Optimized Commercial Synthesis of SHR6390: Technical Breakthroughs for Scalable API Production
The development of efficient synthetic routes for next-generation oncology therapeutics remains a critical priority for the global pharmaceutical industry. Patent CN112778303A discloses a robust preparation method for SHR6390, a potent and selective small molecule CDK4/6 kinase inhibitor currently advancing through phase 3 clinical research for HR-positive, HER2-negative breast cancer. This technology addresses significant bottlenecks in traditional synthesis by leveraging the high reactivity of the pyrimidine 4-position under mild conditions, enabling a streamlined 6-step chemical sequence. The process is specifically engineered to minimize operational complexity while maximizing yield, offering a viable pathway for the commercial scale-up of complex kinase inhibitors. By integrating advanced catalytic strategies with simplified workup procedures, this method ensures consistent quality and supply continuity for high-value active pharmaceutical ingredients.
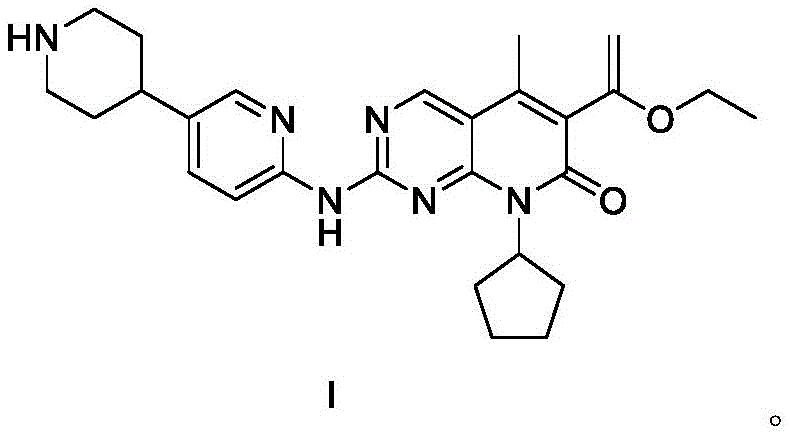
The structural complexity of SHR6390 necessitates a precise assembly strategy to maintain stereochemical integrity and functional group tolerance throughout the synthesis. The disclosed methodology capitalizes on convergent synthesis principles, where distinct molecular fragments are coupled efficiently to construct the final pyrido[2,3-d]pyrimidin-7-one core. This approach not only enhances the overall atom economy but also reduces the accumulation of impurities that often plague multi-step linear syntheses. For procurement and supply chain stakeholders, understanding the underlying chemical architecture is essential for evaluating raw material sourcing risks and production scalability. The ability to produce this high-purity CDK4/6 inhibitor reliably positions manufacturers to meet the rigorous demands of international regulatory bodies and clinical trial timelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing fused heterocyclic systems like SHR6390 often rely on harsh reaction conditions that compromise safety and operational efficiency. Conventional methods frequently require elevated temperatures, strong acidic or basic environments, and excessive use of protecting groups, which inevitably increase the number of unit operations and waste generation. Furthermore, legacy processes often depend heavily on repetitive column chromatography for intermediate purification, creating significant bottlenecks in production throughput and driving up solvent consumption costs. These inefficiencies result in prolonged lead times and inconsistent batch-to-batch quality, posing substantial risks for reliable pharmaceutical intermediate supplier networks aiming to support large-scale API manufacturing. The accumulation of trace metal catalysts and organic impurities from these aggressive conditions also complicates downstream processing and final drug substance release testing.
