Advanced Synthesis of 8-Methoxy-Quinolone Antibiotics for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for high-potency antibiotics, particularly within the fluoroquinolone class where the 8-methoxy substitution pattern confers superior Gram-positive and Gram-negative activity. Patent CN1200938C discloses a transformative methodology for producing these critical 8-methoxy-quinolone carboxylic acids, addressing longstanding inefficiencies in prior art. This innovation centers on a nucleophilic substitution strategy that replaces harsh, high-pressure conditions with a streamlined process utilizing potassium tert-butoxide in ether solvents. The significance of this technical breakthrough lies in its ability to synthesize complex intermediates like Bay 12-8039 and Gatifloxacin precursors with drastically improved operational simplicity. By shifting the reaction paradigm to atmospheric pressure and moderate temperatures, the patent offers a viable pathway for industrial manufacturers aiming to optimize their API supply chains. The core chemical transformation involves the selective displacement of a halogen atom at the 8-position of the quinolone nucleus, a step that has historically been a bottleneck in production due to sluggish kinetics and difficult purification requirements.
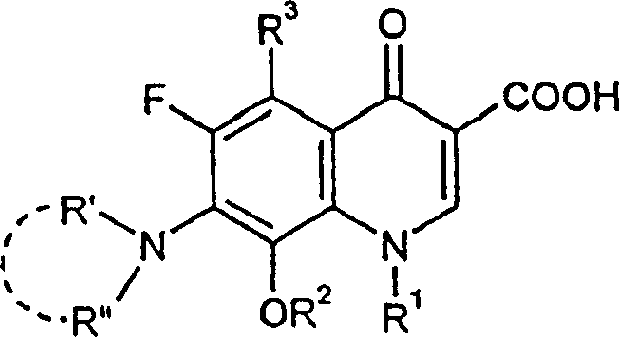
Traditional methods for introducing the 8-alkoxy group onto the quinolone scaffold have been plagued by severe practical limitations that hinder large-scale manufacturing efficiency. Prior art, such as the processes described in EP-A-106 489 and EP-A-230 295, often relies on the use of sodium methylate in polar aprotic solvents like DMF or DMSO, necessitating extreme reaction conditions. These conventional approaches frequently require sealed pressure vessels operating at temperatures between 140°C and 150°C to drive the conversion, posing significant safety risks and equipment costs for production facilities. Furthermore, the reaction kinetics in these older methods are notoriously slow, with some protocols requiring reflux times extending up to 70 hours to achieve acceptable conversion levels, as evidenced by comparative data showing minimal progress even after extended periods. The use of excess alkali metal alcoholates in these traditional routes also complicates the downstream processing, as separating the resulting salts from high-boiling solvents like DMF is energetically expensive and technically challenging. Additionally, certain sensitive substrates, such as the specific bicyclic amine derivatives found in Bay 12-8039, are prone to decomposition or simply fail to react under these forcing conditions, rendering the old methods unsuitable for modern, high-value antibiotic production.
In stark contrast, the novel approach detailed in the patent utilizes a specific combination of potassium tert-butoxide or potassium tertiary amyloxide in aliphatic or cyclic ether solvents to overcome these kinetic and thermodynamic barriers. This new methodology operates effectively under atmospheric pressure at temperatures ranging from 20°C to the boiling point of the solvent, typically around 60-70°C for THF, which represents a massive reduction in thermal stress on the equipment and the molecule. The choice of solvent is critical; the patent highlights that ethers like tetrahydrofuran (THF), dioxane, or glycol dimethyl ether provide a superior reaction medium compared to the polar aprotic solvents of the past. This solvent system facilitates a much faster reaction rate, reducing the total process time from days to merely a few hours, specifically noting completion within approximately 2.5 hours in optimized embodiments. The use of a bulky base like potassium tert-butoxide appears to enhance the nucleophilicity of the alcohol species without promoting unwanted side reactions, allowing for high yields even with sterically demanding amine substituents at the 7-position. This shift not only improves safety by eliminating high-pressure reactors but also simplifies the workup procedure significantly.
