Advanced Palladium-Catalyzed Synthesis of 3-Alkenyl Quinolin-2(1H)ones for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocyclic scaffolds, particularly quinolin-2(1H)-one derivatives, which serve as critical building blocks for a vast array of bioactive compounds. As detailed in the groundbreaking patent CN114478375A, a novel preparation method has been disclosed that fundamentally shifts the paradigm for synthesizing 3-alkenyl quinolin-2(1H) ketone derivatives. This technology leverages a sophisticated palladium-catalyzed reductive aminocarbonylation strategy, utilizing o-nitrobenzaldehyde as a dual nitrogen and formyl source, thereby streamlining the synthetic route significantly. For R&D Directors and Procurement Managers alike, this innovation represents a pivotal opportunity to access high-purity pharmaceutical intermediates through a process that is not only chemically elegant but also commercially viable for large-scale production. The ability to synthesize these complex heterocycles from readily available starting materials addresses long-standing challenges in supply chain continuity and cost efficiency within the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolinone derivatives has relied heavily on transition metal-catalyzed carbonylation reactions involving aryl or vinyl halides, which often necessitate the use of hazardous and difficult-to-handle gaseous carbon monoxide under high pressure. Furthermore, conventional approaches frequently employ allyl chlorides, acetates, or carbonates as electrophiles, which can present significant stability issues and toxicity concerns during storage and handling on an industrial scale. These traditional electrophiles often require stringent anhydrous conditions and specialized equipment to prevent decomposition, leading to increased operational expenditures and complex waste management protocols. Additionally, the limited substrate scope of many classical methods restricts the ability to introduce diverse functional groups late in the synthesis, forcing chemists to adopt lengthy protecting group strategies that erode overall yield and atom economy. The reliance on high-pressure gas infrastructure also poses substantial safety risks, creating bottlenecks for facilities that lack the necessary certifications for handling toxic gases, thereby limiting the pool of potential contract manufacturing partners.
The Novel Approach
In stark contrast to these legacy techniques, the methodology outlined in patent CN114478375A introduces a transformative approach by utilizing allyl aryl ethers as the electrophilic coupling partners, which are inherently more stable, less toxic, and operationally simpler to manage in a production environment. This novel route employs molybdenum hexacarbonyl as a solid carbon monoxide surrogate, effectively eliminating the need for high-pressure CO gas cylinders and allowing the reaction to proceed in standard sealed vessels at moderate temperatures. By using o-nitrobenzaldehyde as both the nitrogen source and the formyl source, the process achieves a remarkable level of atom economy, reducing the number of discrete synthetic steps required to reach the target 3-alkenyl quinolin-2(1H)one core. This simplification of the synthetic pathway not only accelerates the timeline from laboratory discovery to commercial manufacturing but also drastically reduces the generation of chemical waste, aligning with modern green chemistry principles that are increasingly demanded by global regulatory bodies and end-user pharmaceutical clients.
Mechanistic Insights into Pd-Catalyzed Reductive Aminocarbonylation
The core of this technological advancement lies in the intricate palladium-catalyzed cycle that facilitates the coupling of o-nitrobenzaldehyde with allyl aryl ethers under mild conditions. The reaction initiates with the oxidative addition of the palladium catalyst into the allyl ether bond, a step that is traditionally challenging due to the poor leaving group ability of the alkoxide moiety compared to halides. However, the presence of tetrabutylammonium iodide (TBAI) as a crucial additive likely assists in activating the allyl ether through halide exchange or by stabilizing the cationic palladium intermediates, thereby lowering the activation energy for this key step. Following oxidative addition, the insertion of carbon monoxide, released in situ from the thermal decomposition of molybdenum hexacarbonyl, generates an acyl-palladium species that is poised for nucleophilic attack.
Subsequently, the nitro group of the o-nitrobenzaldehyde undergoes a reductive transformation, potentially mediated by the molybdenum species or the phosphine ligand system, to generate the requisite amine functionality in situ. This intramolecular cyclization then occurs, forging the new carbon-nitrogen bond that closes the quinolinone ring system with high regioselectivity. The use of tris(3-methoxyphenyl)phosphine as a ligand provides the optimal electronic and steric environment to stabilize the active palladium species throughout the catalytic cycle, preventing premature catalyst deactivation via palladium black formation. 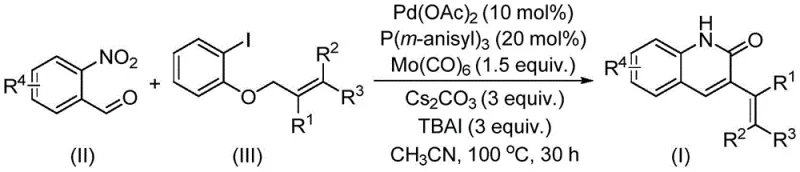
Furthermore, the choice of cesium carbonate as the base ensures efficient neutralization of acidic byproducts without promoting unwanted side reactions such as hydrolysis of the sensitive allyl ether or the newly formed lactam ring. The reaction proceeds efficiently in acetonitrile, a solvent that offers excellent solubility for both the organic substrates and the inorganic bases, ensuring a homogeneous reaction mixture that facilitates heat and mass transfer. This mechanistic understanding underscores the robustness of the process, demonstrating wide functional group tolerance that allows for the incorporation of halogens, methoxy groups, and various alkyl chains without compromising the integrity of the catalytic cycle or the purity of the final product.
