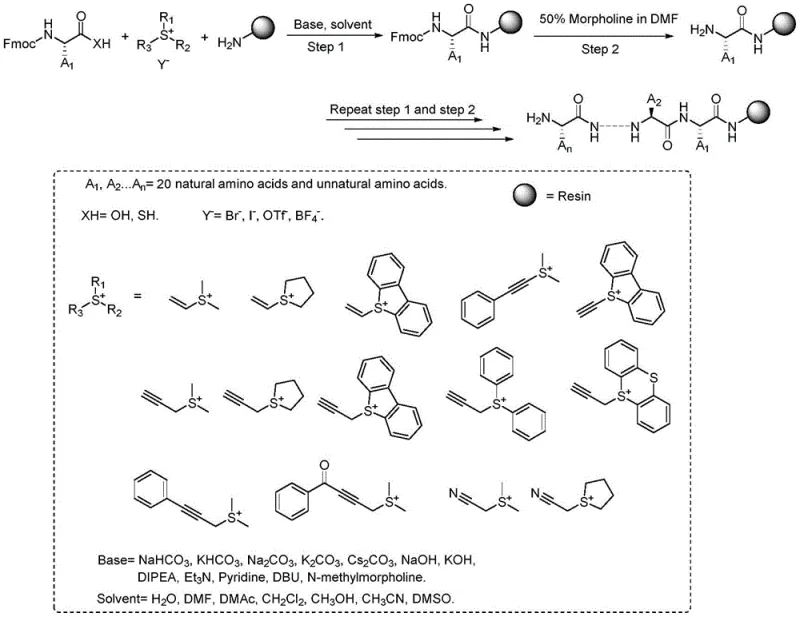
Advanced Solid-Phase Polypeptide Synthesis via Unsaturated Sulfonium Salts for Commercial Manufacturing
Advanced Solid-Phase Polypeptide Synthesis via Unsaturated Sulfonium Salts for Commercial Manufacturing
The landscape of polypeptide manufacturing is undergoing a significant transformation driven by the urgent need for safer, more cost-effective, and environmentally sustainable synthetic methodologies. As detailed in the groundbreaking patent CN111978369A, filed in late 2020, a novel approach has emerged that challenges the decades-old dominance of traditional coupling agents like HATU and HCTU. This technology introduces the use of unsaturated sulfonium salts as highly efficient carboxyl activators for solid-phase peptide synthesis (SPPS). By replacing hazardous phosphonium and uranium-based reagents with stable, low-toxicity sulfonium derivatives, this method addresses critical pain points in both laboratory research and industrial production. For R&D directors and procurement managers alike, this represents a paradigm shift towards a more robust supply chain for reliable polypeptide intermediate suppliers, offering a pathway to reduce dependency on expensive and dangerous condensing agents while maintaining high coupling efficiencies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional solid-phase polypeptide synthesis has long relied on in-situ activation methods using reagents such as HATU, HCTU, PyBOP, and DIC. While these methods are standard, they suffer from severe inherent drawbacks that impact both safety and cost structures in a commercial setting. HATU and HCTU, for instance, are known to be highly sensitive to moisture, requiring strictly anhydrous conditions that increase solvent consumption and operational complexity. Furthermore, these reagents pose significant safety hazards; they are often explosive and exhibit high toxicity, necessitating specialized handling protocols and expensive waste disposal procedures. From a supply chain perspective, the reliance on these complex, multi-step synthesized coupling agents creates vulnerability to price volatility and availability issues. Additionally, the formation of racemic byproducts and the difficulty in removing certain activating residues can compromise the purity profile of the final high-purity polypeptide, leading to costly downstream purification challenges.
The Novel Approach
The innovative methodology disclosed in the patent circumvents these issues by employing α,β- and β,γ-unsaturated substituted sulfonium salts as the primary carboxyl activating reagents. This approach fundamentally alters the reaction dynamics by facilitating an amine transesterification mechanism that is remarkably tolerant to water and operates efficiently under mild basic conditions. Unlike the explosive nature of traditional uranium salts, these sulfonium activators are safe, stable, and low-toxicity, making them ideal for commercial scale-up of complex peptides. The process involves reacting Fmoc-protected amino acids with the sulfonium salt in the presence of a base, followed by coupling to the resin-bound amine. This not only simplifies the reaction setup by removing the need for rigorous drying of solvents but also enhances the overall safety profile of the manufacturing facility. The result is a streamlined workflow that maintains high yields comparable to conventional methods while drastically reducing the environmental and safety footprint.