Mechanistic Insights into Potassium Tert-Butoxide Mediated Nucleophilic Substitution
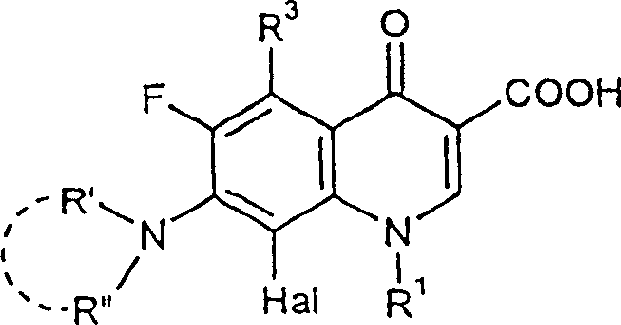
The mechanistic advantage of this process lies in the unique interplay between the bulky alkoxide base and the ether solvent environment, which likely enhances the effective concentration of the reactive methoxide species in situ. When potassium tert-butoxide is introduced into the mixture containing methanol and the 8-halo-quinolone substrate, an equilibrium is established that generates the active nucleophile required for the displacement of the halogen at the 8-position. Unlike sodium methylate, which can form tight ion pairs that reduce reactivity in certain solvents, the potassium cation paired with the bulky tert-butoxide anion creates a more dissociated and reactive environment in THF. This facilitates a smoother SNAr (Nucleophilic Aromatic Substitution) mechanism on the electron-deficient quinolone ring system. The presence of the electron-withdrawing carbonyl group at the 4-position and the carboxylic acid (or ester) at the 3-position activates the ring towards nucleophilic attack, but the steric hindrance at the 8-position usually retards this process. The novel conditions overcome this steric barrier efficiently, ensuring that the substitution occurs selectively at the 8-position without affecting other sensitive functional groups, such as the cyclopropyl group at N-1 or the complex diazabicyclo nonane moiety at the 7-position.
Impurity control is another critical aspect where this mechanism offers distinct advantages over conventional routes. In traditional high-temperature methods, the prolonged exposure to strong bases and heat often leads to the degradation of the quinolone core or the hydrolysis of the ester groups if present. The milder conditions of the new process minimize thermal degradation pathways, resulting in a cleaner crude reaction profile. Furthermore, the specific choice of reagents avoids the formation of difficult-to-remove byproducts that often arise from solvent decomposition or over-alkylation. The patent describes a straightforward isolation method where the reaction mixture is directly treated with dilute hydrochloric acid to precipitate the product as a hydrochloride salt. This acidification step not only neutralizes the excess base but also converts the free acid or salt form of the product into a crystalline solid that can be easily filtered. This direct precipitation bypasses the need for complex extraction or chromatographic purification steps, which are major sources of yield loss and cost in pharmaceutical manufacturing. The resulting solid can then be further purified by simple recrystallization from water or water-alcohol mixtures, achieving purity levels suitable for medicinal application without extensive processing.
How to Synthesize 8-Methoxy-Quinolone Efficiently
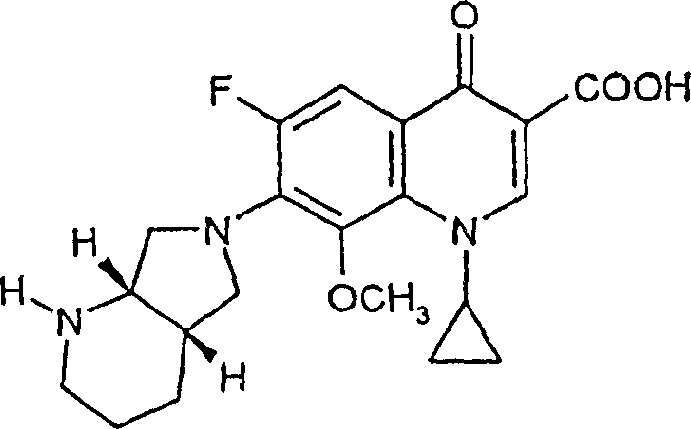
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for implementing this technology in a pilot or production setting. The process begins with the dissolution of the 8-halo-quinolone precursor, such as the 6,8-difluoro derivative, in a suitable volume of tetrahydrofuran combined with a stoichiometric amount of methanol. This mixture is heated to initiate the reaction, followed by the controlled addition of a potassium tert-butoxide solution, typically added in portions to manage the exotherm and ensure complete conversion. The reaction is maintained at reflux for a short duration, monitored by HPLC to confirm the disappearance of the starting material. Upon completion, the mixture is cooled, and the product is isolated via acidification and filtration. For a comprehensive understanding of the precise stoichiometric ratios, temperature profiles, and specific workup parameters required to replicate these results, please refer to the standardized synthesis steps provided below.