The Novel Approach
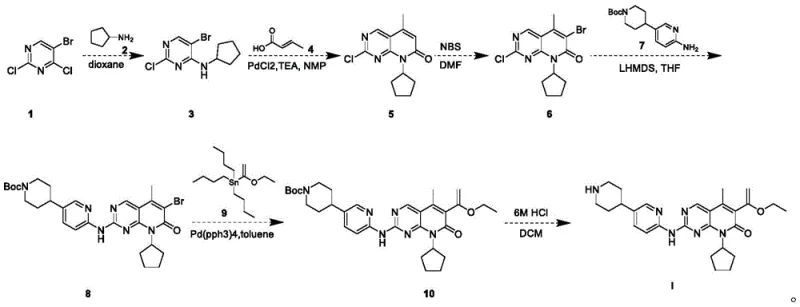
In stark contrast, the novel approach detailed in the patent utilizes a series of optimized transformations that prioritize mild conditions and operational simplicity. The synthesis initiates with a nucleophilic substitution at room temperature, eliminating the need for energy-intensive heating during the initial bond formation. Subsequent steps employ palladium-catalyzed cross-coupling reactions under controlled nitrogen atmospheres to ensure high selectivity and yield, such as the conversion of Compound 3 to Compound 5 with yields reaching 94.8%. A standout feature of this route is the post-treatment strategy for Compound 8, where the target product precipitates as a white solid upon quenching, allowing for isolation via simple filtration and washing without further purification. This drastic reduction in purification steps significantly lowers cost reduction in API manufacturing by minimizing solvent usage and processing time while maintaining high chemical fidelity.
Mechanistic Insights into Pd-Catalyzed Coupling and Cyclization
The core of this synthetic strategy relies on precise mechanistic control during the construction of the central heterocyclic scaffold. The transformation of Compound 1 to Compound 3 exploits the inherent electrophilicity of the 4-chloro position on the pyrimidine ring, facilitating a smooth displacement by cyclopentanamine in dioxane at room temperature. This step is critical as it sets the stage for subsequent cyclization, achieving a crude yield of 100% which indicates near-perfect conversion efficiency. Later stages involve a Heck-type or Suzuki-Miyaura coupling sequence using palladium chloride and tetrakis(triphenylphosphine)palladium, respectively, to install the vinyl and aryl components. The use of ligands and specific bases like triethylamine ensures that the catalytic cycle turns over rapidly, preventing catalyst deactivation and minimizing the formation of homocoupling byproducts that could affect the impurity profile of the final high-purity pharmaceutical intermediates.
Impurity control is meticulously managed through the physical properties of the intermediates, particularly during the formation of Compound 8. The reaction involves the deprotonation of Compound 6 using Lithium Hexamethyldisilazide (LHMDS) at low temperatures (0-10°C) to generate a reactive nucleophile that attacks Compound 7. The resulting product precipitates out of the tetrahydrofuran solution upon quenching with saturated ammonium chloride and acetic acid, effectively excluding soluble impurities from the crystal lattice. This crystallization-driven purification is a powerful tool for reducing lead time for high-purity pharmaceutical intermediates as it bypasses the need for time-consuming chromatographic separation. The final deprotection step using 6M hydrochloric acid cleanly removes the Boc protecting group, yielding the target molecule with an HPLC purity of 99.5%, demonstrating the robustness of the impurity rejection mechanism throughout the entire synthetic sequence.
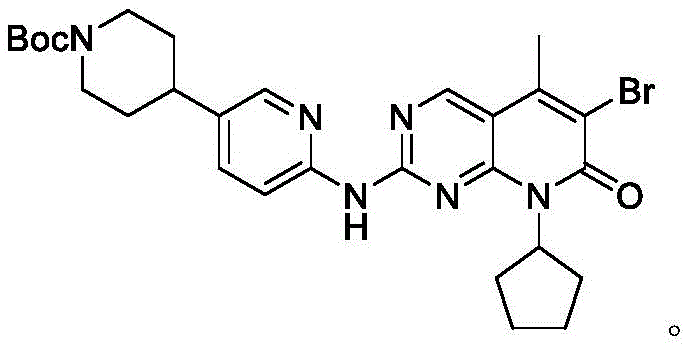
How to Synthesize SHR6390 Efficiently
Implementing this synthesis requires strict adherence to the specified reaction parameters to ensure optimal yield and safety profiles across all six steps. The process begins with the room temperature coupling of halogenated pyrimidines, followed by palladium-catalyzed cyclization and bromination sequences that build the core rigidity of the molecule. Operators must maintain inert atmospheres during metal-catalyzed steps to prevent oxidation of sensitive reagents like phosphine ligands and organometallic species. The detailed standardized synthesis steps见下方的指南 outline the precise molar ratios, solvent volumes, and workup procedures necessary to replicate the patent's success in a GMP environment.
