Advanced Manufacturing of Difluoro-Oxobutyrate Intermediates for Next-Generation Fungicides
Introduction to Patent CN102791694A and Strategic Value
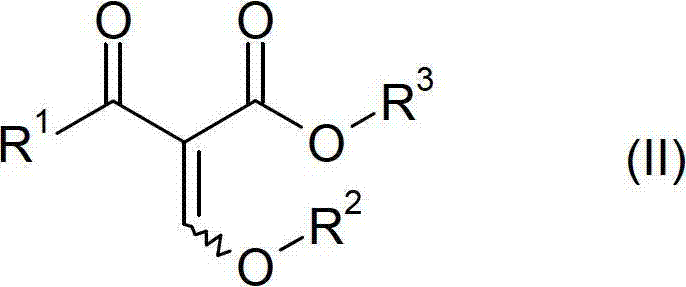
The global demand for high-performance succinate dehydrogenase inhibitor (SDHI) fungicides continues to drive innovation in the synthesis of their core heterocyclic intermediates. Patent CN102791694A discloses a highly efficient preparation method for compounds of Formula II, specifically alkyl 2-alkoxymethylene-4,4-difluoro-3-oxobutyrate derivatives. These molecules serve as indispensable precursors in the manufacture of 3-difluoromethyl-1-methyl-1H-pyrazole-4-carboxylic acid, a structural motif found in blockbuster agrochemicals like Isopyrazam and Sedaxane. The technical breakthrough described in this intellectual property lies in the optimization of the reaction sequence, specifically targeting the elimination of redundant purification steps that have historically plagued this synthetic route. By re-engineering the workflow to tolerate inorganic salts within the reaction matrix, the patent offers a pathway to substantially lower manufacturing costs and improved throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of these fluorinated beta-keto ester intermediates relied on processes described in earlier literature, such as WO2009/106619. In these conventional workflows, the synthesis typically involves the formation of an enolate species which must then be protonated to liberate the free beta-keto ester. A critical bottleneck in these legacy methods is the mandatory requirement to remove the inorganic salt by-product generated during the acidification step before proceeding to the next transformation. This salt removal often necessitates the addition of water to facilitate filtration, as the salts can exist in forms that are difficult to filter on a large scale without aqueous modification. However, the introduction of water creates a cascade of negative effects, including the increased consumption of moisture-sensitive reagents like orthoesters and acetic anhydride in subsequent steps. Furthermore, the physical handling of salt cakes and the associated filtration equipment adds significant capital expenditure and operational time to the manufacturing cycle, creating a barrier to efficient commercial scale-up.
The Novel Approach
The methodology outlined in CN102791694A represents a paradigm shift by challenging the necessity of intermediate salt removal. The inventors surprisingly discovered that the enolate protonation and subsequent salt filtration steps are entirely superfluous for the successful conversion to the target alkoxymethylene compound. Instead, the process allows the reaction to proceed in a telescoped manner where the inorganic salt remains suspended or dissolved in the reaction mixture during the critical condensation step with the orthoester. This approach not only drastically simplifies the unit operations by removing a filtration stage but also enables the reaction to be conducted in the substantial absence of water. By avoiding water addition, the process minimizes the hydrolysis of valuable reagents, thereby improving atom economy and reducing the overall raw material intensity. This streamlined logic transforms a multi-step batch process with isolation into a more continuous and fluid operation, directly addressing the pain points of cycle time and waste generation inherent in the older technology.
Mechanistic Insights into Enolate Formation and Orthoester Condensation
The core chemical transformation begins with the generation of a metal enolate from readily available starting materials. As depicted in the reaction scheme, compounds of Formula IV (such as ethyl acetate), Formula V (an alkali metal alkoxide like sodium ethoxide), and Formula VI (ethyl difluoroacetate) are reacted to form the enolate of Formula VII. This step is exothermic and requires precise temperature control, typically maintained between 0°C and 80°C, to ensure high conversion while minimizing side reactions. The choice of base (lithium, sodium, or potassium) and the specific alkyl groups on the esters can be tuned to optimize solubility and reactivity. The resulting enolate is a nucleophilic species that holds the potential energy required for the subsequent carbon-carbon bond formation. In traditional chemistry, this intermediate would be quenched immediately, but here it is preserved or transiently formed to feed directly into the acidification stage without workup.
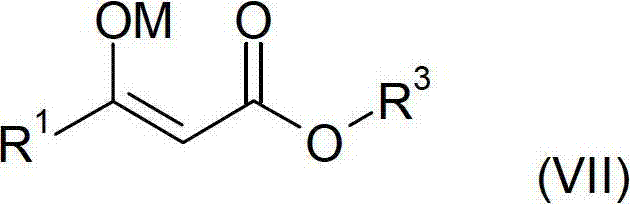
Following enolate formation, the mixture is treated with a mineral acid, preferably hydrogen chloride, to liberate the beta-keto ester of Formula VIII. Crucially, this acidification is performed under anhydrous conditions to prevent the degradation of the sensitive fluorinated backbone. The resulting mixture, containing the free beta-keto ester and the precipitated inorganic salt (e.g., NaCl), is then subjected to condensation with an orthoester (Formula IX). The presence of the salt does not inhibit the orthoester reaction; rather, the process leverages reduced pressure (0 to 750 mbar) to continuously distill off low-boiling by-products like ethanol. This dynamic removal of volatiles drives the equilibrium towards the formation of the desired alkoxymethylene product (Formula II). The ability to perform this condensation in the presence of suspended solids demonstrates a robust tolerance for impurities that allows for significant process intensification. Finally, the crude mixture containing Formula II can be directly utilized in the subsequent cyclization with methylhydrazine to form the pyrazole ring of Formula III, further exemplifying the telescoped nature of this superior synthetic design.
