Scalable Manufacturing of SHR6390: A Novel 6-Step Synthetic Route for High-Purity Intermediates
Scalable Manufacturing of SHR6390: A Novel 6-Step Synthetic Route for High-Purity Intermediates
The rapidly evolving landscape of oncology therapeutics demands robust, scalable, and cost-effective synthetic routes for next-generation kinase inhibitors. Patent CN112778303A discloses a groundbreaking preparation method for SHR6390, a highly selective CDK4/6 kinase inhibitor currently in Phase 3 clinical trials for HR-positive, HER2-negative breast cancer. This technical insight report analyzes the proprietary 6-step synthesis, highlighting its strategic advantages for R&D directors seeking high-purity intermediates and procurement managers focused on supply chain optimization. The disclosed route effectively addresses previous limitations by utilizing mild reaction conditions and simplifying post-treatment protocols, thereby establishing a new benchmark for the commercial scale-up of complex pharmaceutical intermediates.
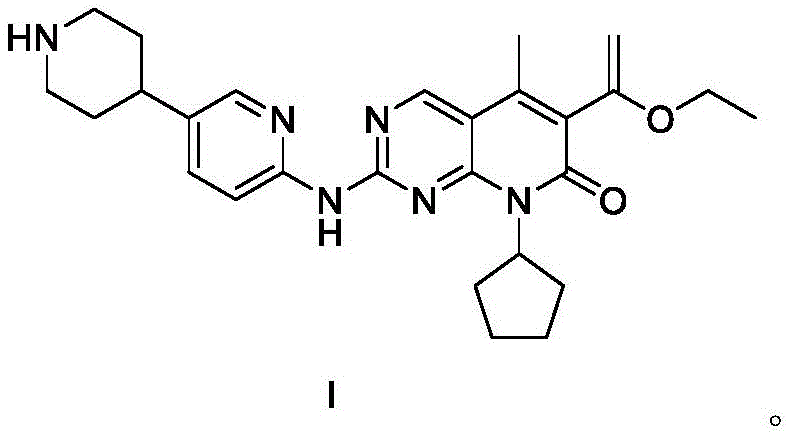
SHR6390 functions by preventing the phosphorylation of the Rb protein by the CDK4/6-Cyclin D complex, effectively arresting cell cycle progression from the G1 to S phase. As a critical asset in the fight against malignant tumors, the demand for reliable production methods is paramount. The invention provides a comprehensive pathway that not only ensures high chemical fidelity but also enhances process safety and environmental compliance. By leveraging the intrinsic reactivity of pyrimidine derivatives and optimizing palladium-catalyzed cross-couplings, this method delivers a white solid product with exceptional purity profiles, making it an ideal candidate for reliable pharmaceutical intermediate supplier partnerships aiming to secure long-term API availability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fused heterocyclic kinase inhibitors like SHR6390 has been plagued by harsh reaction conditions and cumbersome purification workflows. Traditional routes often require extreme temperatures or strong acidic/basic environments that can degrade sensitive functional groups, leading to complex impurity profiles that are difficult to separate. Furthermore, conventional methods frequently rely on multiple column chromatography steps for intermediate purification, which drastically increases solvent consumption, extends production lead times, and inflates the overall cost of goods sold (COGS). These inefficiencies create significant bottlenecks in cost reduction in pharmaceutical manufacturing, particularly when scaling from gram-scale laboratory synthesis to multi-kilogram commercial production. The accumulation of heavy metal residues from catalysts and the generation of hazardous waste streams further complicate regulatory compliance and environmental sustainability goals.
The Novel Approach
The methodology outlined in CN112778303A represents a paradigm shift towards greener and more efficient chemical manufacturing. A standout feature of this novel approach is the strategic exploitation of the high reaction activity at the 4-position of the pyrimidine ring in the initial step, allowing for the formation of intermediate 3 under mild room temperature conditions. Perhaps most critically for industrial application, the post-treatment for key intermediates, specifically compound 8, is remarkably simple; a good white solid product is obtained basically by filtration and washing, eliminating the need for energy-intensive purification columns. This streamlined workflow not only accelerates the synthesis timeline but also substantially reduces solvent usage and waste generation. By integrating robust catalytic cycles with simplified isolation techniques, this route offers a viable solution for reducing lead time for high-purity pharmaceutical intermediates while maintaining rigorous quality standards.
