Advanced Green Synthesis of Alpha-Acyloxy Thioether Derivatives for Commercial Scale-Up
Advanced Green Synthesis of Alpha-Acyloxy Thioether Derivatives for Commercial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for greener, more efficient synthetic routes that minimize environmental impact while maximizing yield. A significant breakthrough in this domain is detailed in patent CN112898188A, which discloses a novel method for preparing alpha-acyloxy thioether derivatives. These compounds serve as vital building blocks in organic synthesis, particularly for constructing complex aldehyde and ketone architectures found in bioactive molecules. The disclosed technology represents a paradigm shift by utilizing a simple, one-pot reaction system involving carboxylic acids, their sodium salts, and the commercial reagent Selectfluor. This approach effectively bypasses the traditional reliance on toxic sulfoxide precursors, offering a streamlined pathway that aligns perfectly with modern green chemistry principles and the rigorous demands of the global pharmaceutical supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-acyloxy thioethers has been plagued by significant operational and safety challenges that hinder large-scale adoption. The primary conventional strategy involves the Pummerer rearrangement, which necessitates the use of sulfoxide compounds as starting materials. The preparation of these sulfoxides often requires separate oxidation steps involving hazardous oxidants, introducing unnecessary complexity and toxicity into the manufacturing process. Furthermore, alternative methods utilizing thioether compounds typically depend on expensive hypervalent iodine reagents or transition metal catalysts to achieve the desired oxidative functionalization. These reagents are not only cost-prohibitive for bulk production but also pose difficulties in terms of waste disposal and metal residue removal, which are critical concerns for regulatory compliance in API manufacturing. The cumulative effect of these drawbacks is a process that is economically inefficient and environmentally burdensome.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN112898188A introduces a direct and highly efficient catalytic system that transforms simple thioethers into valuable alpha-acyloxy derivatives. By leveraging the unique reactivity of Selectfluor (1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octane bis(tetrafluoroborate)), the process activates the sulfur center directly without the need for pre-oxidation to sulfoxides. This eliminates an entire synthetic step and removes the associated safety risks of handling unstable sulfoxide intermediates. The reaction proceeds smoothly in acetonitrile at elevated temperatures, utilizing inexpensive carboxylic acids and their sodium salts as the acyloxy source. This simplification of the reagent profile drastically reduces the raw material costs and simplifies the downstream purification process, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Selectfluor-Mediated C-H Functionalization
The success of this transformation relies on a sophisticated yet elegant mechanistic pathway initiated by the electrophilic fluorinating agent. Upon exposure to Selectfluor, the sulfur atom of the N-substituted-2-methylthiobenzamide substrate undergoes activation to form a fluorosulfonium intermediate. This highly reactive species facilitates an intramolecular cyclization, assisted by the sodium carboxylate base, to generate a cyclic cationic intermediate. Subsequent elimination of a proton at the alpha-position of the sulfur atom, followed by N-S bond cleavage, yields a key sulfur cation intermediate. Finally, the nucleophilic attack by the carboxylate anion on this cationic center delivers the target alpha-acyloxy thioether product. This mechanism highlights the dual role of the reagents, where Selectfluor acts as the oxidant and the carboxylate serves as both the base and the nucleophile, ensuring high atom economy.
Crucially, the patent data reveals strict structural requirements that govern the reaction's success, providing valuable insights for impurity control and substrate scope optimization. Comparative experiments demonstrate that the presence of an ortho-amide group with an available N-H proton is indispensable; substrates lacking this specific arrangement, such as simple thioanisole or N,N-diethyl substituted analogs, fail to react. This suggests that the amide group participates in stabilizing the intermediate or directing the reaction trajectory, thereby preventing non-specific side reactions. Understanding this mechanistic constraint allows process chemists to predict substrate compatibility accurately, ensuring that only viable candidates are selected for scale-up, thus minimizing resource wastage and enhancing the overall robustness of the synthetic route.
How to Synthesize Alpha-Acyloxy Thioether Derivatives Efficiently
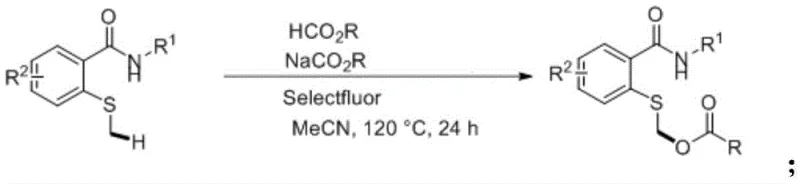
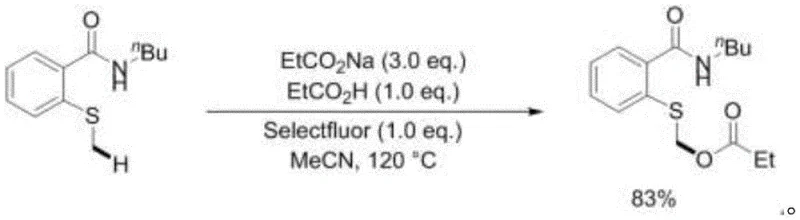
Implementing this novel synthesis route requires precise control over reaction parameters to maximize yield and purity. The standard protocol involves charging a sealed, pressure-resistant tube with the thioether substrate, the corresponding carboxylic acid, its sodium salt, and Selectfluor in acetonitrile. The mixture is then heated to a temperature range of 100°C to 150°C, with 120°C identified as the optimal condition for balancing reaction rate and safety. Maintaining these conditions for 12 to 36 hours ensures complete conversion of the starting material. Following the reaction, the solvent is removed via distillation, and the crude product is purified using standard column chromatography techniques. For detailed operational specifics and stoichiometric ratios, please refer to the standardized guide below.
