Scalable Biaryl Synthesis Using Reusable Heterogeneous Palladium Catalysts for Commercial Manufacturing
The landscape of organic synthesis for high-value aromatic structures is undergoing a significant transformation, driven by the urgent need for greener, more cost-effective manufacturing protocols. A pivotal advancement in this domain is detailed in patent CN103864567A, which discloses a novel method for preparing coupled arene compounds, specifically biaryls, utilizing a heterogeneous palladium-based catalyst system. This technology addresses critical bottlenecks in traditional cross-coupling reactions by enabling a seamless, two-step continuous process where the activation of the silicon nucleophile and the subsequent carbon-carbon bond formation occur without intermediate isolation. For global pharmaceutical and fine chemical manufacturers, this represents a paradigm shift away from fragile homogeneous catalytic systems toward robust, recyclable heterogeneous alternatives that promise enhanced operational efficiency and reduced environmental impact.
By leveraging nanoporous palladium or supported palladium variants, this methodology facilitates the in-situ conversion of readily available hydrosilanes into reactive silanols or silyl ethers using benign activators like water or alcohols. The strategic integration of these activation steps directly with the coupling phase eliminates the need for complex ligand systems and harsh oxidative conditions typically associated with classical Hiyama couplings. As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative pathways is essential for maintaining competitiveness in a market that increasingly demands sustainable and scalable synthetic solutions for complex molecular architectures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methodologies for constructing biaryl scaffolds, such as the ubiquitous Suzuki-Miyaura or Stille couplings, while effective, are fraught with significant logistical and economic challenges that hinder optimal commercial scalability. These conventional routes typically rely on homogeneous palladium catalysts, such as palladium acetate or tetrakis(triphenylphosphine)palladium(0), which are notoriously sensitive to oxygen and moisture, necessitating expensive inert atmosphere equipment and rigorous exclusion of air throughout the reaction lifecycle. Furthermore, the nucleophilic partners in these reactions, often organoboron or organotin reagents, present their own set of difficulties; organoboron species can be thermally unstable and prone to protodeboronation, while organotin reagents are highly toxic and pose severe regulatory and disposal hurdles in modern GMP manufacturing environments. The separation of residual homogeneous palladium from the final product is another critical pain point, often requiring costly scavenging resins or extensive chromatographic purification to meet stringent heavy metal limits imposed by regulatory bodies for active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the technology outlined in CN103864567A introduces a streamlined approach that circumvents these historical limitations through the deployment of stable, heterogeneous palladium catalysts. This novel strategy allows for the direct utilization of hydrosilanes, which are generally more stable and easier to handle than their boron or tin counterparts, and activates them using simple, green reagents like water or methanol. The true brilliance of this system lies in its telescoped nature; the catalyst performs the dual function of activating the silicon center and facilitating the cross-coupling with the halogenated arene in a single reactor vessel. This eliminates the need for isolating the intermediate silanol or silyl ether, thereby reducing solvent consumption, minimizing material loss during transfer, and significantly shortening the overall production cycle time. Moreover, the heterogeneous nature of the catalyst means it can be recovered via simple filtration, washed, and reused, offering a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing by lowering both raw material and waste disposal expenses.
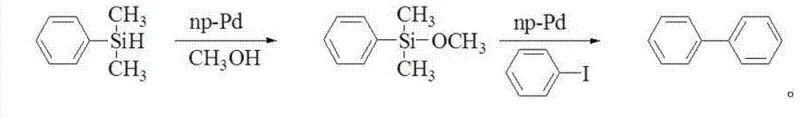
Mechanistic Insights into Heterogeneous Palladium-Catalyzed Silane Activation and Coupling
The mechanistic underpinning of this transformative process involves a sophisticated interplay between the heterogeneous palladium surface and the silicon-hydrogen bond of the aryl silane substrate. In the initial activation phase, the nanoporous palladium catalyst, characterized by its high surface area and specific pore dimensions ranging from 2 to 500 nanometers, facilitates the oxidative addition or sigma-bond metathesis of the Si-H bond. When conducted in an alcoholic medium like methanol or ethanol, this interaction leads to the evolution of hydrogen gas and the formation of the corresponding alkoxysilane species. Alternatively, in the presence of water, the catalyst promotes the hydrolysis of the Si-H bond to generate the highly reactive aryl silanol intermediate. This activation step is crucial because unactivated hydrosilanes are generally inert towards transmetallation in cross-coupling cycles; the conversion to a hypervalent silicon species (silanol or silicate) is the key that unlocks their nucleophilic potential for the subsequent carbon-carbon bond formation.
