Advanced Zinc-Catalyzed Silane Reduction for Scalable Pharmaceutical Intermediate Production
Advanced Zinc-Catalyzed Silane Reduction for Scalable Pharmaceutical Intermediate Production
The landscape of organic synthesis is continuously evolving towards safer, more sustainable, and cost-effective methodologies, particularly in the production of high-value pharmaceutical intermediates. Patent CN1208295C introduces a groundbreaking process for the selective reduction of carbonyl compounds, including aldehydes, ketones, esters, and lactones, utilizing silanes as reducing agents in the presence of novel zinc catalysts. This technology represents a significant departure from traditional stoichiometric hydride reductions, offering a catalytic cycle that operates under mild conditions with exceptional chemoselectivity. By leveraging monomeric zinc complexes coordinated by basic ligands, this method overcomes the historical limitations of zinc-catalyzed reductions, specifically enabling the efficient transformation of esters and lactones which were previously resistant to such systems without hazardous activators. For industrial manufacturers, this patent provides a robust framework for developing scalable routes to chiral alcohols and fatty alcohols, addressing critical needs for purity and process safety in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of carbonyl functionalities to their corresponding alcohols has relied heavily on stoichiometric anhydride reducing agents such as lithium aluminum hydride (LiAlH4) and sodium borohydride (NaBH4). While effective for simple aldehydes and ketones, these reagents present substantial challenges for industrial application, particularly when dealing with esters and lactones where reactivity is often limited or requires harsh conditions. A primary concern is the inherent safety risk associated with these hydrides; they are pyrophoric, react violently with moisture to release explosive hydrogen gas, and necessitate rigorous inert atmosphere controls that drive up operational costs. Furthermore, the stoichiometric nature of these reactions generates significant amounts of metal salt waste, complicating downstream purification and environmental compliance. The inability of traditional zinc-silane systems to reduce esters without prior activation by dangerous reducing agents like NaBH4 further highlighted the need for a more sophisticated catalytic approach that could bridge the gap between safety and reactivity.
The Novel Approach
The innovative methodology described in the patent data circumvents these issues by employing a catalytic system based on monomeric zinc compounds activated by specific ligands, paired with polymethylhydrosiloxane (PMHS) as a safe and economical silane source. Unlike prior art which required the in situ generation of zinc hydrides using hazardous activators, this process utilizes pre-formed or in situ generated monomeric zinc complexes that are intrinsically active towards a broad spectrum of carbonyl substrates. The key breakthrough lies in the use of ligands such as amines, amino alcohols, and imines to disrupt the polymeric structure of common zinc salts, creating electron-deficient monomeric species capable of activating the silane. This approach not only eliminates the need for stoichiometric hydrides but also ensures high chemoselectivity, allowing for the reduction of esters and lactones in the presence of other sensitive functional groups like olefins, thereby streamlining synthetic routes for complex pharmaceutical intermediates.
Mechanistic Insights into Zinc-Ligand Activated Reduction
The core mechanistic advantage of this technology stems from the structural modification of the zinc catalyst. Standard zinc carboxylates typically exist as oligomeric or polymeric structures due to the metal's tendency to achieve higher coordination numbers, rendering them electronically saturated and catalytically inert for ester reduction. The patent elucidates that by introducing basic ligands—such as dimethylaminoethanol, bipyridine, or TMEDA—the polymeric zinc network is depolymerized into discrete monomeric complexes. These monomeric species possess open coordination sites that facilitate the activation of the silane reducing agent, initiating the hydride transfer to the carbonyl substrate. This ligand-induced activation is crucial, as it transforms inexpensive, stable zinc salts into highly reactive catalysts without altering the oxidation state of the metal through dangerous pre-reduction steps.
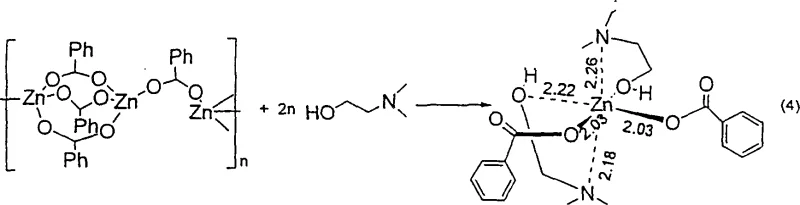
Furthermore, the versatility of the ligand system allows for fine-tuning the electronic and steric environment around the zinc center, optimizing activity for specific substrate classes. As illustrated in the structural data, complexes like [Zn(benzoate)2(Me2NCH2CH2OH)2] demonstrate how bidentate coordination stabilizes the monomeric form, ensuring sustained catalytic turnover. This mechanistic understanding is vital for R&D teams aiming to replicate the process, as it underscores the importance of ligand selection in achieving high conversion rates. The preservation of stereochemistry in unsaturated substrates is another critical mechanistic feature; the mild nature of the zinc-silane interaction prevents the isomerization or hydrogenation of carbon-carbon double bonds, a common side reaction in traditional catalytic hydrogenation. This selectivity is paramount for synthesizing bioactive molecules where geometric integrity dictates biological activity.
