Scalable Manufacturing of Thienopyrimidine Derivatives for Advanced Pharmaceutical Applications
The pharmaceutical landscape for cardiovascular and urological treatments has been significantly advanced by the discovery of potent cGMP-specific phosphodiesterase (PDE) inhibitors. Patent CN1346358A discloses a novel class of thieno[2,3-d]pyrimidine compounds and their salts, which exhibit remarkable inhibitory activity against PDE5. These structures serve as critical scaffolds for developing next-generation therapeutics aimed at managing hypertension, heart failure, angina pectoris, and erectile dysfunction. As a leading manufacturer in the fine chemical sector, we recognize the immense commercial potential of these intermediates. The patent outlines a versatile synthetic methodology that allows for extensive structural modification at the 4-position of the pyrimidine ring, enabling the tuning of pharmacokinetic properties while maintaining high potency. This technical insight report analyzes the synthetic feasibility and commercial viability of these compounds for global supply chains.
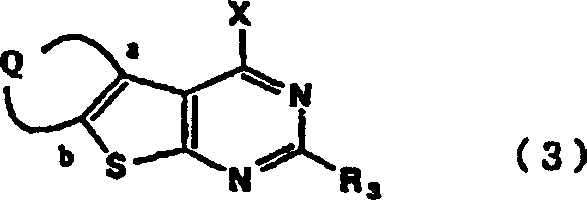
![General formula of thieno[2,3-d]pyrimidine compounds showing variable substituents Q, R1, R2, and R3](/insights/img/thienopyrimidine-pde5-inhibitor-intermediate-supplier-20260306072608-03.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fused heterocyclic systems like thienopyrimidines has presented significant challenges in process chemistry. Conventional routes often rely on harsh cyclization conditions that require high temperatures and strong bases, leading to poor regioselectivity and the formation of difficult-to-remove impurities. Traditional methods for introducing amino groups at the 4-position frequently involve multiple protection and deprotection steps, which drastically increase the step count and reduce overall yield. Furthermore, older methodologies often utilize expensive transition metal catalysts or exotic reagents that complicate waste stream management and increase the cost of goods sold (COGS). The inability to easily purify intermediates via crystallization often forces manufacturers to rely on column chromatography, a technique that is notoriously difficult to scale for commercial production of active pharmaceutical ingredients (APIs).
The Novel Approach
The methodology described in CN1346358A offers a transformative solution to these bottlenecks by employing a direct halogenation-substitution strategy. The core innovation lies in the activation of the thienopyrimidinone precursor using phosphorus oxychloride (POCl3) to generate a highly reactive 4-chloro intermediate. This activated species then undergoes a facile nucleophilic aromatic substitution with various substituted benzylamines. This two-step sequence effectively bypasses the need for complex catalytic cycles or protecting group manipulations. The use of common industrial solvents such as dimethyl sulfoxide (DMSO) and dimethylformamide (DMF) ensures that the reaction can be performed in standard glass-lined or stainless steel reactors. Moreover, the final products often precipitate directly from the reaction mixture upon aqueous workup, facilitating isolation through simple filtration rather than energy-intensive distillation or chromatography.
Mechanistic Insights into Phosphorus Oxychloride-Mediated Activation
The chemical mechanism underpinning this synthesis is a classic example of activating a carbonyl group for nucleophilic displacement. Initially, the oxygen atom of the 4-carbonyl group on the thienopyrimidine ring attacks the phosphorus atom of POCl3, forming an imidoyl chloride intermediate. This transformation converts a poor leaving group (oxide) into an excellent leaving group (chloride), significantly lowering the activation energy for the subsequent substitution step. When the substituted benzylamine is introduced, the nitrogen lone pair attacks the electron-deficient carbon at the 4-position of the heterocyclic ring. The aromaticity of the pyrimidine system is temporarily disrupted to form a Meisenheimer-like complex, which then rapidly collapses to expel the chloride ion and restore aromaticity. This mechanism is highly robust and tolerant of various functional groups on the benzylamine, including methoxy and chloro substituents, as evidenced by the broad scope of compounds exemplified in the patent data.
