Advanced Crystallization Technology for Ultra-High Purity Trelagliptin Succinate Manufacturing
Advanced Crystallization Technology for Ultra-High Purity Trelagliptin Succinate Manufacturing
The global pharmaceutical landscape is constantly evolving, driven by the demand for higher purity active pharmaceutical ingredients (APIs) that ensure patient safety and therapeutic efficacy. In the realm of diabetes treatment, Trelagliptin Succinate stands out as a critical ultra-long-acting dipeptidyl peptidase IV (DPP-4) inhibitor, marketed under the trade name Zafatek®. A recent technological breakthrough detailed in patent CN112480075A introduces a sophisticated refining method that addresses the persistent challenges of impurity control in DPP-4 inhibitor synthesis. This innovation allows manufacturers to elevate the purity of Trelagliptin Succinate from a standard crude level of 99.76% to an exceptional 99.99% through a optimized crystallization protocol. For R&D directors and procurement specialists, this development signifies a pivotal shift towards more robust, environmentally friendly, and cost-effective manufacturing pathways that align with modern Good Manufacturing Practice (GMP) standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional purification strategies for complex heterocyclic compounds like Trelagliptin often rely on repetitive recrystallization or column chromatography, which can be inherently inefficient and costly. Conventional solvents frequently employed in these processes may possess high toxicity profiles, posing significant risks to operator safety and creating substantial burdens for waste management and environmental compliance. Furthermore, standard cooling crystallization techniques often struggle to discriminate between the target molecule and structurally similar impurities, leading to products that hover around 99.5% to 99.8% purity. This marginal gap can be critical for regulatory approval, necessitating additional processing steps that erode profit margins and extend lead times. The inability to effectively recycle mother liquors in older methods also results in significant material loss, driving up the overall cost of goods sold (COGS) and complicating the supply chain for reliable API intermediate suppliers.
The Novel Approach
The methodology disclosed in the patent presents a paradigm shift by utilizing a precise binary solvent system comprising acetonitrile and ethanol in a 1:1 ratio. This approach leverages the differential solubility characteristics of the target compound versus its impurities across a specific temperature gradient. By dissolving the crude material at a controlled temperature range of 50-60°C and subsequently inducing crystallization at temperatures between 0°C and 10°C (or even down to -10°C), the process maximizes the yield while simultaneously excluding trace contaminants. This novel technique not only simplifies the operational workflow—eliminating the need for complex separation equipment—but also facilitates the recycling of the mother solution solvent. Consequently, this leads to a drastic reduction in solvent consumption and waste generation, positioning this method as a superior choice for cost reduction in pharmaceutical manufacturing and enhancing the sustainability profile of the production facility.
Mechanistic Insights into Crystallization Kinetics and Impurity Rejection
Understanding the physicochemical interactions during the refining of Trelagliptin Succinate is essential for replicating these results on a commercial scale. The core of this mechanism lies in the thermodynamic stability of the crystal lattice formed by the succinate salt of the DPP-4 inhibitor. When the crude material is introduced into the acetonitrile-ethanol mixture at elevated temperatures, the solvation shell around the molecules is disrupted, allowing for a homogeneous distribution. As the system cools, the supersaturation point is reached in a controlled manner, promoting the nucleation of pure Trelagliptin Succinate crystals. The specific polarity and hydrogen bonding capabilities of the ethanol-acetonitrile mix appear to selectively solvate potential impurities, keeping them in the liquid phase while the target molecule precipitates out. This selective precipitation is key to achieving the reported 99.99% purity, as it prevents the occlusion of impurities within the growing crystal matrix, a common defect in rapid or uncontrolled crystallization processes.
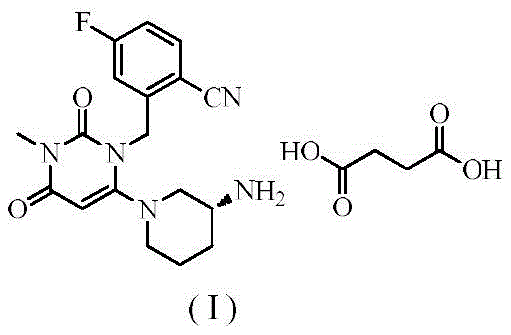
Furthermore, the structural integrity of the final product is maintained through careful temperature management during the drying phase, typically conducted at 50°C. This ensures that the crystal habit remains consistent, which is vital for downstream tableting and formulation processes. The ability to consistently produce high-purity [high-purity API intermediate] batches reduces the variability in bioavailability, a critical parameter for once-weekly dosing regimens like Zafatek. From a quality control perspective, this mechanism provides a robust defense against genotoxic impurities or residual starting materials that might persist in less refined batches. For technical teams, mastering these crystallization parameters—specifically the cooling rate and the solvent ratio—is the gateway to unlocking the full commercial potential of this refining technology.