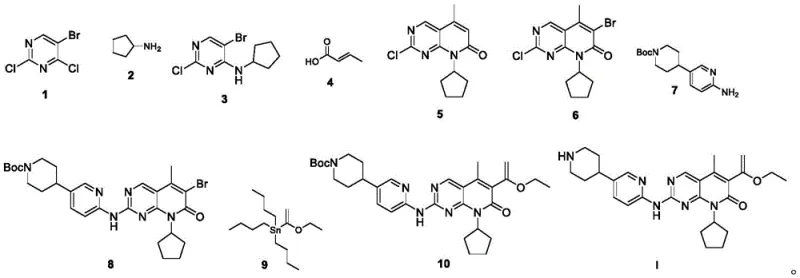
Mechanistic Insights into Pd-Catalyzed Cyclization and Stille Coupling
The core of this synthetic strategy relies on sophisticated transition metal catalysis to construct the rigid pyrido[2,3-d]pyrimidinone scaffold. In Step 2, the transformation of intermediate 3 to compound 5 involves a palladium-catalyzed reaction with crotonic acid (formula 4) in N-methylpyrrolidone (NMP). This step likely proceeds through a mechanism involving oxidative addition of the aryl halide to the Pd(0) species, followed by coordination and insertion of the alkene moiety from crotonic acid. The presence of triethylamine serves as a base to facilitate the elimination steps necessary for aromatization and ring closure. Operating at 70-80°C ensures sufficient kinetic energy for the catalytic cycle while avoiding thermal degradation, yielding compound 5 in impressive yields of up to 94.8%. This high efficiency underscores the robustness of the catalytic system, which is crucial for minimizing residual metal contaminants in the final API.
Further downstream, the installation of the acrylate side chain in Step 5 utilizes a Stille cross-coupling reaction between the brominated intermediate 8 and the vinyl tin reagent 9. Catalyzed by tetrakis(triphenylphosphine)palladium(0) in refluxing toluene (130°C), this reaction forms a carbon-carbon bond with high regioselectivity. The mechanism involves the transmetallation of the vinyl group from tin to palladium, followed by reductive elimination to release the coupled product 10. Although this step requires careful control of stoichiometry (preferably 1:2 molar ratio of substrate to tin reagent) to drive conversion, the subsequent purification via column chromatography (DCM:MeOH 50:1) effectively removes tin byproducts. The final deprotection in Step 6 uses 6M hydrochloric acid to cleave the Boc protecting group, a straightforward acid-base reaction that unveils the free piperidine amine, delivering the final active pharmaceutical ingredient with an HPLC purity of 99.5%.
How to Synthesize SHR6390 Efficiently
The synthesis of SHR6390 described in this patent is optimized for operational simplicity and high throughput, making it highly attractive for contract development and manufacturing organizations (CDMOs). The process begins with the nucleophilic substitution of 2,4-dichloro-5-bromopyrimidine with cyclopentylamine, followed by a cascade of cyclization, bromination, and coupling reactions. Each step has been fine-tuned to maximize yield and minimize impurity carryover. For instance, the bromination in Step 3 uses N-bromosuccinimide (NBS) in DMF at a controlled 50°C, ensuring selective monobromination without over-halogenation. The detailed standardized synthetic steps, including precise molar ratios, solvent volumes, and workup procedures, are critical for reproducibility. For a comprehensive guide on executing these reactions in a GMP environment, please refer to the technical protocol below.
- React 2,4-dichloro-5-bromopyrimidine with cyclopentylamine in dioxane at room temperature to form intermediate 3.
