Advanced Synthesis of Beraprost Sodium: Technical Upgrades for Commercial Manufacturing
Advanced Synthesis of Beraprost Sodium: Technical Upgrades for Commercial Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for complex prostacyclin derivatives, particularly Beraprost Sodium, a critical therapeutic agent for pulmonary hypertension and chronic arterial occlusive disease. The patent CN112778254A discloses a groundbreaking synthetic method that addresses longstanding challenges in the production of cyclopenta[b]benzofuran-5-sodium butyrate. This innovation focuses on the selective protection of secondary hydroxyl groups under weak acidic conditions, a strategic modification that drastically simplifies the overall synthetic route. By optimizing reaction conditions and reagent selection, this methodology not only improves overall yield but also enhances the feasibility of industrial production. For R&D directors and procurement specialists, understanding this technical evolution is vital for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials consistently.
![Chemical structure of Beraprost Sodium showing the cyclopenta[b]benzofuran core and side chains](/insights/img/beraprost-sodium-synthesis-pharma-supplier-20260309110821-01.png)
Beraprost Sodium acts as a stable analogue of prostacyclin (PGI2), exhibiting potent platelet aggregation inhibition and vasodilation effects. The molecular complexity, featuring multiple chiral centers, traditionally poses significant hurdles in synthesis. However, the disclosed method leverages silicon protecting groups to navigate these complexities with precision. This approach ensures that the sensitive functional groups remain intact during subsequent transformations, thereby preserving the stereochemical integrity required for biological activity. As we delve deeper into the technical specifics, it becomes evident that this process represents a substantial leap forward in cost reduction in API manufacturing, offering a viable alternative to legacy methods that often suffer from low efficiency and hazardous byproduct generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of prostacyclin analogues has been plagued by the reliance on Swern oxidation for converting secondary alcohols to ketones. While chemically effective on a small scale, Swern oxidation presents severe drawbacks for commercial scale-up of complex pharmaceutical intermediates. The reaction requires cryogenic temperatures and generates dimethyl sulfide as a byproduct, which is notorious for its extremely foul odor and difficulty in containment. Furthermore, the workup procedures associated with Swern oxidation are cumbersome, often requiring extensive purification steps to remove sulfur-containing impurities that can poison downstream catalysts. These factors collectively contribute to increased operational costs, longer processing times, and significant environmental compliance burdens, making traditional routes less attractive for modern green chemistry initiatives.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a selective silyl protection strategy followed by oxidation with reagents such as Dess-Martin periodinane or pyridinium chlorochromate (PCC). This shift eliminates the need for malodorous sulfur reagents and allows reactions to proceed under much milder conditions, typically ranging from 0°C to 50°C. The use of alkyl silicon chlorides, such as tert-butyldimethylsilyl chloride, provides a robust protecting group that withstands subsequent reaction conditions yet can be easily removed when necessary. This methodological refinement results in a cleaner reaction profile, higher crude purity, and a streamlined workflow that is inherently safer for plant operators. By adopting this route, manufacturers can achieve significant cost savings through reduced waste treatment needs and simplified equipment requirements.
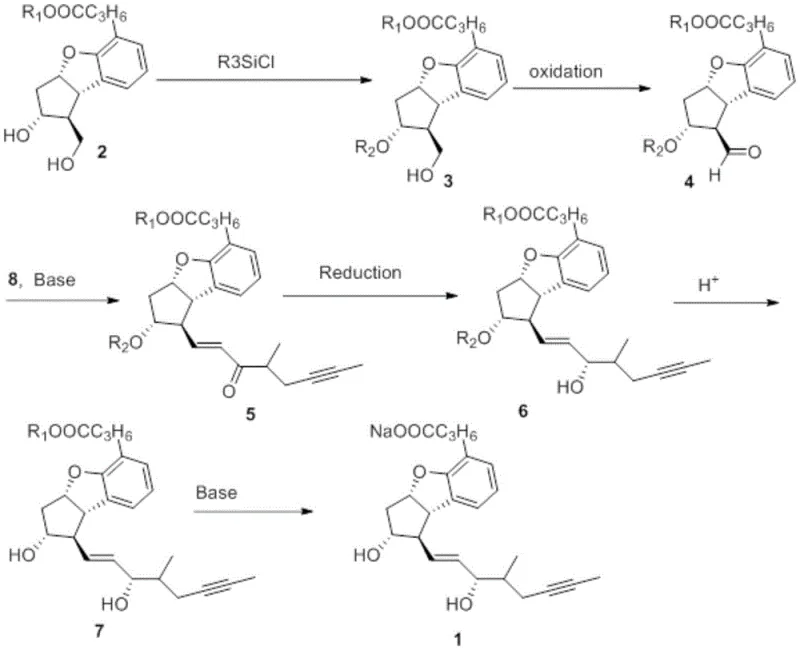
Mechanistic Insights into Selective Hydroxyl Protection and Asymmetric Reduction
The core of this synthetic breakthrough lies in the precise manipulation of stereochemistry and functional group reactivity. The initial step involves the reaction of the starting diol with an alkyl silicon chloride in the presence of a base like imidazole or triethylamine. This step is critical because it selectively protects the secondary hydroxyl group while leaving the primary hydroxyl or other sensitive moieties available for further transformation, depending on the specific derivative being targeted. The choice of silyl group—whether TBS, TES, or TBDPS—is tuned to balance stability and lability, ensuring that the protecting group remains intact during the oxidation and olefination steps but can be cleaved efficiently under mild acidic conditions later in the sequence. This selectivity is paramount for maintaining the structural fidelity of the cyclopenta[b]benzofuran core.
