Advanced Resolution Process for High-Purity Beraprost Acid Manufacturing
Advanced Resolution Process for High-Purity Beraprost Acid Manufacturing
The pharmaceutical industry continuously seeks robust and scalable methodologies for producing optically active prostaglandin analogs, particularly for the treatment of peripheral arterial disease. Patent CN109219601B discloses a groundbreaking process for the preparation of optically active beraprost of formula (I) and its salts, addressing the critical challenge of separating the four possible stereoisomers inherent to this complex molecular architecture. Unlike traditional approaches that rely on early-stage chiral pool synthesis or expensive catalytic asymmetric reactions, this invention leverages a strategic late-stage resolution protocol starting from readily available racemic beraprost alkyl esters. The core innovation lies in a multi-step purification sequence involving hydrolysis, selective crystallization, and diastereomeric esterification, which collectively ensure the isolation of the biologically active alpha 2/1 isomer with exceptional purity. This technical advancement represents a significant leap forward for manufacturers aiming to secure a reliable pharmaceutical intermediates supplier status while maintaining rigorous quality standards.
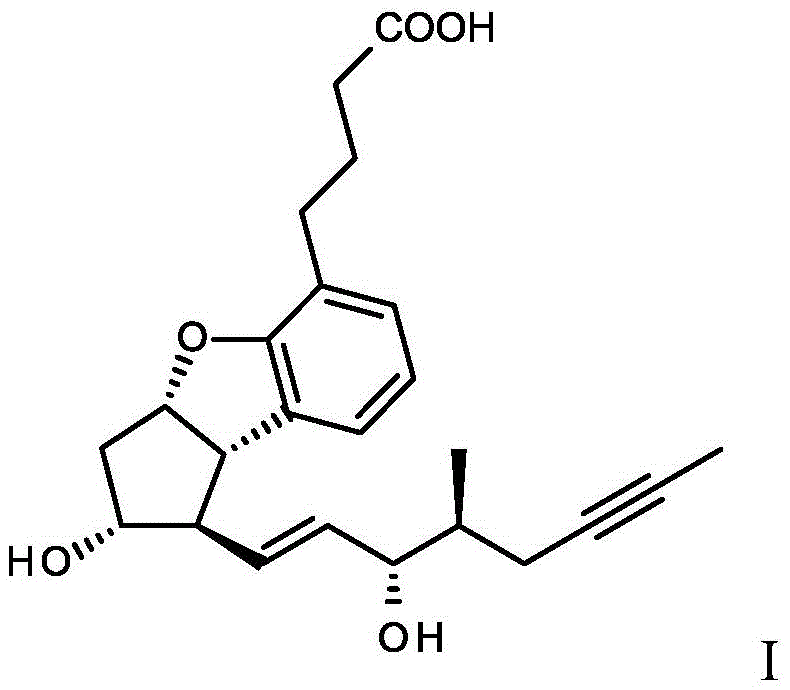
Beraprost sodium is commercially recognized for its efficacy, yet its chemical structure presents a formidable synthetic hurdle due to the presence of multiple chiral centers, resulting in a mixture of four isomers in non-stereoselective syntheses. The target molecule, specifically the (1 alpha 2/1) isomer, possesses the requisite therapeutic potency, necessitating a manufacturing route that can effectively discriminate between enantiomers and diastereomers. The disclosed method initiates with the hydrolysis of a racemic beraprost ester, followed by a crucial crystallization step that enriches the mixture with the desired diastereomeric pair (alpha 2/1 and alpha 2/2). Subsequent derivatization with a chiral acid, such as R-acetyl-chloromandelic acid, transforms these enantiomers into diastereomers with divergent physical properties, enabling their separation via standard chromatographic techniques. This logical progression from racemic starting materials to optically pure final products underscores the process's viability for industrial application.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beraprost has been dominated by routes that introduce chirality at the very beginning of the synthetic sequence, often relying on complex natural products or specialized chiral catalysts. For instance, the original Toray synthesis utilizes cyclopentadiene and involves a Grignard reaction followed by a copper-catalyzed cyclization to establish the tricyclic core, as illustrated in earlier literature. While effective for small-scale research, these pathways often suffer from low overall yields due to the accumulation of impurities across numerous steps. Furthermore, alternative methods disclosed in patents like WO2012/174407A1 employ lanthanide catalysts, specifically Eu(hfc)3, to facilitate Diels-Alder cycloadditions. The reliance on such rare-earth metals introduces significant supply chain vulnerabilities and necessitates stringent heavy metal clearance protocols, which drastically inflate the cost of goods sold (COGS) and complicate regulatory filings for API manufacturing.