- Perform Pd-catalyzed cyclization with crotonic acid in NMP at 70-80°C to generate the pyrido[2,3-d]pyrimidinone core (compound 5).
- Brominate compound 5 using NBS in DMF at 50°C to yield intermediate 6.
- Couple intermediate 6 with the protected piperidine-pyridine amine (7) using LHMDS in THF to form intermediate 8.
- Execute Stille coupling with vinyl tin reagent 9 using Pd(PPh3)4 in toluene at reflux to install the acrylate moiety (compound 10).
- Deprotect the Boc group using 6M HCl in DCM to obtain the final API SHR6390 with >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this synthetic route are profound. The elimination of complex purification steps for early intermediates translates directly into lower operational expenditures and reduced dependency on expensive chromatographic resins and solvents. By simplifying the workflow, manufacturers can achieve faster batch turnover rates, enhancing the overall agility of the supply chain. This efficiency is vital for maintaining continuity of supply for critical oncology medications, where market demand is high and uninterrupted availability is non-negotiable. The use of commercially available starting materials and standard reagents further mitigates supply risk, ensuring that production schedules remain resilient against raw material shortages.
- Cost Reduction in Manufacturing: The most significant cost driver in fine chemical synthesis is often the downstream processing. By enabling the isolation of intermediate 8 through simple filtration and washing without the need for column chromatography, this process drastically cuts down on solvent consumption and labor hours. The high yield in the cyclization step (up to 94.8%) minimizes raw material waste, ensuring that expensive precursors are converted efficiently into value-added products. Additionally, the avoidance of exotic reagents in favor of standard catalysts like PdCl2 and Pd(PPh3)4 helps stabilize reagent costs, contributing to substantial cost savings in pharmaceutical manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions—such as performing the initial amination at room temperature and the bromination at moderate heat (50°C)—reduces the risk of batch failures due to thermal runaway or equipment limitations. This reliability allows for more accurate forecasting and planning. Furthermore, the synthesis utilizes widely accessible solvents like dioxane, NMP, DMF, and toluene, which are staples in the global chemical supply chain. This accessibility ensures that production is not bottlenecked by the scarcity of specialized reagents, thereby securing a stable supply of high-purity pharmaceutical intermediates for downstream API production.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies waste generation, but this route is designed with green chemistry principles in mind. The high atom economy of the coupling reactions and the reduced solvent load from simplified workups lower the environmental footprint of the manufacturing process. The ability to produce a white solid product with high purity directly from filtration reduces the volume of liquid waste requiring treatment. This alignment with environmental compliance standards not only reduces disposal costs but also positions the manufacturing process favorably within increasingly strict regulatory frameworks regarding industrial emissions and waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of SHR6390. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on process capabilities and quality outcomes. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this route for commercial production.
Q: What are the key advantages of this new SHR6390 synthesis method?
A: The patented method (CN112778303A) offers mild reaction conditions, particularly in the first step which utilizes the high reactivity of the pyrimidine 4-position. Crucially, intermediates 3 and 8 can be obtained via simple filtration and washing without column chromatography, significantly simplifying post-treatment and reducing production costs.
Q: What is the overall purity achievable with this process?
A: The final deprotection step yields SHR6390 (Formula I) with an HPLC purity of up to 99.5%, meeting stringent specifications required for clinical-grade pharmaceutical intermediates.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process is designed for industrial scale-up. Key steps avoid complex purification techniques like preparative HPLC for early intermediates, and utilize robust reagents like NBS and standard Pd catalysts, ensuring supply chain reliability and cost reduction in pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable SHR6390 Supplier
The synthesis of CDK4/6 inhibitors like SHR6390 represents a pinnacle of modern medicinal chemistry, requiring a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions and sensitive heterocyclic chemistry, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to process optimization, we guarantee the delivery of high-quality intermediates that align perfectly with the demanding timelines of global drug development programs.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthetic technology. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can enhance your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us be your trusted partner in bringing life-saving oncology therapies to market faster and more efficiently.
