Advanced Palladium-Catalyzed Synthesis of 3-Alkenyl Quinolin-2(1H)one Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 3-Alkenyl Quinolin-2(1H)one Derivatives for Commercial Scale-Up
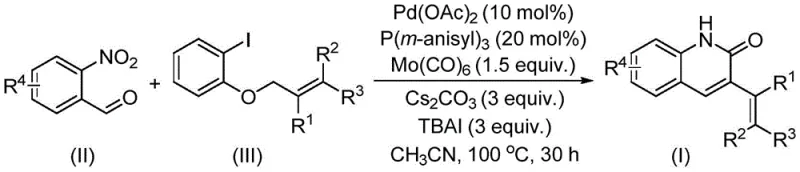
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for nitrogen-containing heterocycles, particularly quinolin-2(1H)-one derivatives, due to their profound biological activities ranging from antibiotic to antitumor properties. A significant breakthrough in this domain is documented in patent CN114478375A, which discloses a highly efficient preparation method for 3-alkenyl quinolin-2(1H) ketone derivatives. This innovative approach utilizes a palladium-catalyzed reductive aminocarbonylation strategy, reacting o-nitrobenzaldehyde with allyl aryl ethers under optimized conditions. For R&D directors and procurement managers seeking reliable pharmaceutical intermediate suppliers, this technology represents a pivotal shift towards more economical and operationally simple manufacturing processes. The method not only streamlines the synthetic pathway but also leverages readily available starting materials, thereby addressing critical supply chain vulnerabilities often associated with complex heterocyclic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of quinolin-2(1H)-one scaffolds has relied on methodologies that often involve harsh reaction conditions, multi-step sequences, or the use of expensive and toxic reagents. Conventional transition metal-catalyzed carbonylation reactions have extensively utilized aryl and vinyl halides; however, the application of allyl compounds, particularly allyl ethers, has remained a challenging and underexplored area. Most existing carbonylation protocols depend on allyl chlorides, acetates, carbonates, or phosphates as electrophiles, which can introduce additional waste streams and purification burdens. Furthermore, many traditional routes fail to efficiently incorporate the nitrogen atom and the carbonyl group in a single convergent step, leading to lower overall atom economy and increased production costs. These limitations pose significant hurdles for the commercial scale-up of complex pharmaceutical intermediates, necessitating the development of more direct and sustainable catalytic systems.
The Novel Approach
The methodology described in patent CN114478375A overcomes these historical barriers by introducing a novel palladium-catalyzed reductive aminocarbonylation of o-nitrobenzaldehyde and allyl aryl ethers. This reaction is groundbreaking as it represents the first instance where allyl aryl ethers are successfully employed as electrophiles in the aminocarbonylation synthesis of 3-alkenyl quinolin-2(1H)one derivatives. By utilizing o-nitrobenzaldehyde as a dual-purpose reagent serving as both the nitrogen source and the formyl source, the process drastically simplifies the reaction setup. The use of allyl aryl ethers, which are characterized by their low toxicity and ease of handling compared to other allyl derivatives, further enhances the operational safety and environmental profile of the synthesis. This novel approach offers a direct, one-pot solution that significantly reduces the number of unit operations required, making it an attractive option for cost reduction in API manufacturing.
Mechanistic Insights into Palladium-Catalyzed Reductive Aminocarbonylation
The core of this synthetic innovation lies in the sophisticated interplay between the palladium catalyst system and the unique reactivity of the substrates. The reaction employs palladium acetate as the catalyst precursor, coordinated with tris(3-methoxyphenyl)phosphine ligands to facilitate the oxidative addition and subsequent migratory insertion steps. Molybdenum hexacarbonyl acts as a solid carbon monoxide source, releasing CO in situ to drive the carbonylation process without the need for high-pressure gas equipment, which is a major safety advantage for industrial applications. The presence of cesium carbonate as a base and tetrabutylammonium iodide as an additive is crucial for activating the allyl aryl ether and stabilizing the catalytic cycle. This specific combination of reagents enables the cleavage of the allyl ether bond and the subsequent formation of the new carbon-carbon and carbon-nitrogen bonds required to construct the quinolinone core. Understanding this mechanistic nuance is vital for R&D teams aiming to replicate or modify the process for specific analog synthesis.
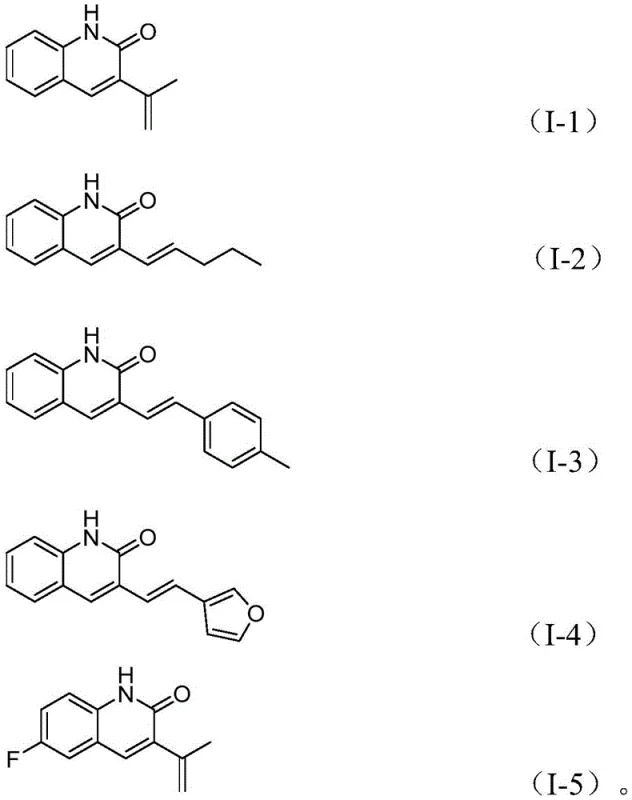
From an impurity control perspective, the high selectivity of this catalytic system is paramount for producing high-purity pharmaceutical intermediates. The reaction conditions, specifically maintaining a temperature of 100°C for 30 hours in acetonitrile, are optimized to maximize the conversion of the starting materials while minimizing side reactions such as homocoupling or incomplete cyclization. The broad substrate tolerance observed in the patent data indicates that the catalytic cycle is robust against various electronic and steric influences from substituents on the aromatic rings. For instance, the system accommodates electron-donating groups like methoxy and electron-withdrawing groups like trifluoromethyl or halogens without significant loss in efficiency. This robustness ensures a cleaner crude reaction profile, which simplifies downstream purification and reduces the burden on quality control laboratories tasked with meeting stringent purity specifications for clinical grade materials.