- Dissolve the 8-halo-quinolone carboxylic acid derivative (such as the 6,8-difluoro precursor) in an aliphatic or cyclic ether solvent like tetrahydrofuran (THF) along with a stoichiometric amount of methanol.
- Add a solution of potassium tert-butoxide (or potassium tertiary amyloxide) in THF to the mixture while heating to reflux temperature, maintaining atmospheric pressure.
- After the reaction completes (typically within 2.5 hours), cool the mixture and precipitate the hydrochloride salt by adding dilute hydrochloric acid, followed by filtration and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers substantial strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the manufacturing infrastructure required. By eliminating the need for high-pressure autoclaves and the associated safety protocols, facilities can utilize standard glass-lined or stainless steel reactors that are already common in fine chemical plants. This reduces capital expenditure (CAPEX) for new production lines and allows for greater flexibility in scheduling campaigns. The reduction in reaction time from several days to just a few hours significantly increases the throughput of existing assets, allowing manufacturers to respond more rapidly to market demand fluctuations without needing to build additional capacity. Furthermore, the use of common, commodity chemicals like THF, methanol, and potassium tert-butoxide ensures a stable and reliable supply of raw materials, mitigating the risk of shortages that can occur with specialized reagents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of energy-intensive steps and the reduction of solvent usage. Conventional methods requiring high temperatures and long reaction times consume significant amounts of energy for heating and cooling cycles, whereas this atmospheric pressure process operates at much lower thermal loads. Additionally, the ability to isolate the product directly as a hydrochloride salt through simple filtration removes the need for expensive chromatographic purification or complex solvent swaps, which are major cost drivers in API production. The higher yield and purity achieved in the crude stage mean less material is lost during downstream processing, directly improving the overall cost of goods sold (COGS). By avoiding the use of high-boiling solvents like DMF or DMSO, the process also reduces the cost and environmental burden associated with solvent recovery and waste disposal.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method translates directly into improved supply chain continuity. Because the reaction conditions are mild and tolerant, the process is less prone to batch failures caused by minor deviations in temperature or pressure, leading to more consistent production outcomes. The reliance on widely available reagents ensures that the supply chain is not vulnerable to the bottlenecks often seen with exotic catalysts or specialized intermediates. This reliability allows for more accurate forecasting and inventory management, reducing the need for excessive safety stock. Moreover, the simplified workup procedure shortens the overall cycle time from raw material intake to finished goods, enabling faster turnaround times for customer orders and reducing lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous high-pressure steps and the use of standard unit operations like filtration and crystallization. The process aligns well with green chemistry principles by reducing energy consumption and minimizing waste generation through higher atom economy and simpler isolation techniques. The avoidance of chlorinated solvents and the use of recyclable ethers like THF further enhance the environmental profile of the manufacturing process. This compliance with stringent environmental regulations reduces the regulatory burden on the manufacturer and minimizes the risk of production shutdowns due to environmental non-compliance. The ability to produce high-quality intermediates with a smaller environmental footprint is increasingly becoming a key differentiator in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 8-methoxy-quinolone synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this process into your existing manufacturing portfolio.
Q: Why is potassium tert-butoxide preferred over sodium methylate for this synthesis?
A: Potassium tert-butoxide allows the reaction to proceed under much milder conditions (atmospheric pressure, lower temperatures) compared to sodium methylate, which often requires sealed vessels at 140-150°C and excessively long reaction times up to 70 hours.
Q: What solvents are compatible with this novel 8-methoxylation process?
A: The process specifically utilizes aliphatic or cyclic aliphatic ethers with 4 to 6 carbon atoms, with tetrahydrofuran (THF), dioxane, and glycol dimethyl ether being the most effective for achieving high conversion rates and short reaction times.
Q: How is the final product purified without column chromatography?
A: The method enables direct precipitation of the hydrochloride salt by acidifying the reaction mixture, followed by simple recrystallization from water or water/alcohol mixtures, eliminating the need for complex chromatographic separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Methoxy-Quinolone Supplier
The technological advancements described in patent CN1200938C represent a significant leap forward in the production of next-generation fluoroquinolone antibiotics, offering a pathway to higher purity and lower costs. NINGBO INNO PHARMCHEM stands ready to leverage this expertise as your trusted partner, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this chemistry, ensuring that stringent purity specifications are met consistently. With our rigorous QC labs and commitment to process optimization, we can deliver high-purity 8-methoxy-quinolone intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to engage with our technical team to explore how this innovative synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this method for your API projects. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless transition to a more efficient and reliable supply source.