Furthermore, the reduction of the enone intermediate (Compound 5) to the allylic alcohol (Compound 6) is executed using chiral reducing agents such as (-)-diisopinocampheylchloroborane ((-)-DIP-Cl). This asymmetric reduction is a pivotal moment in the synthesis, as it establishes the correct stereochemistry at the newly formed chiral center. Conventional reducing agents like sodium borohydride often produce mixtures of isomers that are challenging and costly to separate. By employing a chiral borane reagent at controlled temperatures (e.g., -30°C), the process minimizes the formation of unwanted diastereomers. This high level of stereocontrol directly translates to improved purity profiles in the final active pharmaceutical ingredient, reducing the burden on downstream purification processes and ensuring consistent quality for clinical applications.
How to Synthesize Beraprost Sodium Efficiently
Implementing this synthetic route requires careful attention to reaction parameters and reagent quality to maximize efficiency and yield. The process is designed to be modular, allowing for optimization at each stage from protection to final salt formation. Operators must ensure strict control over moisture levels during the silylation and organometallic steps, as water can quench reactive intermediates and lower yields. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for technical teams aiming to replicate this high-performance synthesis. Detailed procedural specifics regarding stoichiometry and workup are essential for achieving the reported success rates in a production environment.
- React starting compound 2 with alkyl silicon chloride under alkaline conditions to selectively protect the secondary hydroxyl group, yielding compound 3.
- Oxidize compound 3 using Dess-Martin periodinane or PCC in an organic solvent to obtain the aldehyde intermediate compound 4.
- Perform Horner-Wadsworth-Emmons reaction between compound 4 and phosphonate compound 8 under basic conditions to form the enone compound 5.
- Reduce compound 5 using a chiral reducing agent like (-)-DIP-Cl to stereoselectively generate the alcohol compound 6.
- Remove the silyl protecting group from compound 6 using acid to reveal the free hydroxyl group, resulting in compound 7.
- Treat compound 7 with aqueous alkali to form the final sodium salt, Beraprost Sodium (Compound 1).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic method offers tangible benefits beyond mere technical elegance. The elimination of hazardous reagents and the simplification of unit operations directly correlate to enhanced supply chain reliability. By reducing the complexity of the manufacturing process, the risk of batch failures due to operational errors is minimized, ensuring a more consistent flow of materials. Moreover, the use of commercially available and relatively inexpensive oxidants like PCC, as opposed to specialized cryogenic reagents, stabilizes the raw material supply chain. This stability is crucial for maintaining production schedules and meeting the demanding delivery timelines of global pharmaceutical clients without compromising on quality standards.
- Cost Reduction in Manufacturing: The replacement of Swern oxidation with solid-supported or solution-phase oxidants like Dess-Martin or PCC removes the necessity for specialized scrubbing systems to handle volatile sulfur compounds. This qualitative shift in process chemistry leads to substantial cost savings by lowering capital expenditure on environmental control equipment and reducing ongoing maintenance costs. Additionally, the higher yields observed in the protection and reduction steps mean that less raw material is wasted per kilogram of final product, effectively lowering the cost of goods sold (COGS) and improving overall margin potential for the manufacturer.
- Enhanced Supply Chain Reliability: The reagents specified in this protocol, such as imidazole, various silyl chlorides, and common organic solvents like dichloromethane and THF, are commodity chemicals with robust global supply networks. This availability reduces the risk of production stoppages caused by raw material shortages. Furthermore, the milder reaction conditions reduce the stress on reactor vessels and piping, extending the lifespan of manufacturing assets and decreasing downtime for repairs. This operational resilience ensures that the reliable pharmaceutical intermediates supplier can maintain continuous production even during periods of high market demand.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this method mitigates those concerns through inherent design. The absence of toxic gas evolution and the use of manageable temperatures facilitate a smoother transition from pilot scale to commercial tonnage. The waste streams generated are easier to treat and dispose of in compliance with international environmental regulations. This alignment with green chemistry principles not only safeguards the environment but also protects the company's reputation and license to operate, making it a sustainable choice for long-term commercial scale-up of complex prostacyclin derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Beraprost Sodium using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical rationale behind the process. Understanding these details helps stakeholders make informed decisions about sourcing and partnership opportunities. The answers reflect a commitment to transparency and technical excellence, ensuring that all partners have a clear view of the capabilities and limitations of the synthesis route.
Q: Why is this synthetic method preferred over traditional Swern oxidation?
A: Traditional Swern oxidation requires harsh conditions and produces dimethyl sulfide, which has a foul odor and complicates industrial waste treatment. This novel method utilizes mild oxidants like Dess-Martin or PCC, significantly improving operational safety and environmental compliance.
Q: How does this process ensure high stereochemical purity?
A: The process employs chiral reducing agents such as (-)-diisopinocampheylchloroborane in the reduction step. This specifically targets the ketone intermediate to minimize isomer impurities, which are notoriously difficult to separate in prostacyclin synthesis.
Q: Is this route suitable for large-scale commercial production?
A: Yes, the method uses readily available raw materials and avoids cryogenic conditions where possible. The selective protection strategy simplifies the workflow, making it robust for scaling from pilot batches to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beraprost Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to verify that every batch meets the highest industry standards. Our facility is equipped to handle the specific requirements of sensitive prostacyclin synthesis, including moisture-controlled environments and specialized containment for potent compounds, guaranteeing the integrity of the final product.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis of Beraprost Sodium can support your commercial goals. Let us be your partner in bringing high-purity pharmaceutical intermediates to the market efficiently and reliably.
