Industrial Scale-Up of Alkyl 2-Alkoxymethylene-4,4-Difluoro-3-Oxobutyrate via Solvent-Free Claisen Condensation
The global demand for high-performance agrochemicals, particularly fungicides based on pyrazole carboxamide structures, has necessitated the development of more efficient synthetic routes for their key fluorinated intermediates. Patent CN101959840B discloses a groundbreaking process for preparing alkyl 2-alkoxymethylene-4,4-difluoro-3-oxobutyrate (VI), a critical building block in the synthesis of 1-methyl-3-difluoromethylpyrazole derivatives. This innovation addresses long-standing inefficiencies in the production of fluorinated beta-keto esters, shifting away from hazardous solvent-dependent methods toward a streamlined, solvent-free protocol. By optimizing the Claisen condensation and subsequent conversion steps, this technology offers a robust pathway for generating high-purity intermediates essential for modern crop protection agents. As a leading entity in fine chemical manufacturing, understanding the nuances of this patent is vital for securing a competitive edge in the agrochemical supply chain.
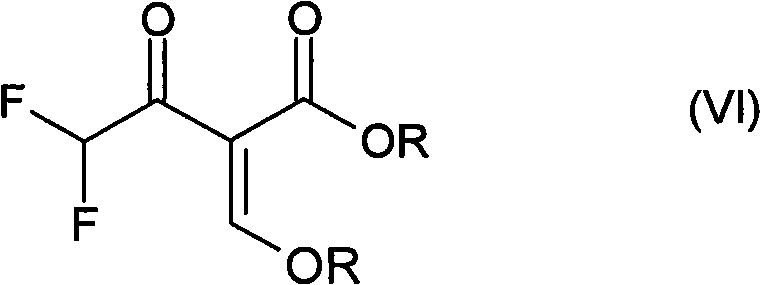
The structural integrity of compound (VI) is paramount for downstream cyclization reactions, and the patented method ensures minimal degradation of the sensitive difluoro moiety. Traditional approaches often suffered from thermal instability and side reactions induced by prolonged exposure to reactive solvents. In contrast, the methodology outlined in CN101959840B leverages precise stoichiometric control and temperature management to preserve the fluorine content while maximizing yield. For R&D directors and procurement specialists alike, this represents a significant opportunity to enhance the quality of raw materials entering the production line, thereby reducing the burden on purification units and improving the overall economic viability of the final active pharmaceutical or agrochemical ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical methods for synthesizing 4,4-difluoroacetoacetate esters, such as the procedure described by Y. Desirant in 1930, relied heavily on the use of dry ether as a solvent. This legacy approach is fraught with industrial disadvantages, primarily due to the extremely low boiling point of ether, which necessitates complex containment systems to prevent evaporation losses and poses significant safety risks regarding peroxide formation. Furthermore, the reaction kinetics in these solvent-heavy environments are sluggish, often requiring up to 5 days to reach completion, which severely bottlenecks production capacity. The reported yields for these conventional methods rarely exceed 65%, largely because the isolation of the intermediate product involves distillation steps where thermal sensitivity leads to substantial decomposition. For a procurement manager, these factors translate into higher raw material costs, increased energy consumption for solvent recovery, and unpredictable lead times that can disrupt the supply of critical agrochemical intermediates.
The Novel Approach
The novel approach detailed in the patent fundamentally reengineers the synthesis by eliminating the external solvent entirely, utilizing the reactants themselves to form a stirrable suspension or homogeneous mixture. This solvent-free strategy not only mitigates the safety hazards associated with volatile ethers but also dramatically accelerates the reaction rate, reducing processing time from days to mere hours. By conducting the Claisen condensation between alkyl acetate, alkoxide, and alkyl difluoroacetate in a concentrated medium, the process achieves yields exceeding 90% for the intermediate (I), a stark improvement over the historical 65% benchmark. The elimination of solvent removal steps prior to the final conversion further simplifies the workflow, allowing for a telescoped process where the crude intermediate is directly transformed into the target alkoxymethylene derivative. This efficiency gain is crucial for cost reduction in agrochemical manufacturing, as it maximizes reactor throughput and minimizes waste generation.
Mechanistic Insights into Solvent-Free Claisen Condensation
The core of this innovative synthesis lies in the formation of the enolate intermediate (V) through a carefully controlled Claisen condensation. In the absence of diluting solvents, the interaction between the alkoxide base (III) and the ester substrates (II and IV) is intensified, promoting rapid deprotonation and nucleophilic attack. The molar ratios are critical here; maintaining an excess of alkyl acetate relative to the alkoxide, typically in a range of 2:1 to 4:1, ensures that the base is fully consumed in generating the reactive enolate species without causing excessive side reactions. The reaction temperature is managed within a window of -20°C to 70°C, allowing for precise control over the exothermic nature of the condensation. This mechanistic precision ensures that the difluoroacetyl group remains intact, preventing defluorination which is a common pitfall in basic conditions.