Mechanistic Insights into Sulfonium Salt-Mediated Amidation
The core of this technological advancement lies in the unique reactivity of the unsaturated sulfonium salt. In the presence of a base, the sulfonium salt activates the carboxyl group of the incoming Fmoc-amino acid, generating a highly reactive intermediate species. This activated complex then undergoes a nucleophilic attack by the free amino group on the solid support (resin), resulting in the formation of the peptide bond and the release of a neutral sulfide byproduct. This mechanism is distinct from the O-acylisourea or active ester pathways typical of carbodiimide or uranium salt chemistry. The tolerance of this system to aqueous environments is particularly noteworthy; the reaction can proceed effectively in solvents containing water or even in purely aqueous conditions with specific inorganic bases. This water tolerance suggests a mechanism where the activated intermediate is sufficiently stable to outcompete hydrolysis, or where the kinetics of amidation are significantly faster than hydrolysis, ensuring high conversion rates even in less-than-perfect anhydrous conditions.
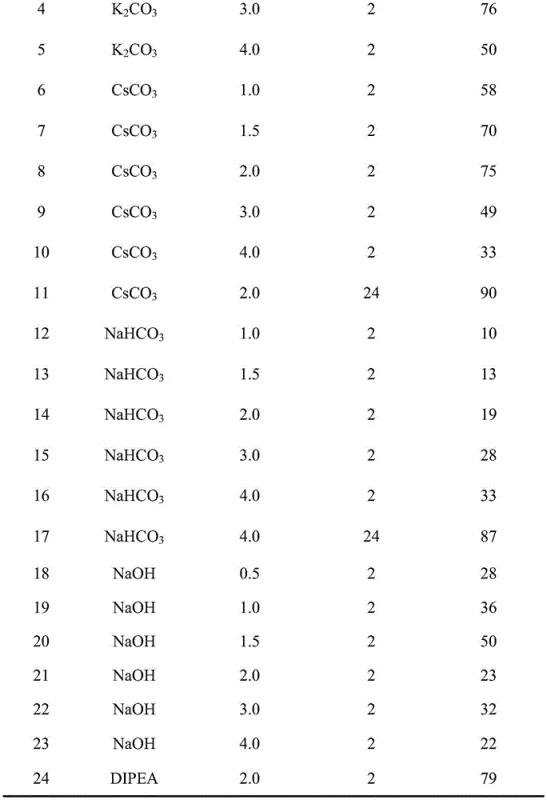
Furthermore, the versatility of the base selection allows for fine-tuning the reaction kinetics to minimize side reactions such as racemization. The patent data indicates that a wide array of bases, ranging from mild inorganic carbonates like sodium bicarbonate and cesium carbonate to stronger organic amines like DIPEA and DBU, can be successfully employed. This flexibility is crucial for optimizing the synthesis of difficult sequences where steric hindrance or aggregation might otherwise impede coupling. The ability to use inexpensive inorganic bases like potassium carbonate in aqueous media opens up new possibilities for green chemistry applications in peptide manufacturing, potentially reducing lead time for high-purity polypeptides by simplifying the workup and purification stages. The structural diversity of the sulfonium salts themselves, including vinyl, ethynyl, and propynyl derivatives, offers additional levers for chemists to optimize reactivity for specific amino acid substrates.
How to Synthesize Polypeptides Efficiently
Implementing this novel synthesis route requires a strategic adjustment to standard SPPS protocols, focusing on the preparation of the sulfonium activator and the optimization of base equivalents. The process begins with the swelling of the resin, typically Rink amide MBHA resin, followed by the standard removal of the Fmoc protecting group using a morpholine/DMF solution. The critical innovation occurs in the coupling step, where the Fmoc-amino acid is mixed with the unsaturated sulfonium salt and the chosen base before being added to the resin. This pre-activation or in-situ activation strategy ensures that the reactive species is generated immediately prior to coupling, maximizing efficiency. While the specific stoichiometry may vary depending on the steric bulk of the amino acid, the patent suggests using equivalents ranging from 1.2 to 5 times the resin loading to drive the reaction to completion. For a detailed breakdown of the specific experimental conditions and step-by-step operational parameters, please refer to the standardized guide below.
