Scalable Nine-Step Synthesis of Pyrroloquinoline Quinone for Commercial Manufacturing
Introduction to Advanced PQQ Manufacturing Technologies
The landscape of oxidoreductase cofactor manufacturing has evolved significantly with the disclosure of patent CN101193888A, which outlines a robust nine-step synthetic methodology for Pyrroloquinoline Quinone (PQQ). This document represents a critical advancement for industrial chemists seeking reliable pharmaceutical intermediate supplier solutions, as it addresses the historical bottlenecks associated with scaling PQQ production beyond milligram quantities. The core innovation lies not merely in the construction of the pyrroloquinoline skeleton, but specifically in the refinement of the terminal hydrolysis and isolation steps, which traditionally plagued earlier methodologies with low yields and complex purification requirements. By leveraging a controlled precipitation of a triacid potassium salt, this process ensures consistent quality and facilitates the transition from laboratory discovery to commercial viability.
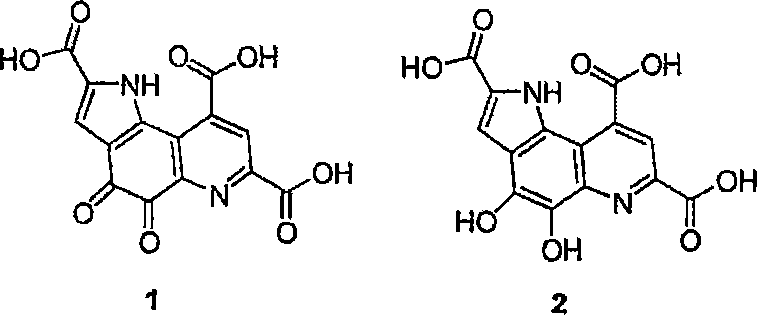
Understanding the redox versatility of PQQ is fundamental to appreciating the value of this synthesis. As depicted in the structural analysis, PQQ functions as a versatile prosthetic group capable of reversible reduction to semiquinone or hydroquinone forms, underpinning its utility in diverse biological applications ranging from antioxidant protection to neuroprotection. The ability to synthesize this molecule with high fidelity and minimal metallic impurities is paramount for its application in high-value nutraceutical and pharmaceutical formulations. This patent provides the technical blueprint for achieving such standards, offering a pathway that balances chemical efficiency with the rigorous purity specifications demanded by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the advancements detailed in this patent, the synthesis of PQQ was largely constrained by the methodologies established by Corey and Martin, which, while scientifically groundbreaking, presented significant hurdles for cost reduction in pharmaceutical intermediate manufacturing. The Corey method, for instance, was limited to small-scale production, typically yielding only around 50 milligrams of product, which is insufficient for any meaningful commercial application. Furthermore, the Martin method, although suitable for larger scales, suffered from a cumbersome final isolation procedure that required a two-stage separation involving both sodium and cesium salts. This dual-salt approach not only extended the processing time but also introduced complexities in removing residual heavy metals and inorganic salts, thereby complicating the downstream purification workflow and increasing the overall cost of goods sold.
The Novel Approach
In stark contrast, the novel approach described in CN101193888A streamlines the terminal phase of synthesis by introducing a single-step isolation of a triacid salt containing a单一 metal element, preferably potassium. This strategic shift eliminates the need for the laborious two-stage separation inherent in the Martin protocol, thereby reducing the operational footprint and enhancing the throughput of the manufacturing line. The process utilizes a specific sequence of hydrolysis followed by precise pH adjustment to precipitate the desired intermediate directly from the reaction matrix. This simplification is a game-changer for commercial scale-up of complex oxidoreductase cofactors, as it minimizes unit operations and reduces the potential for yield loss during transfer and filtration steps. The result is a more agile and economically viable production process capable of meeting multi-gram and kilogram demands.
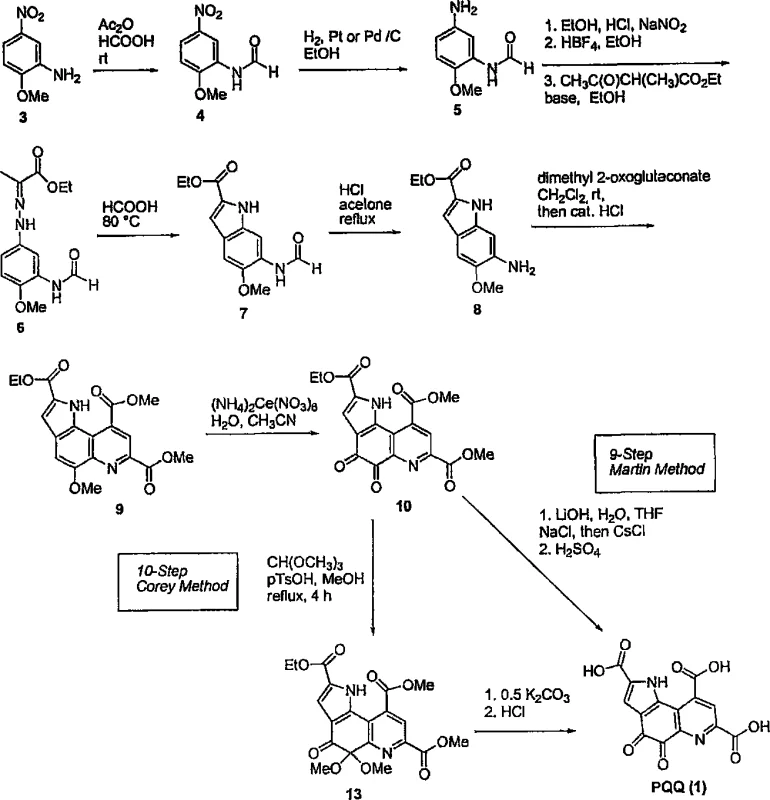
Mechanistic Insights into Controlled Hydrolysis and Salt Precipitation
The heart of this technological breakthrough resides in the mechanistic control exerted during the final hydrolysis step, where the tri-ester precursor is converted into the free acid form. The patent specifies that the reaction mixture, comprising the ester substrate and lithium hydroxide in a tetrahydrofuran-water system, must be maintained at a stringent temperature range of 16°C to 17°C. This narrow thermal window is critical; empirical data within the patent indicates that elevating the temperature beyond this threshold correlates directly with an increase in impurity profiles, likely due to accelerated degradation or side reactions of the sensitive quinone moiety. By adhering to these low-temperature conditions, the process preserves the integrity of the pyrroloquinoline core while ensuring complete saponification of the ester groups.