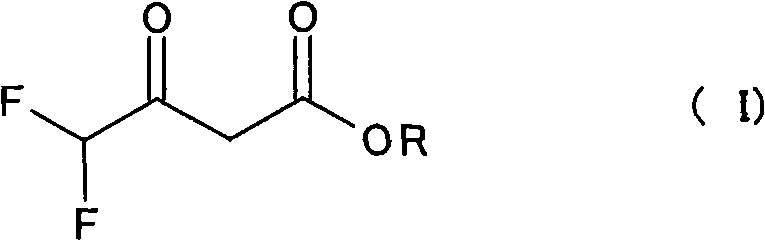
Following the formation of the enolate salt (V), the release of the free 4,4-difluoroacetoacetate (I) is achieved through acidification. The patent highlights the importance of using anhydrous acids or acids with minimal water content, such as gaseous hydrogen chloride or concentrated sulfuric acid. This specific condition is vital because excessive water can hydrolyze the ester functionalities or interfere with the subsequent orthoester reaction. The acidification step precipitates the inorganic salt byproduct (e.g., sodium chloride or sodium sulfate), which can be efficiently removed via filtration. This solid-liquid separation is a key purification point, effectively removing ionic impurities before the final step. The resulting filtrate contains the crude intermediate (I) dissolved in the excess alkyl acetate, ready for immediate conversion without the need for energy-intensive distillation.
How to Synthesize Alkyl 2-Alkoxymethylene-4,4-Difluoro-3-Oxobutyrate Efficiently
The synthesis protocol described in the patent offers a clear roadmap for industrial implementation, focusing on operational simplicity and high yield. The process begins with the metered addition of reactants to control exotherms, followed by a straightforward acidification and filtration sequence. The final conversion step utilizes triethyl orthoformate and acetic anhydride to install the alkoxymethylene group, completing the synthesis of the target molecule (VI). This telescoped approach minimizes unit operations and reduces the overall footprint of the manufacturing process. For technical teams looking to adopt this methodology, the following guide outlines the critical stages involved in executing this high-efficiency route.
- React alkyl acetate, alkoxide, and alkyl difluoroacetate without additional solvent to form an enolate intermediate.
- Release the 4,4-difluoroacetoacetate intermediate using an acid such as hydrogen chloride or sulfuric acid.
- Convert the crude intermediate directly into the final product using triethyl orthoformate and acetic anhydride.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this solvent-free technology offers profound benefits for supply chain stability and cost management. The elimination of ether solvents removes a major variable in raw material sourcing and hazard management, simplifying logistics and storage requirements. Additionally, the significant increase in yield directly correlates to a reduction in the cost of goods sold, as less starting material is required to produce the same amount of final product. The ability to filter off inorganic salts rather than performing aqueous workups also reduces the volume of wastewater generated, aligning with increasingly stringent environmental regulations. These factors combined create a more resilient supply chain capable of meeting the high-volume demands of the agrochemical sector.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic reduction in solvent usage and the elimination of intermediate distillation steps. By avoiding the use of expensive and hazardous ether solvents, manufacturers save on both raw material procurement and the capital expenditure required for explosion-proof solvent recovery systems. Furthermore, the higher reaction yields mean that the effective cost per kilogram of the intermediate is significantly lowered, as less feedstock is wasted in side reactions or lost during purification. The telescoped nature of the process also reduces labor and energy costs associated with multiple isolation and drying steps, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as ethyl acetate, sodium ethoxide, and ethyl difluoroacetate ensures a stable and diversified supply base. Unlike specialized solvents that may face market volatility, these starting materials are widely available from multiple global suppliers, reducing the risk of supply disruptions. The shortened reaction time from days to hours allows for faster turnaround times in production scheduling, enabling manufacturers to respond more agilely to fluctuations in market demand. This reliability is crucial for maintaining continuous production lines for downstream fungicide synthesis, ensuring that customers receive their orders on time without delay.
- Scalability and Environmental Compliance: The solvent-free design inherently supports scalability, as the reaction mixture is more concentrated, allowing for greater production capacity within existing reactor volumes. The simplified waste stream, characterized mainly by solid inorganic salts and recoverable organic byproducts, facilitates easier compliance with environmental discharge standards. The reduction in volatile organic compound (VOC) emissions due to the absence of ether solvents further enhances the environmental profile of the facility. This alignment with green chemistry principles not only mitigates regulatory risk but also improves the corporate sustainability image, which is increasingly important for partnerships with major agrochemical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and process descriptions found in CN101959840B, providing clarity on reaction conditions, impurity profiles, and scalability considerations. Understanding these details is essential for process engineers and quality assurance teams evaluating the feasibility of this technology for their specific manufacturing contexts.
Q: What are the advantages of the solvent-free method over traditional ether-based synthesis?
A: The solvent-free method eliminates the use of hazardous ether solvents, reduces reaction time from 5 days to hours, and significantly improves yield from 65% to over 90%.
Q: How is the purity of the intermediate controlled during the acidification step?
A: Purity is maintained by using anhydrous acids or acids with minimal water content, allowing for the efficient filtration of inorganic salt byproducts before the final conversion step.
Q: Is this process suitable for large-scale manufacturing of fungicide precursors?
A: Yes, the process is designed for industrial scalability, utilizing commercially available starting materials and avoiding complex distillation steps that cause product loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl 2-Alkoxymethylene-4,4-Difluoro-3-Oxobutyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality fluorinated intermediates in the development of next-generation agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alkyl 2-alkoxymethylene-4,4-difluoro-3-oxobutyrate meets the exacting standards required for fungicide synthesis. Our commitment to process excellence allows us to deliver consistent quality while maintaining the flexibility to adapt to custom client requirements.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your production volume. We encourage you to contact us to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial security.
