Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Intermediates for Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those bearing fluorinated motifs which enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in patent CN113045503B, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These scaffolds are ubiquitous in medicinal chemistry, forming the core structure of bioactive molecules such as Methaqualone, Afloqualone, and the natural alkaloid Rutaecarpine. The introduction of the trifluoromethyl group at the 2-position is strategically critical, as it modulates the electronic properties and lipophilicity of the parent molecule, often leading to improved pharmacokinetic profiles. This patent presents a transition metal palladium-catalyzed carbonylation cascade reaction that utilizes cheap and readily available trifluoroethylimidoyl chloride and various amines as starting materials. By leveraging this novel synthetic route, manufacturers can access a diverse library of functionalized quinazolinones with high reaction efficiency and excellent substrate compatibility, addressing a long-standing need for scalable and versatile synthetic tools in API intermediate production.
The limitations of conventional methods for synthesizing 2-trifluoromethyl-substituted quinazolinones have historically hindered their widespread adoption in large-scale manufacturing. Traditional approaches often rely on the cyclization of anthranilamides with ethyl trifluoroacetate or trifluoroacetic anhydride, reactions that frequently suffer from harsh conditions, expensive reagents, and the necessity for pre-activation of substrates. Other methods involving isatoic anhydride or T3P-promoted cascade reactions are similarly constrained by narrow substrate scopes and relatively low yields, making them less attractive for industrial applications where cost and throughput are paramount. In stark contrast, the novel approach described in the patent utilizes a palladium-catalyzed carbonylation strategy that operates under significantly milder and more practical conditions. By employing trifluoroethylimidoyl chloride and amines directly, the process eliminates the need for unstable or costly precursors. The reaction proceeds efficiently in common organic solvents like 1,4-dioxane at 110°C, demonstrating remarkable tolerance for various functional groups including halogens, alkyls, and aryls. This methodological shift not only simplifies the operational workflow but also drastically improves the overall atom economy and yield, with some examples reporting yields as high as 98%, thereby offering a superior alternative for the cost reduction in pharmaceutical intermediate manufacturing.
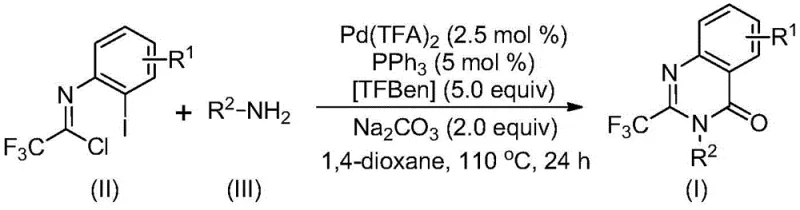
Mechanistic insights into this Pd-catalyzed carbonylation reveal a sophisticated yet streamlined catalytic cycle that ensures high selectivity and purity. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond coupling between the amine and the imidoyl chloride to form a trifluoroacetamidine derivative in situ. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the aromatic ring, generating a divalent palladium intermediate. A crucial step involves the thermal decomposition of TFBen, which releases carbon monoxide safely within the reaction vessel; this CO then inserts into the carbon-palladium bond to form an acyl palladium species. Under the influence of the base, this intermediate undergoes cyclization to form a seven-membered ring palladium complex, followed by reductive elimination to release the final 2-trifluoromethyl-substituted quinazolinone product. This intricate mechanism allows for precise control over the reaction pathway, minimizing the formation of side products and ensuring a clean impurity profile. For R&D directors, understanding this mechanism is vital as it highlights the robustness of the catalytic system, which can accommodate steric and electronic variations in the substrate without compromising the integrity of the final API intermediate, thus facilitating the development of high-purity quinazolinone derivatives.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
To implement this advanced synthetic route in a laboratory or pilot plant setting, operators must adhere to specific procedural guidelines that maximize yield and safety. The process begins with the precise weighing and mixing of the palladium catalyst, ligand, base, and the unique CO source TFBen alongside the key starting materials. The reaction requires sustained heating to drive the carbonylation cascade to completion, followed by a straightforward workup procedure involving filtration and chromatography. While the general protocol is robust, attention to detail regarding solvent quality and temperature control is essential to replicate the high yields reported in the patent literature. For a comprehensive, step-by-step guide tailored to your specific facility capabilities, please refer to the standardized synthesis instructions provided below.