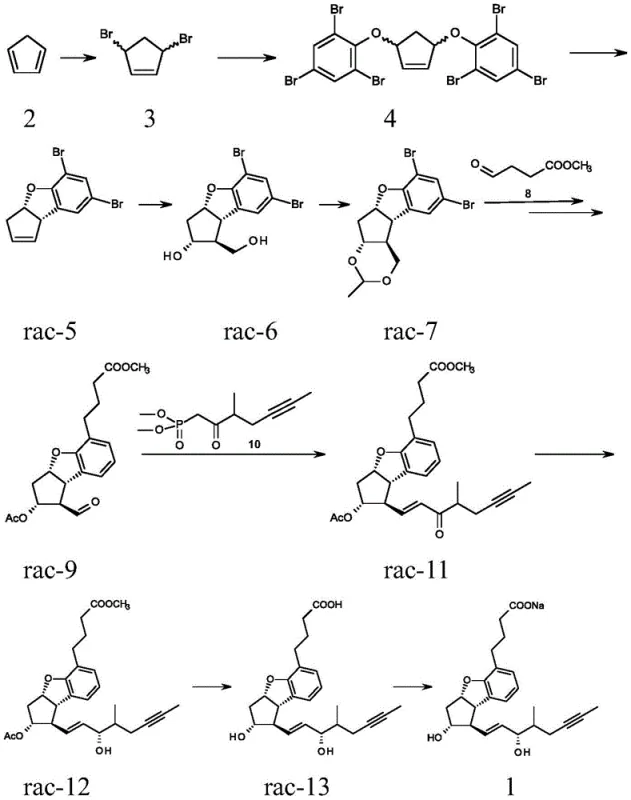
The Novel Approach
In stark contrast, the process detailed in CN109219601B adopts a convergent strategy that postpones the resolution of stereocenters until the molecule is nearly fully assembled. By starting with a racemic beraprost ester, which can be synthesized through established and cost-effective non-stereoselective routes, the method bypasses the need for expensive chiral starting materials. The novelty is anchored in the differential solubility and chromatographic behavior of the intermediate diastereomers. After an initial crystallization removes the less desirable alpha 1/1 and alpha 1/2 isomers, the remaining racemic mixture of the active diastereomeric pair is subjected to chiral derivatization. This creates diacyl-beraprost ester diastereoisomers that can be cleanly separated using atmospheric silica gel chromatography, a unit operation that is far more amenable to scale-up than preparative chiral HPLC. This shift in paradigm significantly simplifies the manufacturing workflow.
