Advanced Synthesis of 6,7-Diacyl-7-deacetylforskolin Derivatives for Pharmaceutical Scale-Up
Advanced Synthesis of 6,7-Diacyl-7-deacetylforskolin Derivatives for Pharmaceutical Scale-Up
The pharmaceutical industry continuously seeks robust, scalable, and high-yielding synthetic routes for complex natural product derivatives, particularly those with potent biological activity such as forskolin analogs. Patent CN1030583A introduces a groundbreaking methodology for the preparation of 6,7-diacyl-7-deacetylforskolin derivatives, addressing critical bottlenecks in regioselectivity and purification that have plagued previous manufacturing attempts. This technical disclosure outlines two distinct yet complementary pathways: a chemical route involving selective acylation followed by controlled solvolysis, and a thermal route utilizing intramolecular acyl migration. For R&D directors and process chemists, this represents a significant leap forward in controlling the impurity profile of these valuable cardiovascular and metabolic intermediates. By shifting away from non-selective direct acylation, manufacturers can now access high-purity materials without relying on labor-intensive chromatographic separations, thereby aligning synthetic efficiency with commercial viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in European Patent 222413A, typically rely on the direct acylation of 7-deacetylforskolin derivatives. The fundamental flaw in this approach lies in the similar reactivity of the hydroxyl groups at the C-1 and C-7 positions. When attempting to introduce an acyl group specifically at the C-7 position (or subsequently manipulating it), the lack of discrimination leads to a mixture of 1-acyl, 7-acyl, and 1,7-diacyl byproducts. This poor regioselectivity necessitates rigorous purification steps, often involving silica gel column chromatography, which is notoriously difficult to scale beyond kilogram quantities due to solvent consumption, silica waste, and throughput limitations. Furthermore, the formation of these isomeric impurities complicates the downstream synthesis of 6-acyl derivatives, as separating the 1-acyl isomer from the desired 7-acyl precursor is chemically challenging and economically inefficient.
The Novel Approach
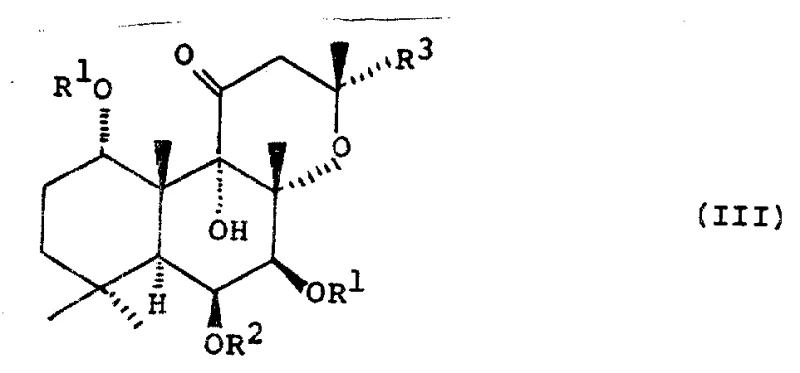
The methodology presented in CN1030583A circumvents these selectivity issues through a clever strategic inversion. Instead of struggling to acylate the C-7 position directly with high fidelity, the inventors propose acylating both the C-1 and C-7 hydroxyls to form a 1,6,7-triacyl intermediate (Formula III), followed by a highly selective solvolysis step that removes the acyl group exclusively at the C-1 position. 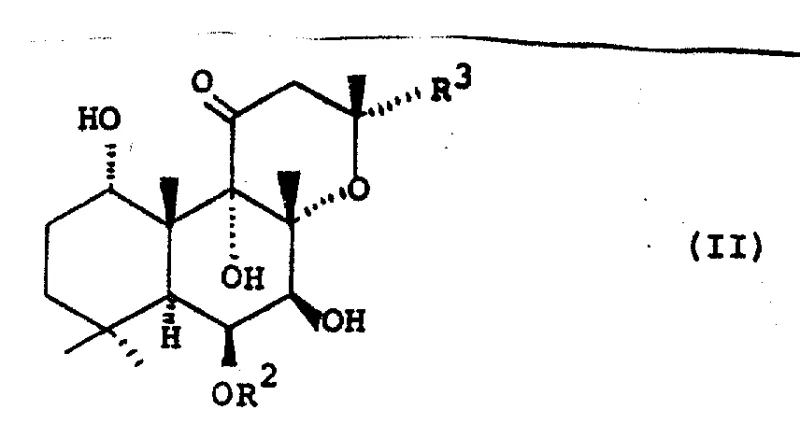
 Alternatively, the patent discloses a thermal rearrangement pathway where a 7-acyl-7-deacetylforskolin derivative (Formula 2) is heated to induce a spontaneous migration of the acyl group from C-7 to C-6. This thermal method is particularly elegant as it can be performed under solvent-free conditions, drastically reducing the environmental footprint and operational complexity associated with large-volume solvent handling.
