Advanced Catalytic Synthesis of Citral Intermediates for Industrial Scale Flavor Manufacturing
Advanced Catalytic Synthesis of Citral Intermediates for Industrial Scale Flavor Manufacturing
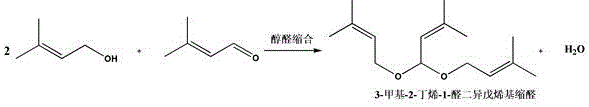
The global demand for high-purity fragrance and flavor intermediates continues to drive innovation in organic synthesis, particularly for key precursors like citral. A groundbreaking technical disclosure found in patent CN112299962B introduces a transformative method for synthesizing 3-methyl-2-butene-1-aldehyde diisopentenyl acetal, a critical intermediate in the production of citral. This patent details a sophisticated catalytic system utilizing supported chiral phosphoric acid, which fundamentally shifts the paradigm from corrosive, homogeneous acid catalysis to a robust, heterogeneous process. For R&D directors and technical leaders in the fine chemical sector, this represents a significant leap forward in process efficiency, offering a pathway to achieve conversion rates exceeding 98% while drastically reducing environmental impact. The technology addresses long-standing industry pain points regarding equipment corrosion and waste generation, positioning it as a cornerstone for next-generation sustainable manufacturing of complex fragrance molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 3-methyl-2-butene-1-aldehyde diisopentenyl acetal has relied heavily on the aldol condensation of isoamylene alcohol and isoamylene aldehyde using traditional inorganic or organic acids such as nitric acid, phosphoric acid, or lithium chloride. These conventional methodologies are fraught with significant operational and economic drawbacks that hinder efficient large-scale production. The use of strong mineral acids creates a highly corrosive environment that severely degrades reactor lifespan, necessitating expensive corrosion-resistant materials and frequent maintenance shutdowns. Furthermore, the homogeneous nature of these catalysts makes separation incredibly difficult, often requiring complex neutralization steps with bases like potassium acetate or sodium carbonate, which subsequently generates substantial volumes of saline wastewater. Prior art, such as US4133836A and US5177265A, highlights yields that often struggle to exceed 75-83%, with significant side reactions complicating downstream purification. The inability to easily recover and reuse these liquid acids results in a linear consumption of reagents, driving up raw material costs and creating a heavy burden on waste treatment facilities.
The Novel Approach
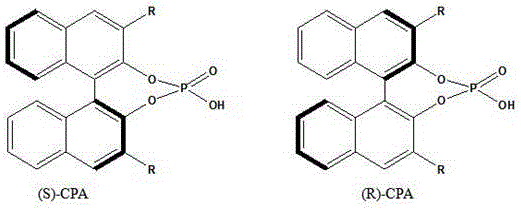
In stark contrast, the novel approach disclosed in CN112299962B leverages a heterogenized catalytic system where chiral phosphoric acid (CPA) is immobilized on advanced porous carriers such as metal-organic frameworks (MOFs), zeolites, or hydrotalcites. This structural innovation transforms the reaction landscape by combining the high selectivity of organocatalysis with the practical handling benefits of solid catalysts. By anchoring the active chiral phosphoric acid species onto carriers like HZIF-8 or Fe-LDH, and critically modifying the surface with polyethylene glycol (PEG), the process achieves a synergistic effect that boosts selectivity to over 99%. This method eliminates the need for corrosive liquid acids, thereby preserving equipment integrity and simplifying the workup procedure to a mere filtration or continuous flow-through. The reaction proceeds smoothly under mild vacuum conditions (1.0 KPa) and moderate temperatures (80-90°C), delivering yields as high as 98.02% in batch tests and demonstrating exceptional stability in continuous fixed-bed operations. This shift not only enhances product purity but also aligns perfectly with green chemistry principles by minimizing waste and energy consumption.
