Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Cost-Effective Iron Catalysis for Global Pharma Supply Chains
Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Cost-Effective Iron Catalysis for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust and economical pathways to access nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which enhance metabolic stability and lipophilicity. A significant advancement in this domain is detailed in patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. This technology represents a paradigm shift from traditional noble-metal catalysis to an earth-abundant iron-based system, addressing critical pain points in cost and scalability. The disclosed method utilizes readily available isatin derivatives and trifluoroethylimidoyl chloride as key building blocks, catalyzed by ferric chloride under relatively mild thermal conditions. For R&D directors and procurement specialists, this innovation offers a compelling value proposition by simplifying the supply chain for complex oncology and anti-inflammatory intermediates.
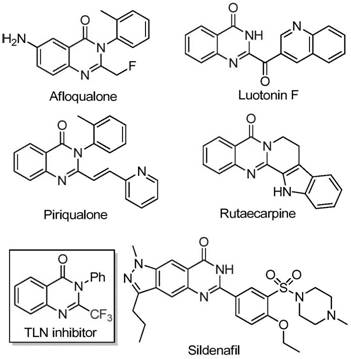
Quinazolinone scaffolds are ubiquitous in medicinal chemistry, serving as the core structure for numerous bioactive agents ranging from Afloqualone to Luotonin F. The introduction of a trifluoromethyl moiety at the 2-position further augments the pharmacological profile of these molecules, often resulting in improved bioavailability and binding affinity. However, accessing these specific substitution patterns has historically been challenging due to the reliance on expensive fluorinating reagents or harsh cyclization conditions. The methodology outlined in the patent overcomes these barriers by employing a tandem decarbonylation and cyclization strategy. This approach not only streamlines the synthetic sequence but also minimizes the generation of hazardous waste, aligning with modern green chemistry principles that are increasingly mandated by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted quinazolinones has relied heavily on the cyclization of anthranilamides or isatoic anhydrides with trifluoroacetic anhydride or ethyl trifluoroacetate. These conventional routes suffer from several inherent drawbacks that hinder their utility in large-scale manufacturing. Firstly, the reaction conditions are often severe, requiring strong bases or high temperatures that can lead to the decomposition of sensitive functional groups. Secondly, the atom economy is frequently poor, generating stoichiometric amounts of acidic or saline byproducts that complicate downstream purification. Furthermore, the starting materials, particularly specialized trifluoromethyl synthons, can be prohibitively expensive and subject to supply chain volatility. These factors collectively increase the cost of goods sold (COGS) and extend the lead time for API production, creating bottlenecks for pharmaceutical developers.
The Novel Approach
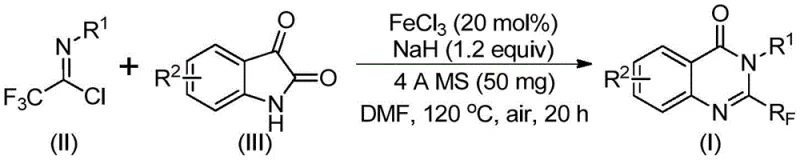
In stark contrast, the novel approach described in CN111675662B leverages a synergistic combination of sodium hydride and ferric chloride to drive the reaction forward efficiently. By utilizing trifluoroethylimidoyl chloride and isatin as precursors, the method bypasses the need for pre-functionalized anthranilic acid derivatives. The reaction proceeds through an initial base-promoted coupling followed by an iron-catalyzed intramolecular cyclization. This two-stage process occurs in a single pot, significantly reducing operational complexity. The use of DMF as a solvent ensures excellent solubility for both polar and non-polar intermediates, facilitating homogeneous reaction kinetics. Most importantly, the tolerance for diverse substituents on the aromatic rings allows for the rapid generation of structural analogs, accelerating the drug discovery process without the need for extensive route re-optimization.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay between base-mediated nucleophilic attack and Lewis acid catalysis. Initially, sodium hydride deprotonates the isatin nitrogen, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step forms a transient trifluoroacetamidine intermediate. Subsequently, the ferric chloride catalyst coordinates with the carbonyl oxygen and the imine nitrogen, activating the system for decarbonylation. This crucial step releases carbon monoxide and drives the equilibrium towards the formation of the quinazolinone ring. The iron center likely stabilizes the transition state, lowering the activation energy required for the cyclization. Understanding this mechanism is vital for process chemists, as it highlights the importance of maintaining anhydrous conditions to prevent catalyst deactivation and ensuring the correct stoichiometric balance between the base and the catalyst.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed alternatives. The mild nature of the iron catalyst reduces the likelihood of side reactions such as polymerization or over-fluorination, which are common pitfalls in trifluoromethylation chemistry. The patent data indicates that the reaction is highly selective for the desired 2-trifluoromethyl isomer, minimizing the formation of regioisomers that are difficult to separate. Additionally, the use of 4A molecular sieves in the reaction mixture plays a critical role in scavenging trace moisture, which could otherwise hydrolyze the imidoyl chloride starting material. This attention to detail in the reaction design ensures a cleaner crude profile, thereby reducing the burden on purification teams and improving the overall recovery of the final active pharmaceutical ingredient.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis requires careful attention to reagent quality and thermal management to ensure reproducibility and safety. The protocol involves a sequential addition of reagents to manage the exothermic nature of the initial deprotonation step. Operators must ensure that the sodium hydride is handled under inert atmosphere to prevent ignition, while the subsequent heating phases require precise temperature control to optimize the cyclization yield. The patent documentation provides a robust framework for scaling this reaction, detailing specific molar ratios and solvent volumes that have been empirically validated. For a comprehensive understanding of the operational parameters, please refer to the standardized synthesis guide below.
- Combine ferric chloride catalyst, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in an anhydrous organic solvent such as DMF.
