Advanced Catalytic Synthesis of Citral Intermediates for Industrial Scale-Up
Advanced Catalytic Synthesis of Citral Intermediates for Industrial Scale-Up
The global demand for high-purity citral, a critical compound in the flavor, fragrance, and pharmaceutical industries, necessitates robust and efficient synthetic routes. Patent CN112299962A introduces a groundbreaking methodology for synthesizing 3-methyl-2-butene-1-aldehyde diisopentenyl acetal, a pivotal precursor in citral production. This innovation leverages a novel supported chiral phosphoric acid catalyst system that fundamentally alters the economic and operational landscape of aldol condensation reactions. By transitioning from corrosive homogeneous liquid acids to a sophisticated heterogeneous solid catalyst, this technology addresses long-standing industry pain points regarding equipment longevity, waste management, and process scalability. For R&D directors and procurement managers seeking a reliable flavor & fragrance intermediate supplier, understanding the mechanistic depth and commercial viability of this patent is essential for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 3-methyl-2-butene-1-aldehyde diisopentenyl acetal has relied heavily on homogeneous acid catalysts such as nitric acid, phosphoric acid, or lithium chloride. While these traditional methods can achieve reasonable conversion rates, they are plagued by severe inherent defects that compromise operational efficiency and safety. The use of strong mineral acids creates a highly corrosive environment that aggressively attacks stainless steel reactors and piping, leading to frequent equipment failure, costly maintenance schedules, and potential safety hazards associated with acid leaks. Furthermore, the post-reaction processing is cumbersome; the acidic catalyst must be neutralized using bases like potassium acetate or sodium carbonate, generating substantial quantities of saline wastewater that require expensive treatment before discharge. Additionally, separating the catalyst from the product mixture often involves complex distillation under negative pressure, which increases energy consumption and poses risks of thermal degradation to the sensitive unsaturated intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN112299962A utilizes a supported chiral phosphoric acid (CPA) catalyst, which operates as a heterogeneous system. This approach effectively decouples the catalytic activity from the bulk reaction medium, allowing the reaction to proceed under much milder and controlled conditions. The solid catalyst can be easily separated from the liquid product stream via simple filtration or by utilizing a fixed-bed reactor configuration, completely eliminating the need for neutralization and the associated salt waste. 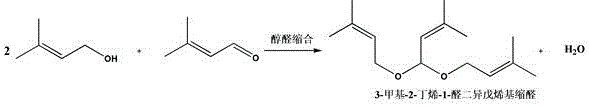 This shift not only streamlines the workflow but also dramatically enhances the purity of the crude product by avoiding side reactions typically induced by strong liquid acids. The ability to operate in a continuous flow mode using a fixed bed further underscores the industrial readiness of this novel approach, offering a pathway to consistent, high-volume production that legacy batch processes cannot match.
This shift not only streamlines the workflow but also dramatically enhances the purity of the crude product by avoiding side reactions typically induced by strong liquid acids. The ability to operate in a continuous flow mode using a fixed bed further underscores the industrial readiness of this novel approach, offering a pathway to consistent, high-volume production that legacy batch processes cannot match.
Mechanistic Insights into Supported Chiral Phosphoric Acid Catalysis
The core of this technological advancement lies in the intricate design of the catalyst, which comprises a porous carrier, a chiral phosphoric acid active component, and a polyethylene glycol (PEG) modifier. The carrier, selected from advanced materials such as hydrotalcite (Fe-LDH), molecular sieves (HZSM-5), or Metal-Organic Frameworks (HZIF-8), provides a high-surface-area scaffold with Lewis acidity. This structure creates a unique three-dimensional microenvironment where reactant molecules—isoamylene alcohol and isoamylene aldehyde—are adsorbed and concentrated within the pore channels. The chiral phosphoric acid, specifically the (R)-CPA or (S)-CPA enantiomers shown below, acts as a Brønsted acid site. 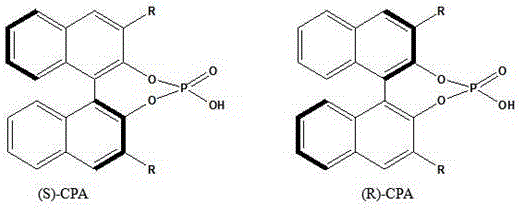 Through hydrogen bonding interactions, the CPA activates the carbonyl group of the aldehyde while simultaneously interacting with the hydroxyl group of the alcohol. This dual activation lowers the activation energy barrier for the aldol condensation, facilitating the formation of the acetal bond with exceptional stereochemical control and regioselectivity.
Through hydrogen bonding interactions, the CPA activates the carbonyl group of the aldehyde while simultaneously interacting with the hydroxyl group of the alcohol. This dual activation lowers the activation energy barrier for the aldol condensation, facilitating the formation of the acetal bond with exceptional stereochemical control and regioselectivity.
Crucially, the inclusion of the PEG modifier serves a stabilizing function that is often overlooked in conventional catalyst design. The PEG chains uniformly coat the carrier surface and form hydrogen bonds with the chiral phosphoric acid molecules, effectively anchoring them to the support. This prevents the leaching of the expensive chiral active component into the reaction mixture, ensuring that the catalyst retains its activity over multiple cycles. The synergistic effect between the Lewis acidic carrier, the Brønsted acidic CPA, and the stabilizing PEG modifier results in a catalytic system that exhibits superior resistance to deactivation and carbon deposition. This mechanistic robustness translates directly to higher yields, often exceeding 90%, and maintains high selectivity even after prolonged operation, making it an ideal solution for cost reduction in synthetic flavors manufacturing.