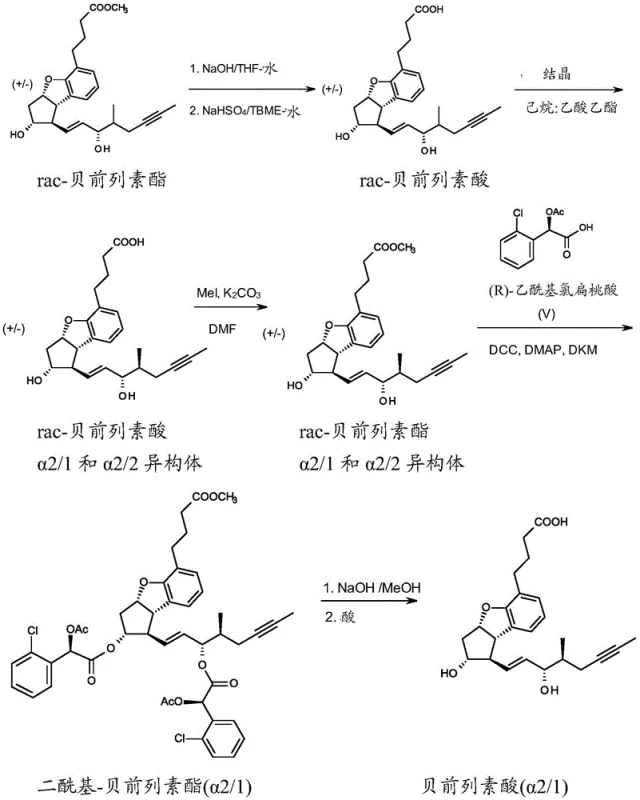
Mechanistic Insights into Diastereomeric Esterification and Resolution
The heart of this technological breakthrough is the formation of diastereomeric esters using a chiral auxiliary, a classic yet highly effective technique in stereochemistry. The process involves reacting the free hydroxyl group of the partially purified racemic beraprost ester with a chiral acid derivative, specifically R-acetyl-chloromandelic acid. This reaction generates two distinct diastereomers, designated as VIa and VIb in the patent documentation, which differ in their three-dimensional arrangement despite having the same connectivity. Unlike enantiomers, which possess identical physical properties in an achiral environment, these diastereomers exhibit different polarities and solubilities. This divergence allows for their separation using standard normal-phase silica gel chromatography with a dichloromethane and ethyl acetate gradient. The ability to separate these intermediates with high fidelity is critical, as it ensures that the subsequent hydrolysis step yields the single, optically active beraprost acid isomer required for therapeutic use.
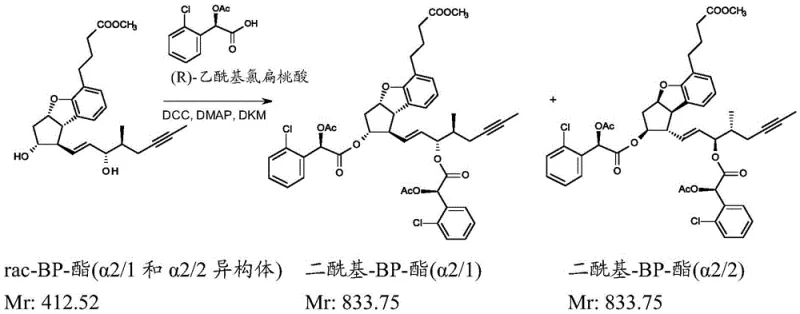
Furthermore, the process incorporates a robust impurity control mechanism through repeated crystallization steps prior to the chiral derivatization. The initial hydrolysis of the racemic ester yields a mixture of four acid isomers. By exploiting the subtle differences in crystal lattice energy and solubility in a hexane and ethyl acetate solvent system, the process selectively precipitates the alpha 2/1 and alpha 2/2 isomer pair while leaving the alpha 1/1 and alpha 1/2 isomers in the mother liquor. This pre-purification step is vital for maximizing the efficiency of the downstream chiral resolution, as it reduces the load of unwanted stereoisomers that would otherwise compete for the chiral resolving agent. The combination of crystallization and chromatographic separation creates a synergistic effect, driving the optical purity of the final product to levels that satisfy stringent pharmacopeial standards without the need for recycling large volumes of unwanted material.
How to Synthesize Optically Active Beraprost Efficiently
The synthesis of this complex prostaglandin analog requires precise control over reaction conditions to maintain stereochemical integrity throughout the sequence. The patented method outlines a clear pathway that begins with the saponification of the racemic ester under mild basic conditions to prevent epimerization of the sensitive alpha-centers. Following the isolation of the enriched acid fraction, the re-esterification and subsequent acylation with the chiral acid must be performed under anhydrous conditions to ensure high conversion rates. The final hydrolysis step removes both the methyl ester and the chiral auxiliary simultaneously, regenerating the free acid and alcohol functionalities of the target molecule. For a comprehensive understanding of the specific reagents, stoichiometry, and workup procedures required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Hydrolyze racemic beraprost ester using an aqueous inorganic base in a water-miscible organic solvent to obtain racemic beraprost acid.