- Initiate pyrimidine substitution at room temperature to form Compound 3 with near-quantitative crude yield.
- Execute Pd-catalyzed coupling and bromination sequences to construct the core scaffold efficiently.
- Finalize synthesis via LHMDS-mediated coupling and acid deprotection to achieve 99.5% HPLC purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers transformative benefits for procurement managers and supply chain heads focused on cost efficiency and reliability. The elimination of complex purification steps for key intermediates directly translates to substantial cost savings by reducing solvent procurement, waste disposal fees, and equipment occupancy time. The reliance on commercially available starting materials and common reagents such as N-bromosuccinimide and LHMDS mitigates supply chain risks associated with exotic or custom-synthesized building blocks. Furthermore, the high yields observed in critical steps, such as the 94.8% yield in the cyclization step, ensure that raw material utilization is maximized, thereby lowering the overall cost of goods sold. This efficiency makes the process highly attractive for cost reduction in API manufacturing where margin pressure is a constant challenge for generic and branded drug producers alike.
- Cost Reduction in Manufacturing: The process design inherently lowers production costs by minimizing the number of unit operations required for purification. By achieving high crude yields and utilizing precipitation-based isolation for intermediates like Compound 8, the method drastically reduces the volume of organic solvents needed for chromatography. This reduction in solvent consumption not only lowers direct material costs but also decreases the environmental burden associated with solvent recovery and waste treatment. Additionally, the use of mild reaction conditions reduces energy consumption for heating and cooling, contributing to a leaner and more economically viable manufacturing model that supports competitive pricing strategies.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain stability by reducing the sensitivity to minor variations in reaction parameters. The use of stable, commercially sourced reagents ensures that production schedules are not disrupted by raw material shortages or long lead times for custom chemicals. The simplified workup procedures also mean that batch cycles are shorter, allowing for increased production capacity and faster response to market demand fluctuations. This reliability is crucial for maintaining the continuity of supply for critical oncology medications, ensuring that patients have uninterrupted access to life-saving therapies without the risk of stockouts due to manufacturing delays.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of hazardous reagents and the simplicity of the isolation techniques. The ability to isolate products via filtration rather than chromatography is a significant advantage for large-scale operations, as it avoids the technical challenges and safety risks associated with packing and running large columns. Moreover, the reduced solvent waste aligns with modern green chemistry principles and environmental regulations, making it easier to obtain necessary permits and maintain compliance with local and international environmental standards. This scalability ensures that the method can support the commercial scale-up of complex kinase inhibitors from kilogram to tonne quantities seamlessly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on process feasibility and quality outcomes. Understanding these details is vital for technical teams evaluating the transfer of this technology to their own manufacturing facilities or for procurement teams assessing the capability of potential partners.
Q: What are the key advantages of this SHR6390 synthesis route?
A: The method features mild reaction conditions, such as room temperature steps, and simplifies post-treatment by allowing direct filtration for key intermediates without column chromatography.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the protocol avoids complex purification in early stages and utilizes common reagents, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Q: What purity levels can be achieved with this method?
A: The final deprotection step yields SHR6390 with an HPLC purity of 99.5%, meeting stringent specifications required for high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable SHR6390 Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate complex synthetic routes like the one described in CN112778303A into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every batch. Our commitment to excellence ensures that the SHR6390 we produce meets the highest standards required for clinical and commercial applications, providing you with a secure and dependable source for this critical oncology intermediate.
We invite you to collaborate with us to explore how this optimized synthesis can benefit your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-quality solutions efficiently. Partnering with us ensures access to top-tier chemical manufacturing services that drive innovation and value in your supply chain.
