imidazole ring from a pre-formed betaine skeleton. This innovative pathway eliminates the need for sulfur protection groups entirely, thereby streamlining the synthetic sequence and reducing the number of unit operations. By reacting a betaine-type compound of general formula (II) with a mercaptan in an aqueous solvent at temperatures between 60°C and 120°C, the process achieves a direct cleavage that forms the target thiocarbonyl functionality with high efficiency. This approach not only avoids the use of deleterious reagents like thiophosgene but also aligns with green chemistry principles by minimizing organic solvent usage. For supply chain heads, this translates to commercial scale-up of complex amino acid derivatives that is safer, faster, and more economically viable, ensuring a stable supply of high-purity ergothioneine for global markets.
Mechanistic Insights into Thiol-Mediated Cleavage Reaction
The core of this technological breakthrough lies in the specific cleavage reaction of the intermediate betaine compound in the presence of a thiol. The reaction mechanism involves the nucleophilic attack of the mercaptan on the intermediate structure, facilitating the scission of the side chain to reveal the desired 2-thione moiety. Crucially, the reaction is conducted in an aqueous medium, which enhances the solubility of hydrophilic mercaptans such as 3-thiohydracrylic acid or cysteine derivatives. The kinetic profile of this reaction is temperature-dependent, requiring heating above 60°C to overcome activation energy barriers, with optimal rates observed between 80°C and 100°C. This thermal requirement ensures complete conversion while maintaining the stability of the sensitive imidazole ring. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters for reducing lead time for high-purity nutraceutical ingredients, as precise temperature control directly impacts impurity profiles and final yield.
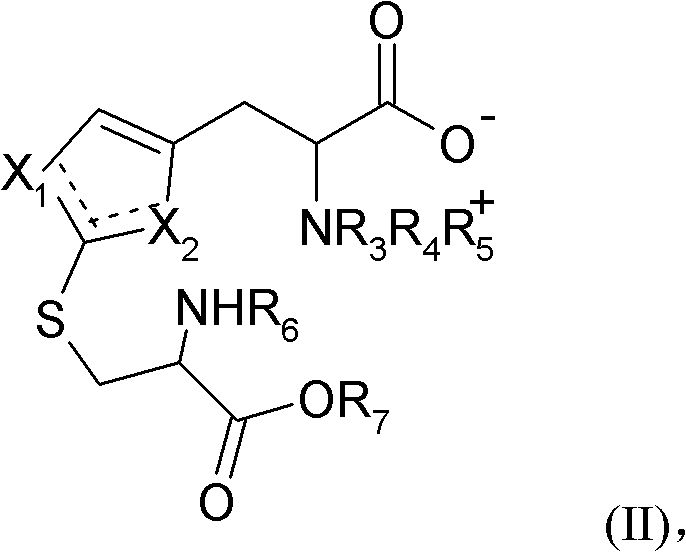
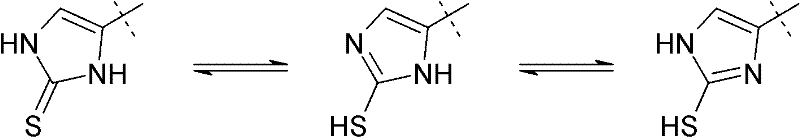
Furthermore, the structural integrity of the ergothioneine molecule is maintained through careful management of tautomeric equilibria. The 2-thiocarbamoyl imidazole ring can exist in multiple tautomeric forms, shifting between thione and thiol configurations depending on solvent pH and polarity. The patented method ensures that the thermodynamically stable thione form is favored in the final product, which is critical for biological activity. The use of specific mercaptans, preferably those with polar substituents like hydroxyl or carboxyl groups, aids in driving the equilibrium towards the desired product while facilitating downstream purification. This level of mechanistic control allows manufacturers to consistently meet stringent pharmacopeial standards, ensuring that the final API or intermediate is free from problematic isomers that could compromise efficacy or safety in downstream formulations.