- Charge a sealed pressure-resistant tube with acetonitrile solvent, N-substituted-2-methylthiobenzamide, carboxylic acid, sodium carboxylate, and Selectfluor reagent.
- Heat the reaction mixture to a temperature between 100°C and 150°C, preferably 120°C, and stir vigorously for 12 to 36 hours.
- Upon completion, distill off the acetonitrile solvent and purify the crude residue via column chromatography using dichloromethane as the developing solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the substantial simplification of the supply chain for raw materials. By replacing expensive and potentially unstable hypervalent iodine reagents or pre-formed sulfoxides with commodity chemicals like carboxylic acids and Selectfluor, manufacturers can significantly mitigate supply risk. These reagents are widely available from multiple global vendors, ensuring continuity of supply even during market fluctuations. Furthermore, the elimination of toxic sulfoxide precursors reduces the regulatory burden associated with hazardous material handling and storage, leading to lower operational overheads and improved workplace safety standards.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the replacement of high-cost reagents with inexpensive alternatives. Traditional methods relying on hypervalent iodine involve reagents that are not only pricey but often have low atom economy, generating significant waste. In contrast, this new method utilizes cheap carboxylic acids and a catalytic amount of base, drastically lowering the bill of materials. Additionally, the simplified workup procedure, which avoids complex extraction steps often needed to remove metal catalysts or iodine byproducts, reduces solvent consumption and energy usage. These factors combine to deliver a manufacturing process with a significantly reduced cost footprint, enhancing the margin potential for downstream API production.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, commercially mature reagents. Selectfluor is a stable, shelf-stable solid that is easy to transport and store, unlike liquid oxidants that may require special containment. The reaction tolerates a variety of substituents on the aromatic ring and the amide nitrogen, as evidenced by the successful synthesis of derivatives with alkyl, aryl, and heteroaryl groups. This versatility means that a single manufacturing platform can be adapted to produce a wide library of intermediates, reducing the need for specialized equipment or distinct process lines. Such flexibility allows suppliers to respond rapidly to changing customer demands without compromising on delivery timelines.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns with the principles of green chemistry by minimizing waste generation and avoiding toxic heavy metals. The absence of transition metal catalysts eliminates the need for expensive and time-consuming metal scavenging steps, which are often a bottleneck in GMP manufacturing. The reaction produces benign byproducts that are easier to treat in standard wastewater facilities. Moreover, the use of acetonitrile, a common industrial solvent, facilitates easy recovery and recycling. These attributes make the process highly scalable from kilogram to multi-ton production, ensuring that environmental compliance costs remain manageable while supporting sustainable growth initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity for R&D teams evaluating this route for their specific projects. Understanding these nuances is essential for successful technology transfer and process validation.
Q: What are the critical structural requirements for the substrate in this synthesis?
A: The patent data indicates that the ortho-amide group is essential for the reaction to proceed. Comparative examples demonstrate that substrates lacking the amide group in the ortho position, or those with N,N-disubstituted amides lacking an N-H proton, fail to yield the desired alpha-acyloxy thioether product.
Q: Why is Selectfluor necessary for this transformation?
A: Selectfluor acts as the crucial electrophilic fluorinating agent that generates the fluorine cation intermediate required to activate the sulfur center. Control experiments without Selectfluor resulted in zero conversion, proving that the carboxylic acid and base alone are insufficient to drive the oxidative functionalization of the thioether.
Q: How does this method improve upon traditional Pummerer rearrangement processes?
A: Traditional methods often rely on pre-synthesized sulfoxide raw materials which can be toxic and require additional synthetic steps. This novel approach utilizes readily available thioethers and carboxylic acids directly, eliminating the need for hazardous sulfoxide precursors and simplifying the overall process flow for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Acyloxy Thioether Derivative Supplier
As the demand for high-quality pharmaceutical intermediates continues to rise, partnering with a technically proficient manufacturer is crucial for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the one described in CN112898188A to deliver superior products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without sacrificing quality. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of alpha-acyloxy thioether derivative meets the highest international standards for potency and impurity profiles.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this green synthesis route can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you navigate the complexities of fine chemical sourcing with confidence and precision.
