Revolutionizing Naproxen Intermediate Production via Continuous Flow Microchannel Oxidation
Introduction to Advanced Naproxen Synthesis
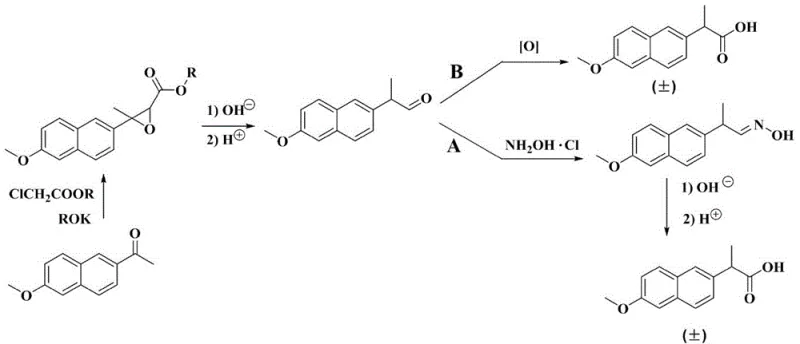
The pharmaceutical industry is constantly seeking robust methodologies to enhance the safety and efficiency of active pharmaceutical ingredient (API) manufacturing. Patent CN113845417A introduces a groundbreaking approach for synthesizing (+/-)-naproxen, a critical nonsteroidal anti-inflammatory drug (NSAID) intermediate, utilizing a continuous flow microchannel reactor. This technology addresses the longstanding challenges associated with the oxidation of 2-(6-methoxy-2-naphthyl) propionaldehyde, a key precursor in the naproxen supply chain. By transitioning from traditional batch processing to a continuous flow regime, this method offers unprecedented control over reaction parameters such as temperature, pressure, and residence time. The innovation lies in the ability to manage highly exothermic oxidation reactions within a confined micro-environment, thereby mitigating safety risks while simultaneously improving product quality and yield. For global supply chain leaders, this represents a significant leap forward in process intensification, promising a more reliable source of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for naproxen often rely on batch reactors, which present inherent limitations when handling vigorous oxidation reactions. Conventional methods, such as the acetylation process followed by oximation and hydrolysis, involve multiple steps that generate substantial amounts of industrial waste, including废气 (waste gas), wastewater, and solid residues. Specifically, the direct oxidation of the aldehyde intermediate in batch tanks using oxidants like potassium permanganate or chromium trioxide poses severe safety hazards due to poor heat dissipation. These batch processes frequently suffer from localized overheating, leading to uncontrolled exotherms that can result in catastrophic equipment failure or explosions. Furthermore, the prolonged reaction times in large vessels increase the likelihood of over-oxidation and the formation of chlorinated byproducts, necessitating complex and costly purification steps to meet stringent pharmaceutical purity standards. The reliance on excessive amounts of scavengers in batch Pinnick oxidations further exacerbates cost and environmental concerns.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the unique advantages of continuous flow chemistry to overcome these historical bottlenecks. By employing a silicon carbide microchannel reactor, the process ensures rapid and homogeneous mixing of the aldehyde substrate and the oxidant solution. This intimate contact allows for precise stoichiometric control and immediate heat removal, effectively eliminating the hot spots that plague batch reactors. The continuous flow system operates under mild conditions with residence times measured in seconds rather than hours, which dramatically suppresses side reactions such as over-oxidation and ring chlorination. Consequently, the process achieves exceptional conversion rates and selectivity without the need for hazardous heavy metal catalysts or excessive scavenger loads. This technological shift not only simplifies the downstream workup but also aligns perfectly with green chemistry principles, offering a sustainable and economically viable pathway for the large-scale production of naproxen intermediates.
Mechanistic Insights into Microchannel Oxidation
The core of this innovation lies in the mechanistic efficiency of the Pinnick-type oxidation performed within a micro-reactor environment. In this system, 2-(6-methoxy-2-naphthyl) propionaldehyde reacts with sodium chlorite (NaClO2) in the presence of a hypochlorous acid scavenger, such as 2-methyl-2-butene. The microchannel geometry facilitates diffusion-controlled mixing, ensuring that the concentration of reactive species remains uniform throughout the reaction zone. This uniformity is critical because it prevents the accumulation of hypochlorous acid, a potent byproduct that can otherwise oxidize the electron-rich methoxy naphthalene ring or consume additional chlorite ions. The rapid quenching of the reaction mixture immediately upon exiting the reactor further stabilizes the product, preventing degradation during the transition to the workup phase. The use of silicon carbide as the reactor material provides exceptional chemical resistance and thermal conductivity, allowing the process to operate safely even with aggressive oxidants.
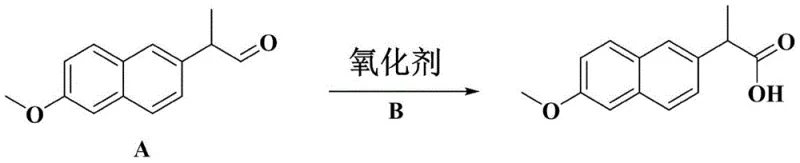
Furthermore, the impurity profile is meticulously managed through the precise control of pH and residence time. In batch systems, fluctuations in pH can lead to the formation of chlorine dioxide radicals, which initiate uncontrollable radical chain reactions. The continuous flow setup maintains a stable acidic environment (pH 2-6) throughout the microchannel, ensuring that the oxidation proceeds via the desired ionic mechanism rather than radical pathways. This control is instrumental in achieving the reported purity levels of greater than 99%, as it effectively minimizes the generation of chlorinated impurities and over-oxidized species. For R&D directors, understanding this mechanistic advantage is crucial, as it validates the scalability of the process from laboratory screening to commercial manufacturing without the fear of impurity spikes that often accompany scale-up in batch processes.