Mechanistic Insights into Supported Chiral Phosphoric Acid Catalysis
The superior performance of this catalytic system stems from a meticulously engineered triple-component architecture comprising the carrier, the active chiral phosphoric acid, and the PEG modifier. The carrier, whether it be a molecular sieve like HZSM-5 or a MOF like HKUST-1, provides a high-surface-area scaffold with inherent Lewis acidity that pre-activates the reactant molecules. Within the pore channels of these carriers, the isoamylene aldehyde molecules are adsorbed and activated through hydrogen bonding interactions with the immobilized chiral phosphoric acid. Simultaneously, the chiral phosphoric acid donates protons to form hydrogen bonds with the isoamylene alcohol, effectively lowering the activation energy for the nucleophilic attack. This dual-activation mechanism ensures that the reaction proceeds with high regioselectivity and stereoselectivity, minimizing the formation of unwanted oligomers or ethers that typically plague acid-catalyzed condensations. The spatial confinement within the carrier pores further restricts the transition state geometry, favoring the formation of the desired acetal product over competing side reactions.
A critical, often overlooked component of this system is the polyethylene glycol (PEG) modifier, which plays a pivotal role in stabilizing the active sites and preventing catalyst deactivation. Without the modifier, the chiral phosphoric acid might leach out of the carrier pores or aggregate, leading to rapid loss of activity. The PEG chains uniformly coat the carrier surface and intercalate with the active acid species through hydrogen bonding, effectively locking them in place while maintaining their accessibility to substrates. This modification creates a microenvironment that enhances the hydrophobicity around the active sites, facilitating the removal of the water byproduct generated during condensation and shifting the equilibrium towards product formation. The result is a catalyst that not only delivers initial high yields but maintains its structural integrity and activity over extended periods, as evidenced by stability tests showing minimal degradation even after thousands of hours of operation. This mechanistic robustness is key to ensuring consistent product quality in a commercial setting.
How to Synthesize 3-Methyl-2-Butene-1-Aldehyde Diisopentenyl Acetal Efficiently
The synthesis protocol outlined in the patent offers a clear roadmap for implementing this technology in both batch and continuous flow settings, providing flexibility for different production scales. The process begins with the rigorous pretreatment of the chosen carrier to ensure all moisture is removed, followed by a swelling step in toluene to open up the pore structure for maximum loading capacity. The subsequent impregnation with the PEG modifier and chiral phosphoric acid requires precise temperature control to ensure uniform distribution of the active species. Once the catalyst is prepared and dried to constant weight, it is packed into a fixed-bed reactor or added to a stirred tank for batch processing. The reactants, isoamylene alcohol and isoamylene aldehyde, are mixed in a molar ratio ranging from 1:2.0 to 1:2.5 and fed into the reactor system. Detailed standard operating procedures for catalyst preparation and reaction conditions are essential for replicating the high yields reported in the patent data.
- Pretreat the selected carrier (e.g., HZIF-8, Fe-LDH) by drying at 70-80°C under vacuum to remove moisture, followed by swelling in toluene.
- Disperse the pretreated carrier in a polyethylene glycol (PEG) modifier, then add the chiral phosphoric acid active component and heat to 80°C for immobilization.
- Conduct the aldol condensation of isoamylene alcohol and isoamylene aldehyde in a fixed-bed reactor at 80-90°C under vacuum (1.0 KPa) using the prepared catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this supported catalyst technology translates directly into tangible operational efficiencies and risk mitigation strategies. The shift from corrosive liquid acids to a solid, reusable catalyst fundamentally alters the cost structure of manufacturing this key intermediate. By eliminating the need for expensive corrosion-resistant alloys in reactors and piping, capital expenditure for new production lines can be significantly optimized, while existing carbon steel infrastructure can potentially be utilized with reduced maintenance overheads. Furthermore, the removal of the neutralization and salt-separation steps drastically simplifies the downstream processing workflow, reducing the consumption of auxiliary chemicals and lowering the volume of hazardous waste that requires disposal. This streamlining of the process flow leads to shorter batch cycle times and higher throughput, allowing manufacturers to respond more agilely to market demand fluctuations without compromising on product quality or safety standards.