Following the in-situ generation of the activated silicon species, the introduction of the base and the aryl halide initiates the catalytic coupling cycle on the catalyst surface. The base, such as tetrabutylammonium fluoride (TBAF) or cesium carbonate, likely assists in forming a pentacoordinate or hexacoordinate silicate complex, which enhances the nucleophilicity of the aryl group attached to the silicon. This activated complex then undergoes transmetallation with the palladium center, which has concurrently undergone oxidative addition with the aryl halide. The final reductive elimination step releases the desired biaryl product and regenerates the active palladium(0) species on the solid support. This mechanism not only ensures high selectivity and yields reaching up to 95% but also inherently suppresses the formation of toxic by-products often seen in stoichiometric activation methods, thereby simplifying the impurity profile and easing the burden on downstream purification processes.
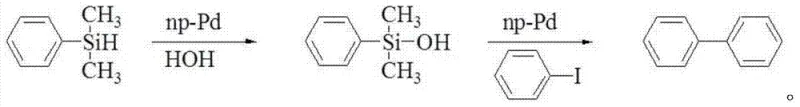
How to Synthesize Biphenyl Efficiently
The practical implementation of this synthesis route is designed for operational simplicity and robustness, making it highly attractive for process chemists aiming to transition from bench-scale discovery to pilot plant operations. The protocol essentially divides the synthesis into two distinct thermal phases within the same reaction vessel, optimizing conditions for both activation and coupling sequentially. By avoiding the isolation of the moisture-sensitive or thermally unstable intermediates, the process minimizes exposure to potential degradation pathways and reduces the overall solvent footprint. For detailed operational parameters, including specific stoichiometric ratios, temperature ramps, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the critical process parameters defined in the patent literature.
- Activate the aryl silane precursor by reacting it with either water or an alcohol solvent in the presence of a heterogeneous palladium catalyst at moderate temperatures (20-80°C) for 2-4 hours.
- Without isolating the intermediate silanol or silyl ether, introduce the halogenated arene coupling partner and a base compound directly into the reaction mixture.
- Heat the combined reaction solution to elevated temperatures (50-150°C) and maintain for 12-36 hours to complete the cross-coupling, followed by standard workup and catalyst recovery.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement and supply chain perspective, the adoption of this heterogeneous catalytic methodology offers profound advantages that extend far beyond mere chemical yield improvements. The ability to utilize a single catalyst for multiple sequential transformations fundamentally alters the cost structure of biaryl production by consolidating unit operations. Traditional multi-step syntheses often incur significant costs related to intermediate isolation, drying, and re-dissolution, all of which consume time, energy, and solvents. By telescoping these steps, manufacturers can achieve a drastic simplification of the production workflow, leading to substantial reductions in labor hours and facility occupancy time. Furthermore, the use of benign activators like water or common alcohols replaces expensive and hazardous specialized reagents, contributing to a safer working environment and lower raw material procurement costs.