- Purify the racemic acid by repeated crystallization from a hexane and ethyl acetate mixture to isolate the alpha 2/1 and alpha 2/2 isomer pair.
- Esterify the purified acid and react with a chiral acid derivative like R-acetyl-chloromandelic acid to form separable diastereomers, followed by chromatographic separation and final hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resolution-based manufacturing route offers compelling economic and logistical benefits over legacy synthetic pathways. By decoupling the construction of the carbon skeleton from the introduction of chirality, the process allows for the use of commodity-grade starting materials that are available in bulk quantities from multiple global suppliers. This diversification of the raw material base significantly mitigates the risk of supply disruptions that often plague specialty chemical markets. Moreover, the elimination of rare-earth catalysts and the reliance on standard purification techniques like crystallization and flash chromatography translate directly into a more predictable and stable cost structure. The process is designed to be inherently scalable, moving seamlessly from laboratory benchtop quantities to multi-ton commercial production without the need for specialized equipment or exotic reagents.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the avoidance of expensive chiral catalysts and the minimization of chromatographic steps. Traditional routes often require preparative chiral HPLC, which is prohibitively expensive at large scales due to the high cost of chiral stationary phases and solvent consumption. By converting enantiomers into diastereomers using a relatively inexpensive chiral acid like R-acetyl-chloromandelic acid, the separation can be achieved using standard silica gel, which is orders of magnitude cheaper. Additionally, the ability to purify intermediates via crystallization reduces solvent usage and energy consumption associated with evaporation, leading to substantial operational expenditure savings.
- Enhanced Supply Chain Reliability: The reliance on racemic starting materials provides a significant buffer against supply chain volatility. Chiral building blocks often have limited suppliers and long lead times, whereas racemic esters can be produced by any competent fine chemical manufacturer with standard capabilities. This flexibility allows procurement teams to negotiate better pricing and secure secondary sources more easily. Furthermore, the robustness of the crystallization steps ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by out-of-specification intermediates. This reliability is crucial for maintaining continuous API production schedules and meeting Just-In-Time delivery commitments to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process aligns well with green chemistry principles by reducing the use of hazardous heavy metals. The absence of lanthanide catalysts eliminates the need for complex metal scavenging steps and the associated waste disposal costs. The solvents employed, such as ethyl acetate, hexane, and dichloromethane, are widely used in the industry with well-established recovery and recycling protocols. The high selectivity of the resolution steps means that less material is wasted as off-spec product, improving the overall atom economy of the synthesis. This makes the process not only economically attractive but also environmentally sustainable, facilitating easier regulatory approval in jurisdictions with strict environmental controls.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of optically active beraprost using this novel resolution methodology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a transparent view of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this supply route into their existing manufacturing networks.
Q: Why is diastereomeric resolution preferred over chiral chromatography for beraprost?
A: Diastereomeric resolution via derivatization allows for separation using standard silica gel chromatography or crystallization, which are significantly more scalable and cost-effective for commercial production than preparative chiral HPLC.
Q: What chiral resolving agent is utilized in this novel process?
A: The process employs R-acetyl-chloromandelic acid as the chiral auxiliary to form diacyl-beraprost ester diastereoisomers, which exhibit distinct physical properties allowing for efficient separation.
Q: Does this method avoid the use of expensive transition metal catalysts?
A: Yes, unlike prior art methods that utilize lanthanide catalysts for Diels-Alder reactions, this resolution approach relies on classical organic transformations, eliminating the need for costly metal removal steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beraprost Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-purity intermediates that meet the exacting standards of the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We are equipped with state-of-the-art rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of beraprost intermediate we supply is free from deleterious impurities. Our commitment to technical excellence allows us to navigate the complexities of chiral resolution and crystallization with precision, delivering a product that supports your drug development and commercialization goals.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out to request specific COA data and route feasibility assessments to verify how our advanced resolution process can enhance your production efficiency. By partnering with us, you gain access to a reliable source of complex pharmaceutical intermediates backed by deep chemical expertise and a proven track record of successful commercialization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
