Optimizing Vismodegib Production: A Novel Three-Step Synthetic Route for Commercial Scale-Up
Optimizing Vismodegib Production: A Novel Three-Step Synthetic Route for Commercial Scale-Up
The pharmaceutical landscape for oncology treatments continues to evolve, with Vismodegib (CAS 879085-55-9) standing out as a critical Hedgehog pathway inhibitor for treating basal cell carcinoma. As global demand for this potent therapeutic agent rises, the efficiency of its supply chain becomes paramount for downstream drug manufacturers. Recent intellectual property developments, specifically patent CN107200708B, have unveiled a groundbreaking preparation method that fundamentally restructures the synthetic approach to this molecule. This new methodology shifts away from complex, multi-step organometallic couplings towards a more direct construction of the biaryl pyridine core. By leveraging a strategic 1,4-addition and cyclization sequence, the process achieves exceptional purity levels, reportedly reaching 99.96%, which is a critical metric for regulatory compliance in API production. 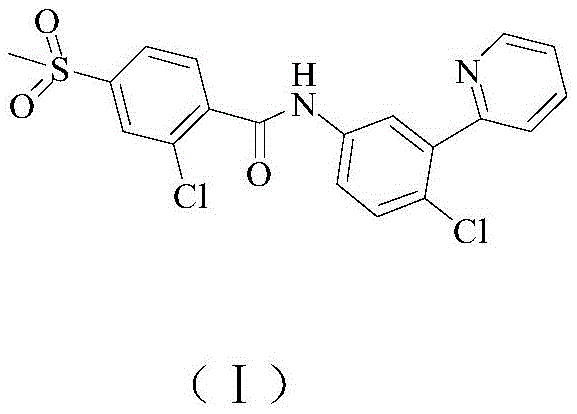
For R&D directors and procurement strategists, understanding the nuances of this patent is essential for securing a reliable API intermediate supplier. The disclosed route not only promises significant cost reduction in pharmaceutical manufacturing but also addresses the growing industry imperative for greener, safer chemical processes. Unlike previous iterations that relied on hazardous reagents, this method utilizes 2-chloro-5-nitroacetophenone as a robust starting block. This shift represents a major technological leap, offering a pathway to stabilize supply chains against the volatility of exotic reagent markets. The following analysis dissects the technical merits and commercial implications of this innovation, providing a comprehensive roadmap for stakeholders looking to optimize their Vismodegib sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Vismodegib has been plagued by reliance on sophisticated and often hazardous chemistries that pose significant barriers to efficient industrial scale-up. One prominent prior art route, exemplified in US7888364, employs an organozinc reagent to construct the critical carbon-carbon bond between the pyridine and phenyl rings. 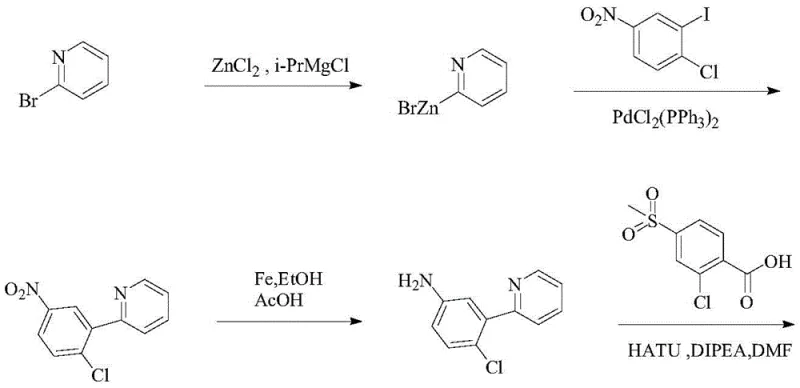
While chemically valid, the generation and handling of organic zinc species introduce severe operational safety risks and require stringent anhydrous conditions, driving up infrastructure costs. Furthermore, these organometallic intermediates often exhibit poor stability, leading to inconsistent yields and complicating inventory management. Another existing pathway utilizes diphenyl phosphorazidate, a highly active and dangerous reagent that necessitates specialized handling protocols and generates substantial toxic waste streams. These conventional methods are characterized by long reaction sequences, low overall yields, and a heavy dependence on expensive transition metal catalysts like palladium, which can contaminate the final product and require costly purification steps to meet residual metal specifications.