Alternatively, the patent discloses a thermal rearrangement pathway where a 7-acyl-7-deacetylforskolin derivative (Formula 2) is heated to induce a spontaneous migration of the acyl group from C-7 to C-6. This thermal method is particularly elegant as it can be performed under solvent-free conditions, drastically reducing the environmental footprint and operational complexity associated with large-volume solvent handling.
Mechanistic Insights into Selective Solvolysis and Thermal Rearrangement
The success of the chemical pathway hinges on the differential stability of the ester bonds at the C-1 and C-7 positions within the 1,6,7-triacyl intermediate. The C-1 acyl group is sterically and electronically more susceptible to nucleophilic attack by hydroxide or alkoxide ions compared to the C-7 acyl group, which is shielded by the rigid diterpene skeleton. By carefully controlling the reaction parameters—specifically maintaining temperatures between 0°C and room temperature and limiting reaction times to minutes rather than hours—the process achieves kinetic control. This ensures that the hydrolysis stops precisely after the cleavage of the C-1 ester, leaving the C-6 and C-7 esters intact. The use of mild bases like sodium bicarbonate or dilute sodium hydroxide in aqueous-alcoholic solvents further fine-tunes this selectivity, preventing the unwanted saponification of the sensitive C-7 ester bond.
In the thermal rearrangement pathway, the mechanism involves a concerted intramolecular acyl shift. Heating the 7-acyl precursor (Formula 2) to temperatures ranging from 130°C to 300°C provides the activation energy required for the acyl group to migrate from the C-7 oxygen to the adjacent C-6 carbon. 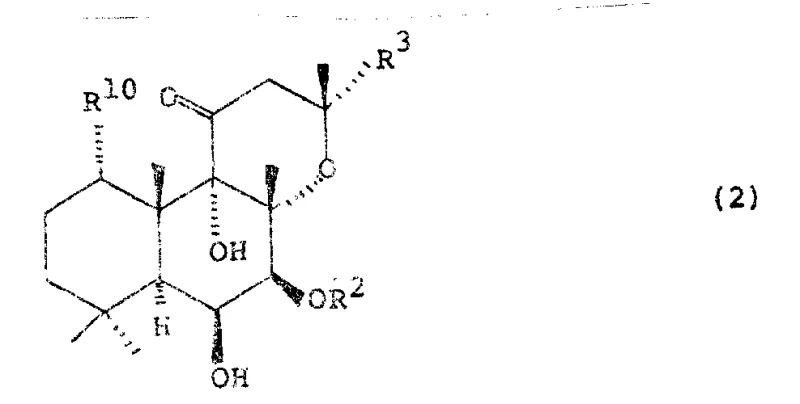 This transformation is driven by the thermodynamic stability of the resulting 6-acyl configuration. Crucially, this rearrangement can be catalyzed or facilitated by the presence of specific amines if the acyl group contains beta-amino substituents, which suppresses side reactions such as elimination or decomposition. The ability to perform this rearrangement in the melt phase (solvent-free) not only accelerates the reaction kinetics due to high concentration but also simplifies the isolation of the product to a straightforward recrystallization, effectively eliminating the need for chromatographic purification entirely.
This transformation is driven by the thermodynamic stability of the resulting 6-acyl configuration. Crucially, this rearrangement can be catalyzed or facilitated by the presence of specific amines if the acyl group contains beta-amino substituents, which suppresses side reactions such as elimination or decomposition. The ability to perform this rearrangement in the melt phase (solvent-free) not only accelerates the reaction kinetics due to high concentration but also simplifies the isolation of the product to a straightforward recrystallization, effectively eliminating the need for chromatographic purification entirely.
How to Synthesize 6,7-Diacyl-7-deacetylforskolin Efficiently
The synthesis of these high-value intermediates can be executed via two primary routes depending on the available starting materials and equipment capabilities. The chemical route offers precise control at lower temperatures, ideal for heat-sensitive substrates, while the thermal route offers superior atom economy and waste reduction for robust scale-up. Both methods converge on the same high-purity product profile, characterized by the absence of 1-acyl isomers. The detailed standardized operating procedures, including specific molar ratios, solvent choices, and crystallization protocols optimized for GMP manufacturing, are outlined below.