How to Synthesize 3-Alkenyl Quinolin-2(1H)one Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and thermal profiles as established in the patent data to maximize yield and minimize impurity formation. The process begins with the careful weighing and charging of palladium acetate, the specialized phosphine ligand, molybdenum hexacarbonyl, cesium carbonate, and the iodide additive into a suitable reaction vessel equipped with efficient stirring capabilities.
- Combine palladium acetate, tris(3-methoxyphenyl)phosphine, molybdenum carbonyl, cesium carbonate, tetrabutylammonium iodide, o-nitrobenzaldehyde, and allyl aryl ether in a reaction vessel.
- Heat the reaction mixture to a temperature range of 90°C to 110°C, preferably 100°C, and maintain stirring for approximately 30 hours to ensure complete conversion.
- Upon completion, perform post-treatment including filtration and silica gel mixing, followed by column chromatography purification to isolate the high-purity 3-alkenyl quinolin-2(1H)one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented methodology offers profound advantages that directly translate into cost reduction in fine chemical manufacturing and enhanced supply chain resilience for critical pharmaceutical intermediates. The substitution of hazardous gaseous carbon monoxide with a solid carbonyl source like molybdenum hexacarbonyl removes the requirement for specialized high-pressure reactor infrastructure, allowing production to be scaled in standard glass-lined or stainless steel vessels available at most contract development and manufacturing organizations. This flexibility significantly broadens the supplier base, reducing dependency on single-source vendors who possess niche high-pressure capabilities and thereby mitigating supply chain risks associated with geopolitical instability or facility downtime. Moreover, the use of allyl aryl ethers, which are often more commercially abundant and cheaper than their halide or carbonate counterparts, drives down the raw material cost basis, enabling more competitive pricing models for the final active pharmaceutical ingredients derived from these intermediates.
- Cost Reduction in Manufacturing: The elimination of high-pressure gas handling systems results in substantial capital expenditure savings, as facilities do not need to invest in costly safety monitoring equipment or specialized containment units for toxic gases. Additionally, the simplified workup procedure, which involves straightforward filtration and chromatography, reduces the consumption of solvents and silica gel compared to multi-step traditional routes, leading to lower operational costs per kilogram of product. The high reaction efficiency and yield reported in the patent imply less raw material waste and higher throughput, which further amplifies the economic benefits when translating this chemistry from gram-scale laboratory experiments to multi-ton commercial production campaigns.
- Enhanced Supply Chain Reliability: By utilizing starting materials such as o-nitrobenzaldehyde and allyl aryl ethers that are widely available from global chemical suppliers, manufacturers can secure a stable and continuous flow of inputs without facing the bottlenecks often associated with custom-synthesized specialty reagents. The robustness of the reaction conditions, which tolerate a wide range of functional groups, means that variations in raw material quality from different suppliers are less likely to cause batch failures, ensuring consistent delivery schedules to downstream pharmaceutical clients. This reliability is crucial for maintaining just-in-time inventory levels and avoiding costly production delays that can ripple through the entire drug development and commercialization timeline.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction parameters that can be easily adjusted for larger batch sizes without encountering the heat transfer limitations typical of exothermic high-pressure reactions. From an environmental standpoint, the avoidance of volatile organic halides and toxic gases aligns with increasingly stringent global environmental regulations, reducing the burden of waste treatment and emissions reporting. This green chemistry profile not only minimizes the environmental footprint of the manufacturing process but also enhances the marketability of the final product to eco-conscious pharmaceutical companies seeking to reduce the carbon footprint of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its adoption for their specific project needs.
Q: Why is the use of allyl aryl ethers considered advantageous over traditional allyl halides in this synthesis?
A: Allyl aryl ethers are generally more stable, less toxic, and easier to handle operationally compared to allyl chlorides or carbonates. This patent demonstrates a breakthrough in activating these challenging electrophiles for carbonylation, expanding the substrate scope while improving safety profiles in manufacturing.
Q: How does the use of Molybdenum Hexacarbonyl impact the safety and cost of the process?
A: Molybdenum hexacarbonyl serves as a solid carbon monoxide source, eliminating the need for handling high-pressure CO gas cylinders. This significantly reduces infrastructure costs related to high-pressure reactors and enhances overall plant safety by mitigating the risks associated with toxic gas leaks.
Q: What is the functional group tolerance of this palladium-catalyzed system?
A: The method exhibits wide substrate functional group tolerance, accommodating various substituents such as halogens, methoxy groups, and trifluoromethyl groups on the aromatic rings. This versatility allows for the synthesis of diverse derivatives without requiring extensive protecting group strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkenyl Quinolin-2(1H)one Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative academic patents like CN114478375A into reliable, commercial-grade supply chains for our global partners. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity 3-alkenyl quinolin-2(1H)one derivatives that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project requirements and volume needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this methodology for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your pharmaceutical development programs.