From an impurity control perspective, this mechanism offers distinct advantages. The primary byproduct of the activation step is phosphoric acid derivatives, which are water-soluble and easily removed during the aqueous quench. The substitution reaction generates hydrochloric acid salts, which are neutralized by the excess amine or added base, forming water-soluble ammonium salts. This inherent cleanliness of the reaction profile means that the crude product typically possesses high purity before any recrystallization. The patent data indicates that simple washing with water and organic solvents like ethyl acetate is sufficient to remove residual starting materials and side products, ensuring that the final intermediate meets the rigorous quality standards required for downstream API synthesis.
How to Synthesize Thienopyrimidine Derivatives Efficiently
The synthesis of these high-value intermediates follows a logical progression designed for operational simplicity and safety. The process begins with the chlorination of the thienopyrimidinone scaffold, followed by the coupling with the appropriate amine component. Detailed protocols regarding stoichiometry, temperature profiles, and workup procedures are critical for maximizing yield and minimizing impurity formation. The following guide summarizes the standardized approach derived from the patent embodiments, providing a roadmap for process chemists looking to implement this technology.
- Halogenation of the thienopyrimidinone precursor using phosphorus oxychloride at elevated temperatures to generate the reactive chloro-intermediate.
- Nucleophilic substitution reaction between the chloro-intermediate and a substituted benzylamine derivative in a polar aprotic solvent like DMSO.
- Purification of the final product through aqueous workup, solvent extraction, and recrystallization to achieve high pharmaceutical grade purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic benefits. The reliance on commodity chemicals such as phosphorus oxychloride, sodium bicarbonate, and common organic solvents ensures a stable and resilient supply chain. Unlike processes dependent on scarce precious metals or specialized ligands, this methodology mitigates the risk of raw material shortages and price volatility. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is not overly sensitive to minor fluctuations in temperature or mixing rates, thereby reducing the likelihood of batch failures and production delays.
- Cost Reduction in Manufacturing: The elimination of expensive catalysts and the reduction in synthetic steps directly contribute to a lower cost of goods. By avoiding chromatographic purification and relying on crystallization for isolation, manufacturers can significantly reduce solvent consumption and waste disposal costs. The high atom economy of the substitution reaction ensures that the majority of the starting mass is incorporated into the final product, optimizing material utilization and reducing the environmental footprint associated with raw material sourcing and processing.
- Enhanced Supply Chain Reliability: The use of widely available reagents means that sourcing can be diversified across multiple global suppliers, preventing single-source bottlenecks. The scalability of the process from laboratory to pilot plant and finally to commercial tonnage is well-supported by the chemistry, as the exothermic nature of the reactions can be managed with standard cooling systems. This predictability allows for accurate lead time forecasting and reliable inventory planning, which is crucial for maintaining continuous API production schedules.
- Scalability and Environmental Compliance: The waste streams generated by this process are primarily aqueous salts and organic solvents that can be readily treated or recycled using standard industrial wastewater treatment facilities. The absence of heavy metal contaminants simplifies the regulatory compliance burden, as there is no need for extensive testing and validation of metal clearance steps. This environmental compatibility aligns with modern green chemistry principles and supports the sustainability goals of major pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these thienopyrimidine intermediates. The answers are derived from the specific experimental data and claims found within the patent documentation, ensuring accuracy and relevance for industry professionals.
Q: What is the primary therapeutic application of these thienopyrimidine compounds?
A: These compounds act as potent and selective inhibitors of cGMP-specific phosphodiesterase (PDE5), making them valuable intermediates for treating cardiovascular conditions such as hypertension, heart failure, and erectile dysfunction.
Q: How does the patented synthesis method improve manufacturing efficiency?
A: The method utilizes a direct halogenation-substitution strategy that avoids complex multi-step sequences, utilizing common industrial reagents like phosphorus oxychloride and standard solvents, which simplifies scale-up and reduces processing time.
Q: What purity levels can be achieved with this synthetic route?
A: The process incorporates robust purification steps including pH-controlled precipitation and recrystallization from organic solvents, allowing for the production of intermediates with stringent purity specifications suitable for API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thienopyrimidine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to bring complex heterocyclic syntheses like those described in CN1346358A to commercial reality. Our team of process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every batch. Our commitment to quality assurance guarantees that the intermediates we supply are fully compliant with international regulatory standards, facilitating a smooth transition into your API manufacturing workflow.
We invite you to collaborate with us to optimize your supply chain for these critical pharmaceutical building blocks. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's efficiency and profitability. Let us be your partner in delivering high-quality thienopyrimidine derivatives to the global market.