How to Synthesize Alkyl 2-alkoxymethylene-4,4-difluoro-3-oxobutyrate Efficiently
Implementing this patented methodology requires careful attention to reagent stoichiometry and thermal management to maximize yield and safety. The process is designed to be flexible regarding solvents, allowing for the use of water-immiscible organic solvents like xylene or toluene which facilitate downstream phase separations. Operators must ensure that the acidification step is strictly controlled to avoid local overheating, and the subsequent distillation under vacuum must be managed to prevent bumping due to the presence of solid salts. The following guide outlines the standardized operational parameters derived from the patent examples, providing a roadmap for R&D teams to replicate the high efficiency reported in the intellectual property documentation.
- React ethyl acetate, alkali metal alkoxide, and ethyl difluoroacetate to form an enolate intermediate at controlled temperatures between 0°C and 80°C.
- Liberate the beta-keto ester compound from the enolate using mineral acid (e.g., HCl) in the substantial absence of water, retaining the formed salt in the mixture.
- Convert the beta-keto ester to the target alkoxymethylene compound using an orthoester under reduced pressure to remove low-boiling by-products.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN102791694A offers tangible strategic benefits beyond mere chemical elegance. The primary value driver is the significant reduction in processing time and equipment utilization. By eliminating the salt filtration and washing steps, manufacturers can reduce the batch cycle time, effectively increasing the capacity of existing reactor trains without capital expansion. This improvement in throughput translates directly to better supply security and the ability to respond more agilely to market fluctuations in fungicide demand. Furthermore, the reduction in unit operations lowers the labor burden and decreases the probability of human error or containment breaches during solid handling, enhancing overall plant safety profiles.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of water usage in the intermediate stages. In conventional routes, water is added to assist salt filtration, which subsequently necessitates the use of excess dehydrating agents or scavengers in later steps to protect moisture-sensitive reagents. By operating anhydrously, this new method drastically cuts the consumption of expensive orthoesters and anhydrides. Additionally, the avoidance of acid anhydrides removes the safety risks associated with thermally unstable distillation fractions, reducing waste disposal costs and insurance liabilities related to hazardous material handling.
- Enhanced Supply Chain Reliability: The robustness of the reaction against the presence of inorganic salts means that the process is less sensitive to variations in filtration efficiency or drying times. This reliability ensures consistent batch-to-batch quality and predictable lead times for the production of high-purity agrochemical intermediates. Suppliers utilizing this technology can offer more stable delivery schedules because the production line is less prone to bottlenecks caused by clogged filters or extended drying cycles. This stability is crucial for long-term contracts with major agrochemical companies who require just-in-time delivery of critical intermediates for their formulation plants.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates less aqueous waste since water is not introduced solely for filtration purposes. This reduction in wastewater volume simplifies effluent treatment and lowers the environmental footprint of the manufacturing site. The ability to scale up without the need for large-scale filtration equipment also reduces the physical footprint of the plant. Moreover, the simplified workflow minimizes the number of transfer steps, reducing the risk of spillage and exposure, which aligns with increasingly stringent global regulations on occupational health and safety in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within the patent specification, providing clarity on the operational boundaries and advantages of the technology. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or licensing agreements.
Q: How does this patent improve upon previous methods like WO2009/106619?
A: The novel method eliminates the unnecessary step of protonating the enolate followed by salt removal and filtration. By telescoping the reaction and keeping the salt in the mixture during the subsequent orthoester conversion, the process significantly reduces cycle times and avoids the need for water addition that typically increases raw material consumption.
Q: What are the safety advantages of avoiding acid anhydrides in this synthesis?
A: Conventional methods often utilize acid anhydrides which can form thermally unstable distillation fractions containing anhydrides, acids, and orthoesters. This new approach preferably operates without anhydrides, simplifying the process safety profile and removing limitations on recycling reaction mixtures.
Q: Can this intermediate be used for major SDHI fungicides?
A: Yes, the compounds of Formula II produced via this method are critical precursors for 3-difluoromethyl-1-methyl-1H-pyrazole-4-carboxylic acid. This acid is a key building block for prominent succinate dehydrogenase inhibitor (SDHI) fungicides such as Isopyrazam, Sedaxane, and Fluxapyroxad.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl 2-alkoxymethylene-4,4-difluoro-3-oxobutyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires deep process engineering expertise. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of CN102791694A are fully realized in our manufacturing facilities. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of difluoro-oxobutyrate intermediate meets the exacting standards required for SDHI fungicide synthesis. Our commitment to quality assurance means that clients receive materials with consistent impurity profiles, facilitating smoother downstream processing in their own plants.
We invite global partners to collaborate with us to leverage this advanced manufacturing technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating these high-efficiency intermediates into your agrochemical portfolios. Let us be your trusted partner in delivering cost-effective and reliable chemical solutions.