Following hydrolysis, the introduction of a halide salt, specifically potassium chloride, prior to acidification plays a pivotal role in the purification mechanism. The subsequent adjustment of the pH to exactly 5.3 using hydrochloric acid triggers the selective precipitation of the triacid potassium salt. This specific pH value is not arbitrary; it represents the isoelectric point or solubility minimum for the potassium salt form, allowing it to crash out of the solution while leaving organic impurities and excess reagents in the supernatant. This phenomenon effectively acts as an in-situ purification step, delivering an intermediate of exceptional purity before the final conversion to PQQ. The structural identity of this key intermediate, the triacid salt, is crucial for understanding the isolation efficiency.
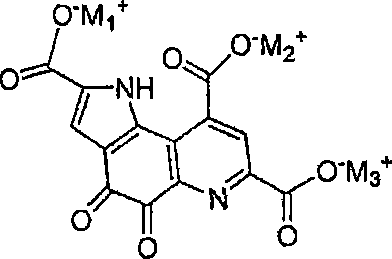
How to Synthesize Pyrroloquinoline Quinone Efficiently
Executing this synthesis requires strict adherence to the nine-step linear sequence outlined in the patent, beginning with the formylation of 2-methoxy-5-nitroaniline and proceeding through reduction, diazotization, and indole formation. The operational success of the route hinges on the meticulous control of reaction parameters in the later stages, particularly the oxidative aromatization and the final hydrolysis. Operators must ensure that the stoichiometry of the oxidizing agents and the timing of the acidification steps are monitored closely to prevent over-oxidation or incomplete hydrolysis. The following guide summarizes the critical operational phases required to replicate this high-yield process in a GMP-compliant environment.
- Perform linear synthesis starting from 2-methoxy-5-nitroaniline through formylation, reduction, and Japp-Klingemann reaction to form the indole core.
- Condense the indole intermediate with dimethyl 2-oxoglutaconate followed by oxidative aromatization to construct the pyrroloquinoline skeleton.
- Execute the critical final hydrolysis at 16-17°C using LiOH, precipitate the triacid potassium salt at pH 5.3, and convert to PQQ using sulfuric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this patented methodology offers tangible benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the isolation workflow, which translates directly into reduced labor costs and shorter cycle times. By removing the complex two-stage salt separation required by previous methods, manufacturers can significantly lower the consumption of specialized reagents like cesium salts, which are often costly and subject to supply volatility. This streamlining enhances the overall resilience of the supply chain, ensuring a more predictable and stable flow of high-purity PQQ to downstream formulation partners.
- Cost Reduction in Manufacturing: The elimination of the dual-salt isolation step fundamentally alters the cost structure of PQQ production. Without the need for sequential precipitation and filtration of sodium and cesium salts, the process consumes fewer resources and generates less waste, leading to substantial cost savings. Furthermore, the use of common reagents like potassium chloride instead of exotic cesium salts reduces raw material expenditure, making the final product more price-competitive in the global marketplace without compromising on quality standards.
- Enhanced Supply Chain Reliability: Reliance on a simplified, robust synthetic route mitigates the risk of production delays caused by complex purification failures. The ability to consistently precipitate the intermediate at a specific pH ensures batch-to-batch reproducibility, which is essential for maintaining long-term supply contracts. This reliability is crucial for reducing lead time for high-purity PQQ intermediates, allowing manufacturers to respond more swiftly to market demand fluctuations and secure their position as a preferred vendor for major pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, having been demonstrated to produce multi-gram quantities with high efficiency. The reduction in solvent usage and the minimization of heavy metal contaminants (by avoiding cesium) simplify the wastewater treatment protocols, aiding in environmental compliance. This eco-friendly profile aligns with modern green chemistry initiatives, making the manufacturing facility more sustainable and reducing the regulatory burden associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this synthetic route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the operational boundaries and quality outcomes of the process.
Q: How does this patent improve upon the Martin method for PQQ synthesis?
A: The patent eliminates the complex two-stage isolation of sodium and cesium salts found in the Martin method. Instead, it utilizes a controlled pH precipitation of a single potassium triacid salt, significantly simplifying the final purification step.
Q: What is the critical temperature control parameter in the final hydrolysis step?
A: The reaction mixture temperature must be strictly maintained between 16°C and 17°C during the hydrolysis with lithium hydroxide. Deviations above this range have been shown to increase impurity formation.
Q: What purity levels can be achieved with this synthetic route?
A: The method allows for the production of PQQ with high purity, demonstrated in examples to reach 98.9% purity by HPLC analysis after final recrystallization from sulfuric acid and water.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrroloquinoline Quinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of mastering complex synthetic pathways like the one described in CN101193888A to deliver superior value to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications and utilizing rigorous QC labs to verify that every batch of PQQ meets the highest international standards for safety and efficacy.
We invite you to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall procurement costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