- Stir the reaction mixture initially at a lower temperature (around 40°C) for approximately 8-10 hours to facilitate initial bond formation.
- Heat the reaction to 120°C and maintain for 18-20 hours to complete the decarbonylation and cyclization, followed by filtration and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed route translates into tangible strategic benefits beyond mere technical feasibility. The shift from precious metals to base metals fundamentally alters the cost structure of the intermediate. Iron salts are commodities with stable pricing, unlike palladium or rhodium which are subject to geopolitical fluctuations and mining constraints. This stability allows for more accurate long-term budgeting and reduces the risk of cost overruns during multi-ton campaigns. Furthermore, the simplicity of the workup procedure, which involves standard filtration and chromatography, eliminates the need for specialized scavenger resins often required to remove trace noble metals. This simplification accelerates the batch cycle time, enabling faster turnover and improved asset utilization in multipurpose manufacturing plants.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the use of commodity chemicals like isatin and DMF result in a drastic reduction in raw material expenditures. The process avoids the need for costly ligand systems often associated with transition metal cross-coupling reactions, further driving down the variable costs per kilogram. Additionally, the high conversion rates observed in the patent examples suggest minimal waste of starting materials, optimizing the overall material efficiency of the process.
- Enhanced Supply Chain Reliability: The starting materials, specifically isatin derivatives and trifluoroethylimidoyl chlorides, are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, ensuring consistent output even when sourcing from different vendors. This resilience is crucial for maintaining continuous supply lines for critical API intermediates in a volatile global market.
- Scalability and Environmental Compliance: The reaction operates at atmospheric pressure and uses standard heating equipment, making it easily transferable from laboratory glassware to industrial stainless steel reactors. The absence of toxic heavy metals simplifies the environmental compliance landscape, reducing the costs associated with wastewater treatment and hazardous waste disposal. The process aligns well with sustainability goals, as the iron catalyst is less toxic and the overall E-factor of the process is favorable compared to traditional multi-step syntheses.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic methodology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the fit of this technology within their existing manufacturing portfolios and R&D pipelines.
Q: What are the primary advantages of using FeCl3 over precious metal catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers significant economic advantages as it is an abundant, non-precious base metal catalyst. Unlike palladium or rhodium systems, iron catalysts drastically reduce raw material costs and simplify the removal of heavy metal residues, which is critical for meeting stringent pharmaceutical purity specifications.
Q: Can this synthetic route tolerate diverse functional groups on the aromatic ring?
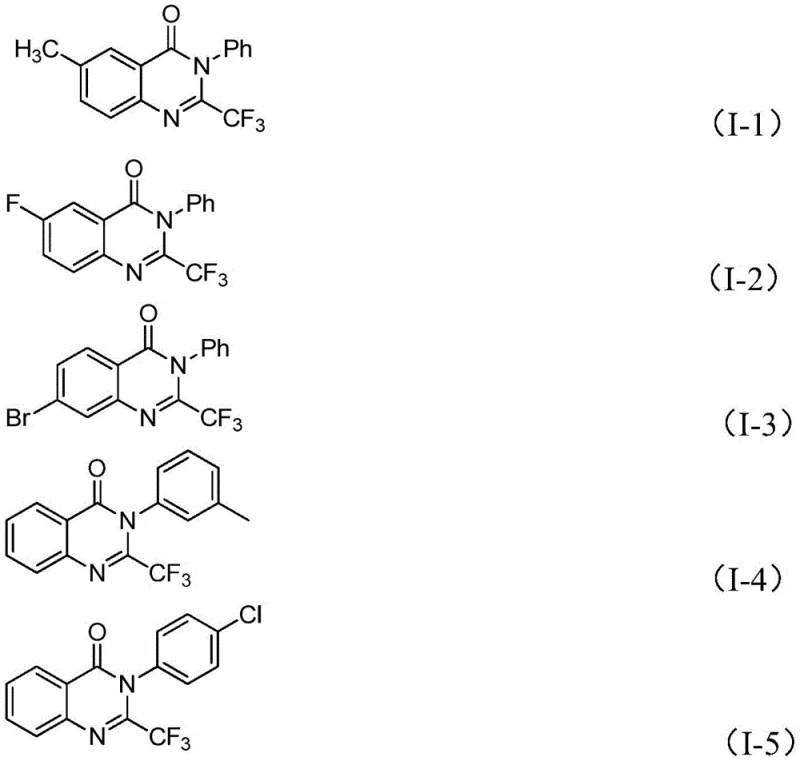
A: Yes, the methodology described in patent CN111675662B demonstrates excellent functional group tolerance. The reaction successfully accommodates various substituents including halogens (fluorine, chlorine, bromine), alkyl groups (methyl), and electron-withdrawing groups (nitro) at ortho-, meta-, and para-positions without compromising the cyclization efficiency.
Q: Is this process suitable for large-scale industrial manufacturing?
A: The process is highly amenable to scale-up due to the use of commercially available starting materials like isatin and trifluoroethylimidoyl chloride. The reaction conditions utilize standard heating and stirring protocols in common solvents like DMF, avoiding the need for specialized high-pressure equipment or cryogenic conditions often required in alternative synthetic pathways.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed technology for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess the technical expertise to translate this patent methodology into a GMP-compliant manufacturing process. Our facilities are equipped to handle the specific thermal and safety requirements of this reaction, ensuring that the transition from gram-scale optimization to commercial production is seamless. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with precision and reliability. Our rigorous QC labs enforce stringent purity specifications, ensuring that every batch of 2-trifluoromethyl quinazolinone meets the highest standards required for downstream drug synthesis.
We invite you to collaborate with us to leverage this cost-effective synthetic route for your next project. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you secure not just a supplier, but a strategic ally committed to optimizing your supply chain and accelerating your time to market.