How to Synthesize 3-methyl-2-butene-1-aldehyde diisopentenyl acetal Efficiently
The synthesis protocol outlined in the patent offers a reproducible pathway for producing high-purity citral intermediates suitable for downstream pharmaceutical and fragrance applications. The process begins with the meticulous preparation of the supported catalyst, involving the pretreatment of the carrier to remove moisture, followed by impregnation with the PEG modifier and the chiral phosphoric acid active species. Once the catalyst is prepared and loaded into a reactor, the aldol condensation proceeds by feeding a mixture of isoamylene alcohol and isoamylene aldehyde. The detailed standardized synthesis steps, including specific molar ratios, temperature gradients, and residence times required to achieve optimal conversion and selectivity, are provided in the technical guide below.
- Pretreat the selected carrier (e.g., HZIF-8, Fe-LDH) by drying at 70-80°C for 10 hours to remove moisture, followed by swelling in toluene.
- Disperse the pretreated carrier in a polyethylene glycol (PEG) modifier at 70-80°C, then add the chiral phosphoric acid (CPA) active component and maintain temperature for 4-8 hours.
- Filter and dry the catalyst precursor at 80°C, then load into a fixed-bed reactor to catalyze the aldol condensation of isoamylene alcohol and isoamylene aldehyde at 80-90°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this supported catalyst technology represents a strategic opportunity to optimize operational expenditures and mitigate supply risks. The transition from corrosive liquid acids to a reusable solid catalyst fundamentally changes the cost structure of production. By eliminating the need for expensive corrosion-resistant alloys in reactors and removing the neutralization and salt disposal steps, manufacturers can achieve substantial cost savings in both capital expenditure (CapEx) and operational expenditure (OpEx). The qualitative improvement in process safety and environmental compliance further reduces the regulatory burden and potential liability associated with handling hazardous mineral acids.
- Cost Reduction in Manufacturing: The heterogeneous nature of the catalyst allows for straightforward recovery and regeneration. Unlike single-use liquid acids that become part of the waste stream, this supported catalyst can be reused multiple times with minimal loss of activity, as demonstrated by stability tests showing consistent performance over thousands of hours. This reusability drastically reduces the recurring cost of catalyst consumption. Furthermore, the high selectivity of the reaction minimizes the formation of by-products, which simplifies downstream purification and increases the overall yield of the valuable acetal intermediate, thereby maximizing the return on raw material investment.
- Enhanced Supply Chain Reliability: The compatibility of this catalyst with continuous flow chemistry, specifically in fixed-bed reactors, offers a significant advantage for supply continuity. Continuous processes are inherently more stable and easier to scale than batch processes, reducing the variability in product quality and lead time. The robustness of the catalyst against deactivation means that production runs can be extended without frequent shutdowns for catalyst replacement or reactor cleaning. This reliability ensures a steady flow of intermediates to downstream customers, mitigating the risk of stockouts and enabling more accurate demand forecasting and inventory management.
- Scalability and Environmental Compliance: As global regulations on industrial emissions tighten, the environmental profile of a chemical process becomes a critical factor for long-term viability. This technology generates significantly less hazardous waste compared to traditional methods, as it avoids the generation of acidic wastewater and salt by-products. The simplified workup procedure reduces the energy intensity of the process, contributing to a lower carbon footprint. These factors make the technology highly scalable, allowing manufacturers to expand capacity from pilot scales to hundreds of tons annually without encountering the bottlenecks associated with waste treatment infrastructure or equipment corrosion limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this supported chiral phosphoric acid catalyst system. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: What are the primary advantages of the supported CPA catalyst over traditional mineral acids?
A: Unlike traditional nitric or phosphoric acid catalysts which cause severe equipment corrosion and generate difficult-to-treat acidic wastewater, the supported chiral phosphoric acid catalyst is heterogeneous. This allows for easy separation, repeated reuse without significant activity loss, and eliminates the need for neutralization steps, thereby drastically reducing three-waste treatment costs.
Q: Can this synthesis method be adapted for continuous flow production?
A: Yes, the patent explicitly demonstrates the efficacy of this catalyst in a fixed-bed reactor setup. The solid nature of the supported catalyst makes it ideal for continuous processing, allowing for stable operation over extended periods (demonstrated up to 3000 hours) with simple regeneration protocols involving toluene and ethanol washing.
Q: How does the PEG modifier influence the catalytic performance?
A: The polyethylene glycol (PEG) modifier acts as a crucial stabilizer. It uniformly covers the carrier surface and forms hydrogen bonds with the active chiral phosphoric acid component. This interaction prevents the leaching of the active ingredient, enhances the dispersion of catalytic sites within the pore channels, and significantly improves the selectivity and stability of the catalyst during the aldol condensation reaction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-methyl-2-butene-1-aldehyde diisopentenyl acetal Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products. Our team of expert chemists has extensively analyzed the potential of the supported catalyst technology described in CN112299962A and is fully equipped to leverage these advancements for your specific needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are designed to handle complex catalytic systems with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards.
We invite you to collaborate with us to explore how this innovative synthesis route can enhance your supply chain efficiency and product quality. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technological excellence can drive value for your organization.