How to Synthesize (+/-)-Naproxen Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this continuous flow technology in a production setting. The process begins with the preparation of two distinct feed streams: Material A, containing the aldehyde substrate and scavenger dissolved in a solvent like sec-butyl alcohol, and Material B, consisting of the aqueous sodium chlorite oxidant adjusted to a specific pH. These streams are then pumped simultaneously into the microchannel reactor using metering pumps, where they mix and react under controlled temperature and pressure conditions. The reaction mixture exits the reactor after a short residence time and is immediately quenched with a reducing agent such as sodium thiosulfate to neutralize any remaining oxidant. Following quenching, a standard liquid-liquid extraction and acidification procedure yields the final crude naproxen product, which can be further purified if necessary.
- Prepare Material A by dissolving 2-(6-methoxy-2-naphthyl) propionaldehyde and a hypochlorous acid scavenger (e.g., 2-methyl-2-butene) in a solvent like sec-butyl alcohol.
- Prepare Material B by adjusting an aqueous sodium chlorite solution to pH 2-6 using acetic acid or buffer salts.
- Pump both solutions into a microchannel reactor at controlled flow rates, maintain temperature between 0-100°C for 60-300 seconds, and quench the effluent with sodium thiosulfate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this continuous flow technology translates into tangible strategic benefits that extend beyond mere technical performance. The primary advantage lies in the drastic reduction of operational risks associated with handling hazardous oxidation reactions. By minimizing the inventory of reactive chemicals within the reactor at any given moment, the facility's overall safety profile is significantly enhanced, potentially lowering insurance premiums and regulatory compliance burdens. Additionally, the elimination of heavy metal catalysts and the reduction in scavenger consumption directly contribute to cost reduction in pharmaceutical intermediates manufacturing. The simplified post-treatment process, which avoids complex distillation or chromatography steps often required to remove batch-generated impurities, further streamlines operations and reduces utility consumption. This efficiency gain allows for a more competitive pricing structure while maintaining high margins.
- Cost Reduction in Manufacturing: The continuous flow process eliminates the need for expensive heavy metal oxidants and significantly reduces the molar equivalents of scavengers required compared to batch methods. This reduction in raw material intensity, combined with the higher yield and purity, leads to substantial cost savings per kilogram of produced API intermediate. Furthermore, the energy efficiency of the microreactor, due to its superior heat exchange capabilities, lowers the overall utility costs associated with heating and cooling large reaction masses. The simplified workup procedure also reduces labor and solvent costs, making the entire production cycle more economically attractive for large-scale contracts.
- Enhanced Supply Chain Reliability: Continuous manufacturing inherently offers greater consistency and reliability compared to batch processing. The steady-state operation of the microchannel reactor ensures that every unit of product meets the same rigorous quality specifications, reducing the variability that often leads to batch rejections or delays. This consistency is vital for maintaining uninterrupted supply lines to downstream formulation partners. Moreover, the compact footprint of the flow chemistry equipment allows for flexible production scheduling and easier capacity expansion without the need for massive infrastructure investments. This agility enables suppliers to respond more rapidly to fluctuating market demands for naproxen and related NSAIDs.
- Scalability and Environmental Compliance: Scaling up continuous flow processes is fundamentally different and often simpler than scaling batch reactions, as it typically involves numbering up reactors rather than increasing vessel size. This approach preserves the optimal reaction conditions achieved at the laboratory scale, ensuring that commercial production maintains the same high yield and purity. From an environmental perspective, the process generates significantly less waste, particularly regarding heavy metal contamination and organic solvent usage. The reduced generation of hazardous byproducts simplifies waste treatment protocols and ensures compliance with increasingly stringent global environmental regulations, safeguarding the long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial implications of this patented synthesis method. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on how this technology compares to established industry standards. Understanding these details is essential for stakeholders evaluating the feasibility of integrating continuous flow oxidation into their existing supply chains.
Q: How does the microchannel reactor improve safety compared to batch oxidation?
A: The microchannel reactor significantly enhances safety by minimizing the hold-up volume of reactive materials and providing superior heat transfer capabilities. This precise thermal control prevents the runaway exothermic reactions often associated with traditional batch oxidation processes using strong oxidants.
Q: What are the purity levels achievable with this continuous flow method?
A: According to patent data, this method consistently achieves product purity exceeding 99% and yields greater than 95%. The short residence time effectively suppresses over-oxidation and chlorination side reactions that typically degrade product quality in batch systems.
Q: Does this process reduce the consumption of scavengers?
A: Yes, the continuous flow nature of the reaction drastically reduces the required amount of hypochlorous acid scavengers. The rapid mixing and short reaction time minimize the generation of byproduct hypochlorous acid, thereby lowering raw material costs and waste treatment burdens.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naproxen Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of continuous flow technology in the synthesis of complex pharmaceutical intermediates like naproxen. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust manufacturing operations. Our state-of-the-art facilities are equipped with advanced flow chemistry reactors and stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards. We are committed to delivering high-purity naproxen intermediates that empower our clients to bring safer and more effective medications to market faster.
We invite you to collaborate with us to explore how this advanced oxidation technology can optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in continuous flow chemistry can become a cornerstone of your strategic sourcing strategy.