The Novel Approach
In stark contrast, the methodology disclosed in CN107200708B introduces a streamlined, three-step synthesis that bypasses the need for fragile organometallic intermediates entirely. 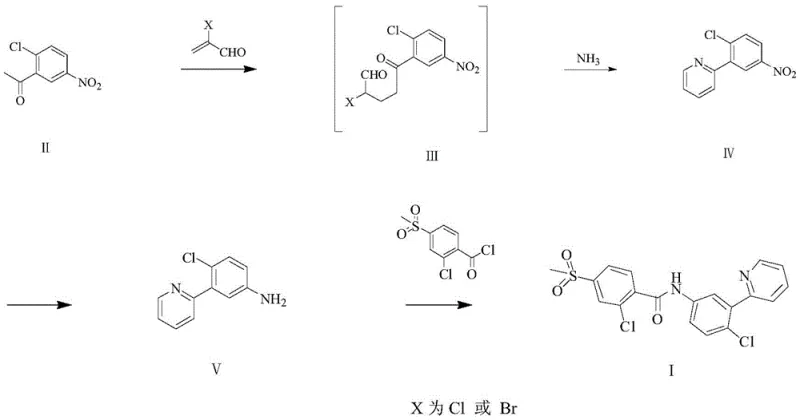
This innovative route initiates with a tandem 1,4-addition and cyclization reaction, directly assembling the 2-(2-chloro-5-nitrophenyl)pyridine scaffold from commodity chemicals. By replacing the cross-coupling strategy with a condensation approach using ammonia, the process eliminates the requirement for expensive boron or zinc reagents. The subsequent reduction of the nitro group and final amidation are performed under mild conditions that are inherently safer and more energy-efficient. This structural simplification of the synthetic tree not only reduces the number of unit operations but also drastically minimizes the generation of three wastes, aligning perfectly with modern environmental, health, and safety (EHS) standards. The result is a robust, high-yielding process capable of delivering high-purity API intermediates with superior economic viability.
Mechanistic Insights into the Tandem Addition-Cyclization Strategy
The cornerstone of this novel synthesis lies in the first step, where 2-chloro-5-nitroacetophenone undergoes a 1,4-addition reaction with 2-haloacrolein. This transformation is catalyzed by organic bases such as piperidine or DBU, facilitating the nucleophilic attack of the enolate derived from the acetophenone onto the electron-deficient double bond of the acrolein derivative. The resulting intermediate, a 1,5-dicarbonyl equivalent, spontaneously undergoes cyclization in the presence of ammonia water to form the pyridine ring. This tandem sequence is mechanistically elegant, as it constructs the heterocyclic core and installs the aryl substituent in a single pot, thereby avoiding the isolation of unstable intermediates. The choice of solvent, typically dichloromethane or 1,2-dichloroethane, plays a crucial role in solubilizing the reactants while allowing for effective phase separation during the workup, ensuring high recovery rates of the valuable pyridine intermediate.
Following the construction of the biaryl scaffold, the process employs a selective reduction of the nitro group to an amine. This step can be achieved via catalytic hydrogenation using palladium on carbon or Raney nickel, or alternatively through chemical reduction with iron powder. The flexibility in reduction methods allows manufacturers to choose the most cost-effective option based on their existing infrastructure; for instance, iron powder reduction avoids the need for high-pressure hydrogenation equipment, further lowering capital expenditure. The final amidation step couples the newly formed aniline with 2-chloro-4-methylsulfonylbenzoyl chloride. Conducted in the presence of an acid-binding agent like sodium carbonate or triethylamine, this reaction proceeds with high selectivity at low temperatures (0-30°C), minimizing the formation of hydrolysis byproducts and ensuring the final product meets stringent purity specifications required for clinical applications.
How to Synthesize Vismodegib Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and purity. The process begins with the careful addition of 2-haloacrolein to the acetophenone substrate, maintaining temperatures between 10-35°C to prevent polymerization of the acrolein. Following the addition-cyclization sequence, the crude pyridine intermediate is isolated and subjected to reduction. Whether using catalytic hydrogenation at 0.5-1.5 MPa or refluxing with iron powder, the goal is complete conversion of the nitro group without affecting the chloro substituents. Finally, the amidation is performed under controlled cooling to manage the exotherm, followed by a recrystallization from ethanol to achieve the final pharmaceutical grade quality.