- Cost Reduction in Manufacturing: The economic impact of switching to this heterogeneous system is driven primarily by the eliminaton of expensive ligand systems and the dramatic reduction in catalyst loading costs due to recyclability. Unlike homogeneous catalysts which are lost in the aqueous waste stream or require complex recovery, the solid palladium catalyst described in this patent can be filtered, washed with solvents like tetrahydrofuran and water, and directly reused in subsequent batches. This closed-loop catalyst usage significantly lowers the cost of goods sold (COGS) over the lifecycle of the product. Additionally, the avoidance of toxic organotin or difficult-to-handle organoboron reagents reduces the regulatory compliance costs associated with hazardous material handling and waste disposal, further enhancing the overall profit margin for high-volume production runs.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available and stable starting materials such as hydrosilanes and simple aryl halides, which are commodity chemicals with robust global supply networks. The insensitivity of the heterogeneous catalyst to air and moisture means that storage and handling requirements are less stringent compared to air-sensitive homogeneous catalysts, reducing the risk of supply disruptions due to improper storage or transportation conditions. This robustness ensures consistent batch-to-batch quality and reliability, which is critical for maintaining uninterrupted supply to downstream API manufacturers. The simplified process flow also reduces the dependency on specialized equipment for inert atmosphere processing, allowing for greater flexibility in manufacturing site selection and capacity allocation.
- Scalability and Environmental Compliance: The environmental profile of this process aligns perfectly with modern green chemistry principles, making it easier to secure regulatory approvals and maintain social license to operate. The generation of hydrogen gas as the sole byproduct during the activation phase is a clean alternative to the heavy metal waste generated by traditional oxidation methods. The heterogeneous nature of the catalyst ensures that metal leaching into the product is minimized, simplifying the purification process and ensuring that the final product meets strict heavy metal specifications required for pharmaceutical applications. This inherent cleanliness facilitates easier scale-up from kilogram to multi-ton scales without the exponential increase in waste treatment costs that often plagues traditional coupling chemistries, ensuring long-term sustainability and compliance with evolving environmental regulations.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing portfolios, we have compiled a set of frequently asked questions based on the specific technical disclosures and experimental data provided in the patent documentation. These answers address common concerns regarding catalyst performance, substrate scope, and operational parameters, providing a clear framework for assessing the potential return on investment for adopting this synthetic route. Understanding these nuances is vital for R&D teams planning process validation and for procurement teams negotiating raw material specifications.
Q: What are the primary advantages of using heterogeneous palladium catalysts over homogeneous systems in biaryl synthesis?
A: Heterogeneous catalysts offer superior stability in air and moisture, eliminating the need for rigorous inert atmosphere conditions. Crucially, they can be easily separated via simple filtration and reused multiple times without significant loss of activity, drastically reducing catalyst consumption costs and simplifying downstream purification.
Q: Can this method utilize water as a reagent, and what are the environmental implications?
A: Yes, the process uniquely allows water to act as an oxidant to convert hydrosilanes into reactive silanols, producing only hydrogen gas as a byproduct. This represents a significant green chemistry advancement over traditional methods that often require toxic oxidants or generate hazardous waste streams.
Q: Is this synthesis route suitable for large-scale commercial production of API intermediates?
A: Absolutely. The protocol operates under relatively mild conditions and utilizes robust, commercially available catalysts like nanoporous palladium or Pd/C. The one-pot nature of the activation and coupling steps minimizes unit operations, making it highly amenable to scale-up from kilogram to multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results observed in patent literature can be reliably translated into industrial reality. We maintain stringent purity specifications through our state-of-the-art rigorous QC labs, employing advanced analytical techniques to monitor impurity profiles and ensure that every batch of biaryl intermediate meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality is matched only by our dedication to process safety and environmental stewardship, making us an ideal partner for long-term supply agreements.
We invite you to engage with our technical team to explore how this advanced heterogeneous catalytic technology can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis to quantify the potential economic benefits for your specific molecule or need to review specific COA data from our pilot trials, our experts are ready to assist. Please contact our technical procurement team today to request route feasibility assessments and discuss how we can collaborate to optimize your supply chain for biaryl compounds, ensuring both cost efficiency and supply security for your critical drug development programs.