- Activate the Fmoc-protected amino acid carboxyl group using an unsaturated sulfonium salt in the presence of a base.
- Perform the amine transesterification reaction with the resin-bound amine to form the peptide bond.
- Remove the Fmoc protecting group using a morpholine/DMF solution and repeat the cycle for chain elongation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of sulfonium salt-mediated peptide synthesis offers tangible economic and logistical benefits that extend far beyond the laboratory bench. The primary advantage lies in the drastic simplification of the reagent supply chain. Traditional coupling agents like HATU are complex molecules that require multi-step synthesis involving hazardous precursors, leading to high costs and potential supply bottlenecks. In contrast, the unsaturated sulfonium salts described in this patent are structurally simpler and can be synthesized from readily available starting materials, suggesting a significantly lower cost of goods sold (COGS) for the activating reagent. Moreover, the stability of these salts eliminates the need for cold chain logistics or specialized inert atmosphere storage, further reducing warehousing and transportation expenses. This stability also translates to a longer shelf life, minimizing waste due to reagent degradation.
- Cost Reduction in Manufacturing: The elimination of expensive uranium and phosphorus-based condensing agents represents a direct material cost saving. Additionally, the tolerance of the reaction to water allows for the use of cheaper, greener solvent systems or reduced solvent drying requirements, which lowers utility costs associated with solvent recovery and drying. The simplified workup procedure, owing to the neutral nature of the sulfide byproducts, reduces the burden on purification teams, potentially increasing overall throughput and reducing the cost per gram of the final API intermediate.
- Enhanced Supply Chain Reliability: By diversifying the chemical toolbox away from a narrow set of proprietary coupling agents, manufacturers can mitigate the risk of supply disruptions. The raw materials for sulfonium salts are commodity chemicals, ensuring a stable and continuous supply. Furthermore, the reduced hazard profile of the reagents simplifies regulatory compliance and shipping classifications, accelerating the movement of materials across international borders and ensuring consistent production schedules without safety-related stoppages.
- Scalability and Environmental Compliance: The safety profile of sulfonium salts makes them inherently more scalable than explosive alternatives. Large-scale reactors do not require the same level of explosion-proof engineering controls, reducing capital expenditure for plant upgrades. Environmentally, the potential to run reactions in aqueous or semi-aqueous media aligns with green chemistry principles, reducing the volume of organic waste generated. This not only lowers waste disposal costs but also enhances the company's sustainability metrics, a key factor for modern pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfonium salt technology in peptide manufacturing. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a factual basis for evaluating the feasibility of this method for your specific projects. Understanding these nuances is essential for R&D teams planning to transition from conventional coupling strategies to this novel, safer alternative.
Q: What are the advantages of using sulfonium salts over HATU in peptide synthesis?
A: Unlike HATU, which is toxic, explosive, and highly sensitive to moisture, unsaturated sulfonium salts are safe, low-toxicity reagents that are stable and effective even in the presence of water, significantly reducing safety risks and handling costs.
Q: Can this method be scaled for industrial production?
A: Yes, the method is specifically designed for industrial applicability. It utilizes stable reagents and tolerates aqueous conditions, simplifying the purification process and making it suitable for large-scale commercial manufacturing of complex polypeptides.
Q: What types of bases are compatible with this synthesis method?
A: The protocol is highly versatile regarding base selection, supporting a wide range of inorganic bases like potassium carbonate and cesium carbonate, as well as organic bases such as DIPEA and DBU, allowing for optimization based on specific substrate solubility and reactivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polypeptide Supplier
At NINGBO INNO PHARMCHEM, we are at the forefront of adopting cutting-edge synthetic methodologies to deliver superior value to our global partners. Our technical team has extensively evaluated the sulfonium salt-mediated synthesis route and confirmed its potential for producing complex polypeptide sequences with exceptional purity and efficiency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of polypeptide intermediate meets the highest international standards required for pharmaceutical applications.
We invite you to explore how this innovative technology can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific peptide targets. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced capabilities can support your drug development pipeline and accelerate your time to market.