- Perform a 1,4-addition reaction between 2-chloro-5-nitroacetophenone and 2-haloacrolein, followed by condensation with ammonia to form the pyridine ring.
- Reduce the nitro group of the intermediate 2-(2-chloro-5-nitrophenyl)pyridine to an amino group using catalytic hydrogenation or iron powder reduction.
- Conduct a final amidation reaction between the resulting amine and 2-chloro-4-methylsulfonylbenzoyl chloride in the presence of an acid-binding agent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers transformative benefits that extend beyond simple chemistry. The primary advantage lies in the radical simplification of the raw material portfolio. By shifting from exotic organozinc or phosphorazidate reagents to commodity chemicals like 2-chloro-5-nitroacetophenone and haloacroleins, the supply chain becomes significantly more resilient. These starting materials are produced on a massive scale by the basic chemical industry, ensuring consistent availability and shielding manufacturers from the price volatility associated with specialized fine chemical reagents. This transition effectively de-risks the procurement strategy, guaranteeing a steady flow of materials necessary for continuous production schedules.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and hazardous reagents directly translates to substantial cost savings. Traditional routes often require stoichiometric amounts of costly zinc or boron species, along with expensive palladium catalysts for cross-coupling, which represent a significant portion of the COGS (Cost of Goods Sold). By replacing these with inexpensive organic bases and ammonia, the variable cost per kilogram of the intermediate is drastically reduced. Furthermore, the shorter reaction sequence reduces utility consumption (steam, cooling water) and labor hours, compounding the economic benefits and allowing for more competitive pricing in the global market.
- Enhanced Supply Chain Reliability: The robustness of the new process enhances overall supply chain reliability. Conventional methods involving sensitive organometallics are prone to batch failures due to moisture ingress or reagent degradation, leading to unpredictable lead times. In contrast, the aqueous ammonia condensation and standard reduction steps described in this patent are forgiving and reproducible. This operational stability ensures that delivery commitments can be met consistently, reducing the risk of stockouts for downstream API manufacturers. The ability to source raw materials from multiple global suppliers further diversifies the supply base, mitigating geopolitical or logistical disruptions.
- Scalability and Environmental Compliance: From a sustainability perspective, this route is exceptionally well-suited for commercial scale-up. The reduction in hazardous waste generation simplifies the permitting process and lowers the costs associated with waste disposal and treatment. The use of common solvents like dichloromethane and ethanol facilitates efficient solvent recovery systems, minimizing environmental impact. Additionally, the mild reaction conditions reduce the energy footprint of the manufacturing process. These factors collectively make the facility more compliant with increasingly strict environmental regulations, future-proofing the production asset against potential regulatory tightening.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Vismodegib synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer or capacity expansion.
Q: What are the primary advantages of this new Vismodegib synthesis route compared to prior art?
A: The novel route described in patent CN107200708B eliminates the need for expensive and unstable organozinc reagents or hazardous diphenyl phosphorazidate. It utilizes readily available starting materials like 2-chloro-5-nitroacetophenone and operates under mild reaction conditions, significantly reducing production costs and safety risks while achieving product purity up to 99.96%.
Q: How does this method address environmental concerns in API manufacturing?
A: By shortening the synthetic route to just three main steps and avoiding heavy metal catalysts like palladium in the coupling steps (using iron or recoverable Pd/C for reduction instead), the process drastically reduces the discharge of three wastes (wastewater, waste gas, solid waste). The use of common solvents like dichloromethane and ethanol further simplifies solvent recovery and recycling.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable. The reaction conditions are mild (0-50°C for the first step, moderate pressure for hydrogenation), and the raw materials are commercially abundant. The high yield (up to 91.5% in the final step) and excellent selectivity minimize side reactions, making it economically viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vismodegib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern pharmaceutical landscape. Our team of expert chemists has thoroughly analyzed the methodology disclosed in CN107200708B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art reactors capable of handling the specific temperature and pressure requirements of this novel route, ensuring that every batch meets stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify the absence of genotoxic impurities and residual metals, guaranteeing a product that is ready for immediate API synthesis.
We invite global partners to collaborate with us to leverage this advanced manufacturing capability. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain for this vital oncology intermediate is optimized for both performance and profitability.