- Cost Reduction in Manufacturing: The implementation of this heterogeneous catalytic system drives down production costs through multiple mechanisms, primarily by enabling the repeated reuse of the catalyst over many cycles without significant loss of activity. Unlike traditional homogeneous acids that are consumed or neutralized in every batch, this supported catalyst can be regenerated and reused, effectively amortizing the cost of the sophisticated chiral phosphoric acid over a much larger volume of product. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, which reduces the load on distillation columns and lowers energy consumption associated with purification. The elimination of wastewater treatment costs associated with neutralizing strong mineral acids further contributes to substantial overall cost savings, making the final product more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and stability of the catalyst, which ensures consistent production output over long durations. The patent data indicates that the catalyst maintains high performance even after 3000 hours of continuous operation in a fixed-bed reactor, reducing the frequency of reactor shutdowns for catalyst replacement or regeneration. This reliability minimizes the risk of production bottlenecks and ensures a steady flow of intermediates to downstream customers. Moreover, the raw materials required for the catalyst synthesis, such as common molecular sieves and polyethylene glycol, are readily available commodities, reducing dependency on scarce or geopolitically sensitive reagents. This stability in raw material sourcing combined with the durability of the catalyst creates a more predictable and secure supply chain for high-purity fragrance intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this technology offers a clear path to compliance with increasingly stringent emission standards. The process generates significantly less hazardous waste compared to traditional methods, as there are no spent acid streams or large quantities of salt byproducts to manage. The ability to operate in a continuous fixed-bed mode facilitates easier containment of volatile organic compounds (VOCs) and allows for more efficient energy integration, further reducing the carbon footprint of the manufacturing process. This environmental advantage not only mitigates regulatory risk but also enhances the brand value of the end products by aligning with the sustainability goals of major multinational consumers in the flavor and fragrance industry. The scalability of the fixed-bed reactor design ensures that production can be ramped up from pilot scale to multi-ton commercial volumes with minimal re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced catalytic synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, aiming to clarify the operational benefits and feasibility for industrial partners. Understanding these nuances is critical for R&D teams evaluating the technology for process transfer and for procurement teams assessing the long-term value proposition of switching to this greener synthetic route.
Q: What are the primary advantages of using supported chiral phosphoric acid catalysts over traditional mineral acids?
A: Unlike traditional nitric or phosphoric acid catalysts which cause severe equipment corrosion and generate difficult-to-treat acidic wastewater, supported chiral phosphoric acid catalysts are heterogeneous. This allows for easy separation via filtration or fixed-bed flow, eliminates the need for neutralization steps, prevents equipment corrosion, and enables catalyst recycling for over 10 cycles with minimal activity loss.
Q: How does the addition of polyethylene glycol (PEG) modifiers impact catalyst performance?
A: The PEG modifier acts as a crucial stabilizing agent that uniformly covers the carrier surface. It forms hydrogen bonds with the chiral phosphoric acid active components, preventing their leaching or loss during the reaction. This synergistic interaction significantly enhances the selectivity of the aldol condensation reaction, pushing yields above 98% compared to roughly 61% yield observed with unsupported homogeneous catalysts.
Q: Is this synthesis method suitable for continuous industrial production?
A: Yes, the technology is specifically designed for scalability. The patent demonstrates successful operation in a fixed-bed reactor system where reactants are pumped continuously through the catalyst bed. The catalyst exhibits exceptional thermal and mechanical stability, maintaining high conversion rates even after 3000 hours of continuous operation, making it ideal for large-scale commercial manufacturing of fragrance intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methyl-2-Butene-1-Aldehyde Diisopentenyl Acetal Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of mastering complex catalytic transformations to deliver high-value intermediates for the global flavor and pharmaceutical markets. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the supported chiral phosphoric acid catalysis described in CN112299962B can be seamlessly translated into robust manufacturing processes. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 3-methyl-2-butene-1-aldehyde diisopentenyl acetal meets the exacting standards required for citral synthesis. Our facility is equipped to handle the specific engineering requirements of fixed-bed continuous reactions, allowing us to offer a stable and cost-effective supply of this critical intermediate to our international partners.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this catalytic route can optimize your total cost of ownership. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples to validate the superior quality of our commercially produced intermediates. Let us partner with you to drive efficiency and sustainability in your fragrance and flavor manufacturing operations.