- Combine palladium trifluoroacetate, triphenylphosphine, TFBen, sodium carbonate, trifluoroethylimidoyl chloride, and amine in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 110°C and stir for 16 to 30 hours to allow the carbonylation cascade reaction to proceed to completion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final 2-trifluoromethyl-substituted quinazolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthesis method offers transformative advantages that directly impact the bottom line and operational reliability. The shift towards using commercially available and inexpensive starting materials, such as various amines and trifluoroethylimidoyl chloride, significantly reduces the raw material costs associated with producing these valuable heterocycles. Furthermore, the elimination of hazardous high-pressure carbon monoxide gas in favor of the solid CO surrogate TFBen enhances workplace safety and simplifies regulatory compliance, which is a critical factor for supply chain continuity. The broad substrate scope means that a single manufacturing platform can be adapted to produce a wide variety of derivatives, reducing the need for specialized equipment changes and minimizing downtime. This flexibility allows for a more agile response to market demands, ensuring that critical API intermediates can be supplied consistently without the bottlenecks often associated with complex multi-step syntheses.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the use of cheap, off-the-shelf reagents and the high efficiency of the catalytic system. By avoiding expensive activating agents and pre-functionalized substrates required in traditional methods, the overall cost of goods sold is substantially lowered. Additionally, the high yields observed across a range of substrates mean that less raw material is wasted, further enhancing the cost-effectiveness of the production process. The simplified post-treatment, which involves standard filtration and purification techniques, also reduces labor and processing time, contributing to significant operational savings.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the accessibility of the starting materials, which are widely available from multiple global suppliers. This diversity in sourcing options mitigates the risk of shortages that can plague proprietary or niche reagents. Moreover, the robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production failures or delays due to sensitivity issues. The ability to scale this reaction from gram scale to industrial quantities without losing efficiency provides a reliable pathway for meeting long-term supply contracts and supporting the continuous manufacturing needs of downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is favorable compared to older methods, as it avoids the generation of excessive toxic waste associated with harsh reagents. The use of a solid CO source eliminates the safety hazards and infrastructure costs related to handling toxic gases, making it easier to scale up in standard chemical facilities. The process generates fewer by-products, simplifying waste treatment and disposal procedures. This alignment with green chemistry principles not only meets increasingly stringent environmental regulations but also appeals to eco-conscious stakeholders, positioning the manufacturer as a leader in sustainable chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the practical aspects of adopting this method. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing production workflows and assessing its fit for their specific product portfolios.
Q: What is the primary advantage of using TFBen in this synthesis?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) serves as a safe and effective carbon monoxide substitute, releasing CO in situ under heating conditions to facilitate the carbonylation step without requiring high-pressure gas equipment.
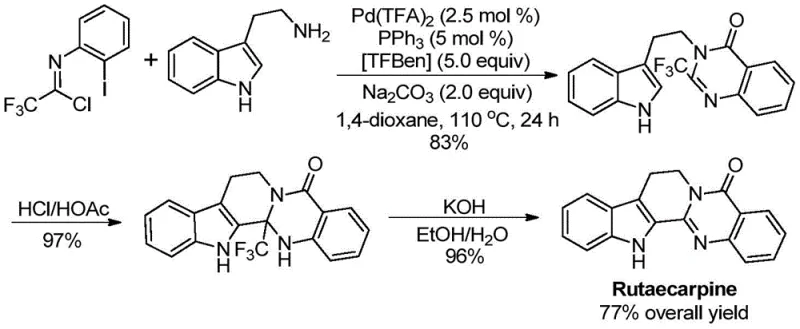
Q: Can this method be applied to complex drug molecules like Rutaecarpine?
A: Yes, the patent explicitly demonstrates the successful application of this method in the high-yield synthesis of Rutaecarpine, achieving a total yield of 77% over three steps, proving its viability for complex heterocyclic drugs.
Q: What are the typical reaction conditions for this transformation?
A: The reaction typically employs Pd(TFA)2 as the catalyst with PPh3 as the ligand in 1,4-dioxane at 110°C for 16 to 30 hours, utilizing sodium carbonate as the base.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and scalable synthetic routes for high-value pharmaceutical intermediates. Our team of expert chemists has extensively evaluated the methodology described in patent CN113045503B and confirmed its potential for delivering high-purity quinazolinone derivatives. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order size. Our state-of-the-art rigorous QC labs and adherence to stringent purity specifications guarantee that every batch meets the exacting standards required for drug development and manufacturing. We are committed to being a partner that not only supplies chemicals but also provides technical solutions that drive your projects forward.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in palladium-catalyzed carbonylation can optimize your supply chain and reduce your overall manufacturing costs.