How to Synthesize Ergothioneine Efficiently
The synthesis of ergothioneine via this patented route involves a sequential process starting from readily available betaine precursors. The initial step entails the reaction of a hercynine derivative with bromine at low temperatures, followed by coupling with a cysteine derivative to form the key intermediate. This intermediate is then subjected to the aforementioned thiol-mediated cleavage without the need for isolation, enabling a telescoped 'one-pot' procedure that maximizes efficiency. Detailed standard operating procedures for this synthesis, including specific molar ratios, temperature ramps, and workup protocols, are essential for successful technology transfer. The following guide outlines the critical stages required to implement this methodology in a GMP-compliant environment, ensuring reproducibility and quality control at every step of the manufacturing process.
- React a betaine-type compound (Formula III) with bromine at low temperature (-5°C to 5°C) followed by addition of a cysteine derivative to form the intermediate (Formula II).
- Perform a cleavage reaction on the intermediate (Formula II) in the presence of a mercaptan (e.g., 3-thiohydracrylic acid) at temperatures ≥60°C in an aqueous solvent.
- Separate and purify the final ergothioneine product (Formula I) from the reaction medium using techniques such as electrodialysis, resin treatment, or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits for procurement managers and supply chain leaders focused on resilience and cost efficiency. By eliminating the reliance on scarce and hazardous reagents like thiophosgene, the process mitigates supply risks associated with regulated chemicals and reduces the overhead costs related to safety compliance and waste disposal. The ability to conduct the reaction in water significantly lowers the consumption of expensive organic solvents, directly contributing to substantial cost savings in raw material procurement. Moreover, the simplified workflow reduces the number of processing days, enhancing the agility of the supply chain to respond to market demand fluctuations without compromising on quality or regulatory adherence.
- Cost Reduction in Manufacturing: The elimination of sulfur protection and deprotection steps drastically reduces the consumption of reagents and solvents, leading to a leaner cost structure. By avoiding expensive heavy metal catalysts or toxic chlorinating agents, the process minimizes the need for costly purification steps such as chromatography or extensive washing, resulting in significant operational expenditure reductions. This efficiency allows for competitive pricing strategies while maintaining healthy margins, making it an attractive option for large-volume contracts in the nutraceutical and pharmaceutical sectors.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials like hercynine and cysteine derivatives ensures a stable supply base that is less susceptible to geopolitical or logistical disruptions. The aqueous nature of the reaction reduces dependency on volatile organic solvent markets, further stabilizing input costs. Additionally, the robustness of the reaction conditions means that production can be scaled across different manufacturing sites with minimal requalification, providing redundancy and security for long-term supply agreements.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with the 'one-pot' capability reducing equipment footprint and processing time. The reduced generation of hazardous waste aligns with increasingly strict environmental regulations, lowering the burden of waste treatment and permitting. This eco-friendly profile not only enhances corporate sustainability goals but also facilitates smoother regulatory approvals in key markets, accelerating time-to-market for new products containing this high-value antioxidant.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ergothioneine synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and benefits of adopting this route for industrial production. Understanding these details is crucial for stakeholders evaluating the potential for technology licensing or contract manufacturing partnerships, ensuring that all technical risks are adequately assessed and managed before project initiation.
Q: How does this synthetic method improve safety compared to conventional routes?
A: Conventional methods often rely on highly toxic reagents like thiophosgene or potassium thiocyanate in acidic media. This novel approach utilizes a thiol-mediated cleavage in aqueous conditions, significantly reducing environmental hazards and operator risk.
Q: What are the key advantages for large-scale manufacturing?
A: The process allows for a 'one-pot' strategy where the intermediate does not require isolation, simplifying the workflow. Additionally, the use of water as a primary solvent minimizes organic waste and facilitates easier downstream processing.
Q: Can this method produce the natural L-enantiomer?
A: Yes, by starting with L-Hercynine and L-Cysteine derivatives, the method effectively produces L-Ergothioneine, which is the naturally occurring and biologically active form required for pharmaceutical and nutraceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ergothioneine Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this advanced synthetic methodology into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial manufacturing is seamless and efficient. We are committed to meeting stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify the identity and quality of every batch. Whether you require custom synthesis or large-volume supply, our facility is equipped to handle the specific demands of ergothioneine production with the highest standards of safety and quality assurance.
We invite you to collaborate with us to leverage this cutting-edge technology for your product pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency and product competitiveness in the global market.