How to Synthesize Trelagliptin Succinate Efficiently
Implementing this refining protocol requires strict adherence to the thermal and stoichiometric parameters outlined in the patent examples to ensure reproducibility. The process begins with the preparation of a crude Trelagliptin Succinate feedstock, which serves as the input for this purification cascade. Operators must ensure that the dissolution phase is complete before initiating the cooling cycle to prevent premature nucleation, which could trap impurities. The following guide outlines the standardized operational steps derived from the patent data, serving as a foundational reference for process engineers aiming to integrate this technology into their existing production lines. Detailed standard operating procedures (SOPs) should be developed based on these principles to maintain batch-to-batch consistency.
- Dissolve crude Trelagliptin Succinate in a mixture of acetonitrile and ethanol (1: 1 ratio) by heating to 50-60°C.
- Maintain stirring for 30 minutes to ensure complete homogeneity of the solution.
- Cool the solution gradually to a crystallization temperature between 0°C and 10°C (or down to -10°C) and stir for 2 hours.
- Filter the resulting crystals, wash with cold solvent mixture, and dry at 50°C to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this refining technology translates into tangible strategic benefits beyond mere chemical purity. The simplification of the purification process directly impacts the operational expenditure (OPEX) by reducing the complexity of unit operations required to bring the API to market readiness. By eliminating the need for hazardous or exotic solvents, facilities can lower their safety compliance costs and reduce the insurance premiums associated with handling dangerous chemicals. Moreover, the ability to recycle the mother liquor solvent creates a closed-loop system that significantly diminishes raw material consumption, offering substantial cost savings over the lifecycle of the product. This efficiency makes the supply chain more resilient against fluctuations in solvent pricing and availability, ensuring a steady flow of materials for continuous manufacturing campaigns.
- Cost Reduction in Manufacturing: The elimination of expensive chromatographic purification steps and the implementation of a recyclable solvent system drastically lower the variable costs associated with production. By optimizing the yield through controlled crystallization, manufacturers can maximize the output from each batch of crude material, effectively reducing the cost per kilogram of the final API. This economic efficiency is further bolstered by the reduced energy requirements for solvent recovery compared to distilling off large volumes of single-component solvents, leading to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: The robustness of this crystallization method ensures high batch success rates, minimizing the risk of production delays caused by failed purification runs. Since the process relies on readily available commodity solvents like acetonitrile and ethanol, the supply chain is less vulnerable to disruptions that might affect specialty reagents. This reliability is crucial for maintaining the continuity of supply for critical diabetes medications, allowing partners to meet their delivery commitments to downstream formulation companies without interruption.
- Scalability and Environmental Compliance: The straightforward nature of the heating and cooling cycles makes this process highly scalable from pilot plant quantities to multi-ton commercial production without significant re-engineering. Additionally, the use of lower toxicity solvents and the capability to recycle waste streams align perfectly with increasingly stringent global environmental regulations. This proactive approach to green chemistry not only mitigates regulatory risk but also enhances the corporate social responsibility (CSR) profile of the manufacturing entity, appealing to eco-conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this refining technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of adopting this process within your current manufacturing infrastructure.
Q: What purity level can be achieved with this refining method?
A: The patented refining method described in CN112480075A allows for the upgrading of crude Trelagliptin Succinate (initially 99.76% pure) to an ultra-high purity of 99.99% as detected by HPLC, meeting the most stringent pharmaceutical standards.
Q: Why is the acetonitrile-ethanol solvent system preferred?
A: This specific binary solvent system offers a balance of solubility and crystallization kinetics that effectively rejects impurities while allowing for the recycling of mother liquor, thereby reducing environmental impact and operational costs compared to more toxic alternatives.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It utilizes simple operations such as heating, cooling, and filtration without requiring complex chromatography or hazardous reagents, making it highly adaptable for commercial scale-up from pilot plants to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trelagliptin Succinate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and process reliability in the production of life-saving medications like Trelagliptin Succinate. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the 99.99% benchmark established by advanced refining protocols. We are committed to delivering excellence in every gram, supporting the global fight against diabetes with superior chemical solutions.
We invite you to collaborate with us to leverage this cutting-edge refining technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and accelerate your time to market.