How to Synthesize Active Zinc Catalysts Efficiently
Implementing this reduction technology requires the precise preparation of the active zinc catalyst species, which can be achieved through straightforward coordination chemistry protocols. The process generally involves reacting a zinc precursor, such as zinc benzoate or zinc diethylacetate, with a molar excess of the chosen ligand in a suitable organic solvent. The reaction is typically exothermic and proceeds rapidly at ambient temperatures, yielding the active monomeric complex as a isolable solid or a stable solution. For detailed operational parameters regarding solvent choices, stoichiometry, and purification techniques, operators should refer to the specific experimental embodiments provided in the patent documentation which outline the synthesis of various active complexes.
- Dissolve zinc carboxylate precursor in a suitable organic solvent such as dichloromethane or diisopropyl ether.
- Add the selected basic ligand, such as dimethylaminoethanol or bipyridine, to the zinc suspension under stirring.
- Stir the mixture at ambient temperature to allow exothermic complexation, then isolate the solid monomeric catalyst via crystallization or filtration.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this zinc-catalyzed silane reduction offers transformative benefits that directly impact the bottom line and operational resilience. The shift from stoichiometric hydrides to a catalytic system drastically reduces the consumption of expensive and hazardous reagents, leading to significant cost reduction in fine chemical manufacturing. By utilizing PMHS, a by-product of the silicone industry that is abundant and low-cost, companies can decouple their production costs from the volatile pricing of specialized reducing agents. Moreover, the elimination of pyrophoric materials simplifies logistics and storage requirements, reducing the need for specialized hazmat handling and insurance costs associated with dangerous goods. This stability enhances supply chain reliability, ensuring that production schedules are not disrupted by the scarcity or regulatory restrictions often placed on high-risk chemicals.
- Cost Reduction in Manufacturing: The transition to a catalytic process fundamentally alters the cost structure of alcohol production by minimizing reagent usage and waste disposal expenses. Traditional hydride reductions generate large volumes of metal sludge that require costly treatment, whereas the zinc-silane system produces siloxane by-products that are easier to manage and potentially recyclable. The ability to use low catalyst loadings, typically in the range of 1 to 5 mole percent, further amplifies these savings, making the process economically viable for large-scale commercial production. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, contributing to overall operational efficiency and lower utility costs for the manufacturing facility.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents like zinc salts and PMHS mitigates the risks associated with the supply of sensitive hydrides which often have short shelf lives and strict transport regulations. This stability allows for bulk purchasing and long-term inventory planning, shielding the supply chain from market fluctuations and delivery delays. The robustness of the catalyst system also means that production can be scaled up with greater confidence, as the reagents are less prone to degradation during storage. For global supply chains, this translates to reduced lead times for high-purity pharmaceutical intermediates, as the manufacturing process is less susceptible to interruptions caused by reagent instability or regulatory compliance issues regarding hazardous material transport.
- Scalability and Environmental Compliance: The environmental profile of this process aligns perfectly with modern green chemistry initiatives, facilitating easier regulatory approval and community acceptance for manufacturing sites. The absence of heavy metal contaminants in the final product reduces the burden on purification steps, while the benign nature of the reagents simplifies wastewater treatment protocols. Scalability is inherently supported by the homogeneous nature of the catalytic system, which allows for efficient heat and mass transfer in large reactors without the safety concerns of runaway exotherms typical of hydride additions. This makes the technology ideal for the commercial scale-up of complex pharmaceutical intermediates, ensuring that production can meet growing market demand without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this zinc-catalyzed reduction technology. These insights are derived directly from the experimental data and claims within the patent, providing clarity on substrate scope and operational parameters. Understanding these nuances is essential for process chemists evaluating the feasibility of this route for their specific target molecules.
Q: Why is the monomeric zinc complex superior to traditional zinc salts for ester reduction?
A: Traditional zinc salts often exist as polymeric or oligomeric structures which are electronically saturated and catalytically inactive for ester reduction. The patented process utilizes specific ligands to break these polymeric structures into active monomeric species, significantly enhancing reactivity towards esters and lactones without requiring hazardous hydride activators.
Q: Does this reduction method affect carbon-carbon double bonds in unsaturated substrates?
A: No, one of the key advantages of this zinc-silane system is its high chemoselectivity. It selectively reduces the carbonyl group (C=O) while leaving olefinic double bonds (C=C) intact, preserving the stereochemistry of unsaturated fatty acid derivatives and pharmaceutical intermediates.
Q: What are the safety advantages of using PMHS over lithium aluminum hydride?
A: Polymethylhydrosiloxane (PMHS) is a stable, non-pyrophoric polymer that does not release hydrogen gas upon contact with moisture, unlike lithium aluminum hydride (LiAlH4). This eliminates explosion hazards and the need for strictly inert atmospheres, greatly simplifying operational safety and waste disposal protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Zinc Catalyst Reduction Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting advanced catalytic technologies like the zinc-silane reduction system to enhance production capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of pharmaceutical intermediate meets the highest global standards. Our expertise in handling sensitive organometallic catalysis allows us to optimize reaction conditions for maximum yield and minimal impurity formation, delivering consistent quality for our partners.
We invite you to collaborate with us to leverage this innovative reduction technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific synthesis requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how this zinc-catalyzed process can optimize your supply chain and reduce manufacturing costs effectively.