- Perform selective acylation on the 1,7-dihydroxy substrate using acylating agents like acetic anhydride in pyridine to form the 1,6,7-triacyl intermediate.
- Execute selective solvolysis (hydrolysis or alcoholysis) under mild alkaline conditions to remove the acyl group specifically at the C-1 position.
- Alternatively, heat the 7-acyl-7-deacetylforskolin precursor at 130-300°C to induce thermal rearrangement of the acyl group from C-7 to C-6.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the processes described in CN1030583A translates directly into substantial cost reductions and enhanced supply reliability. The most significant economic driver is the elimination of silica gel column chromatography. In traditional fine chemical manufacturing, chromatography is a major cost center, consuming vast amounts of solvents and silica, requiring specialized equipment, and creating significant hazardous waste disposal liabilities. By replacing this with simple recrystallization, the new method drastically lowers the cost of goods sold (COGS) and shortens the production cycle time. Furthermore, the thermal rearrangement option allows for solvent-free processing, which not only reduces raw material costs but also mitigates supply chain risks associated with volatile organic compound (VOC) regulations and solvent availability.
- Cost Reduction in Manufacturing: The transition from chromatographic purification to recrystallization represents a paradigm shift in processing economics. Chromatography is inherently batch-limited and resource-intensive, whereas crystallization is easily scalable and continuous. By removing the acyl group at the C-1 position selectively, the process avoids the generation of difficult-to-separate isomers, meaning less material is lost to waste streams. Additionally, the thermal method's ability to operate without solvents removes the cost of solvent purchase, recovery, and incineration, leading to a significantly leaner manufacturing budget.
- Enhanced Supply Chain Reliability: The robustness of the thermal rearrangement reaction, which tolerates a wide range of temperatures and does not require sensitive catalysts or anhydrous conditions, ensures consistent batch-to-batch quality. This reliability is critical for maintaining uninterrupted supply to downstream API manufacturers. The use of commodity chemicals like acetic anhydride, pyridine, and common alcohols as reagents ensures that the supply chain is not dependent on exotic or single-source specialty reagents, thereby reducing the risk of raw material shortages.
- Scalability and Environmental Compliance: The solvent-free nature of the thermal rearrangement pathway aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations. Reducing solvent usage minimizes the facility's VOC emissions and wastewater treatment load. This environmental compliance facilitates faster regulatory approvals and reduces the long-term liability associated with hazardous waste management. The high yields reported in the patent examples, such as the 84% overall yield in the thermal rearrangement of specific derivatives, demonstrate that this efficiency does not come at the expense of productivity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: How does the novel acylation method improve selectivity compared to conventional direct acylation?
A: Conventional direct acylation often lacks selectivity between the C-1 and C-7 hydroxyl groups. The novel method described in CN1030583A utilizes a protection-deprotection strategy or specific thermal rearrangement to ensure the acyl group ends up exclusively at the C-6 position after deacetylation, significantly reducing isomeric impurities.
Q: What are the advantages of the thermal rearrangement method for large-scale production?
A: The thermal rearrangement method allows for solvent-free operation at elevated temperatures (e.g., 160°C). This eliminates the need for large volumes of organic solvents, reduces waste disposal costs, and simplifies the work-up procedure to a simple recrystallization, making it highly suitable for industrial scale-up.
Q: Is chromatographic purification required for the final product?
A: No. A key advantage of this patented process is that the final products can be purified via simple recrystallization from solvents like acetone, ethanol, or hexane/ethyl acetate mixtures. This avoids the expensive and time-consuming silica gel column chromatography required by older methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,7-Diacyl-7-deacetylforskolin Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable, and compliant manufacturing processes for complex pharmaceutical intermediates. Our technical team has extensively analyzed the methodologies disclosed in CN1030583A and possesses the expertise to implement both the selective solvolysis and thermal rearrangement pathways at commercial scale. We understand that moving from gram-scale laboratory synthesis to multi-ton production requires more than just a recipe; it demands rigorous process safety analysis, impurity control strategies, and robust quality assurance systems. Our facility is equipped to handle diverse synthetic pathways, ranging from cryogenic reactions to high-temperature melt processes, ensuring that your specific forskolin derivative requirements are met with precision.
We invite you to leverage our capabilities to optimize your supply chain for these high-value intermediates. Our commitment to excellence means we can offer a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating exactly how switching to our optimized process can reduce your overall procurement costs. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us partner with you to bring these advanced forskolin derivatives from the patent lab to the global market efficiently and reliably.