How to Synthesize 3-Alkenyl Quinolin-2(1H)one Derivatives Efficiently
Implementing this synthesis requires precise adherence to the stoichiometric ratios and reaction parameters outlined in the patent to achieve the reported excellent yields. The process begins with the careful weighing of the palladium catalyst, ligand, and carbonyl source, followed by the addition of the substrates and base in a sealed vessel to maintain the necessary pressure and prevent solvent loss. The reaction mixture is then subjected to thermal energy to initiate the catalytic cycle, after which standard workup procedures involving filtration and silica gel column chromatography are employed to isolate the pure product. While the general procedure is straightforward, attention to detail regarding the purity of reagents and the exclusion of air can further enhance reproducibility. For detailed operational specifics, please refer to the standardized synthesis guide below.
- Combine palladium acetate, tris(3-methoxyphenyl)phosphine, molybdenum carbonyl, cesium carbonate, and tetrabutylammonium iodide in a sealed tube.
- Add o-nitrobenzaldehyde and the specific allyl aryl ether substrate along with acetonitrile solvent to the mixture.
- Heat the reaction mixture at 100°C for 30 hours, then filter and purify via column chromatography to obtain the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage stems from the utilization of o-nitrobenzaldehyde and allyl aryl ethers, which are commercially available, inexpensive, and widely produced commodity chemicals. This reliance on bulk chemicals rather than specialized, custom-synthesized building blocks significantly de-risks the supply chain, ensuring continuity of supply even during market fluctuations. Furthermore, the elimination of high-pressure carbon monoxide gas cylinders in favor of a solid carbonyl source (Mo(CO)6) simplifies facility requirements and reduces regulatory compliance costs associated with hazardous gas storage. These factors collectively contribute to a more resilient and cost-effective manufacturing infrastructure for producing valuable quinolinone intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the dual functionality of o-nitrobenzaldehyde, which eliminates the need for separate nitrogen and carbonyl sources, thereby reducing raw material inventory costs. Additionally, the use of palladium acetate, while a precious metal, is employed at low loading levels (10 mol%), and the overall simplicity of the workup reduces solvent consumption and labor hours. The avoidance of complex protection-deprotection strategies common in alternative syntheses further streamlines the production timeline, leading to substantial cost savings in the overall manufacturing budget without compromising on yield or quality.
- Enhanced Supply Chain Reliability: By relying on generic starting materials like substituted benzaldehydes and allyl ethers, manufacturers can source inputs from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, which is a common cause of batch failures in more sensitive catalytic systems. This reliability translates directly into more predictable lead times for high-purity pharmaceutical intermediates, allowing downstream drug developers to plan their clinical trial material production with greater confidence and reduced buffer stock requirements.
- Scalability and Environmental Compliance: The transition from laboratory to commercial scale is facilitated by the use of standard heating and stirring equipment, avoiding the need for specialized high-pressure reactors. The reaction generates minimal hazardous waste compared to traditional methods that might produce stoichiometric amounts of salt byproducts or require toxic solvents. The ability to run the reaction in acetonitrile, a common and recoverable solvent, aligns with green chemistry principles and simplifies waste treatment protocols. This environmental compatibility is increasingly critical for maintaining operational licenses and meeting the sustainability goals of modern pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this technology. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production pipelines or for procurement specialists negotiating supply contracts for these specific intermediates.
Q: What are the key advantages of using o-nitrobenzaldehyde in this synthesis?
A: According to patent CN114478375A, o-nitrobenzaldehyde serves a dual role as both the nitrogen source and the formyl source, simplifying the reaction stoichiometry and reducing the need for multiple expensive reagents.
Q: What is the typical reaction temperature and time for this process?
A: The optimized protocol specifies a reaction temperature of 100°C maintained for approximately 30 hours to ensure complete conversion and high yields of the 3-alkenyl quinolin-2(1H)one derivatives.
Q: Does this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates wide substrate functional group tolerance, successfully accommodating substituents such as halogens, methoxy groups, and various alkyl chains on the allyl aryl ether component.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkenyl Quinolin-2(1H)one Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to accelerate drug development timelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity intermediates that meet rigorous QC labs standards, leveraging our state-of-the-art facilities to implement cutting-edge methods like the palladium-catalyzed reductive aminocarbonylation described herein. Our dedication to quality and scalability makes us an ideal partner for organizations seeking to secure a stable supply of complex heterocyclic building blocks.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this novel route for your supply chain. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial viability.
