Revolutionizing Axial Chirality: Scalable Synthesis of Indole Carbazole Derivatives via Rh(II) Catalysis
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex chiral scaffolds, particularly those exhibiting axial chirality, which are pivotal in the development of next-generation therapeutics and advanced organic optoelectronic materials. Patent CN113087714B introduces a groundbreaking advancement in this domain by disclosing a novel class of axial chiral aryl indole carbazole derivatives and a highly efficient preparation method thereof. This technology leverages a sophisticated Rh(II)-catalyzed intermolecular N-H insertion reaction to selectively construct the challenging C-N axial chirality. Unlike conventional approaches that often struggle with regioselectivity and stereocontrol, this invention provides a pathway to achieve high yields and exceptional enantioselectivity, thereby establishing a critical research foundation for late-stage functionalization, novel drug development, and the creation of advanced phosphoric acid catalysts. The structural versatility of these derivatives, as depicted in the general formula, allows for extensive modification to tune electronic and steric properties for specific applications.
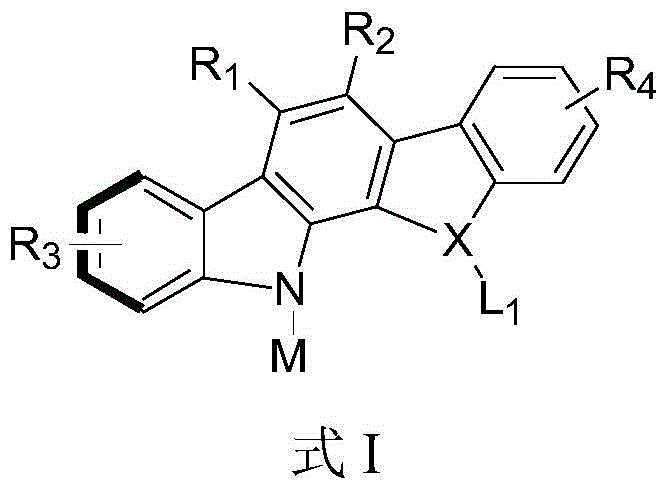
For R&D directors and process chemists, understanding the limitations of existing technologies is crucial for evaluating new synthetic routes. Conventional methods for constructing C-N axial chirality, such as transition metal-catalyzed cross-coupling reactions like Ullmann or Buchwald-Hartwig aminations, have long been the benchmark. However, these traditional schemes often suffer from significant drawbacks, including the requirement for harsh reaction conditions, the necessity of pre-activated substrates like aryl halides, and frequently poor control over atroposelectivity. The inherent low rotational barrier of the C-N bond combined with the competitive reactivity of C-H bonds makes achieving high regional and enantiomer control exceptionally difficult using these older methods. Furthermore, photoinduced radical couplings, while innovative, often result in racemic mixtures that require tedious resolution steps, drastically reducing overall process efficiency and increasing waste generation in a manufacturing setting.
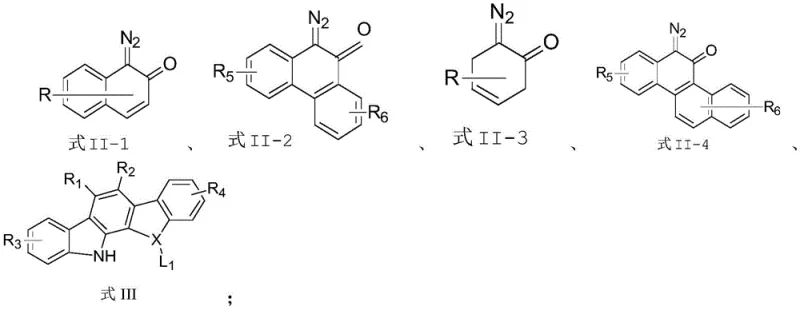
In sharp contrast, the novel approach detailed in this patent utilizes a carbene insertion strategy that fundamentally changes the reaction landscape. By employing diazonaphthoquinone derivatives as carbene precursors, the method generates reactive intermediates in situ that can distinguish between different C-H and N-H bonds with high precision. This strategy bypasses the need for pre-functionalized halides and avoids the high thermal energy inputs typically associated with C-N bond formation. The use of a chiral dirhodium tetracarboxylate catalyst, specifically Rh2(S-PTAD)4, enables the direct and selective insertion into the N-H bond of indolo[2,3-a]carbazoles. This not only simplifies the substrate scope but also ensures that the axial chirality is induced directly during the bond-forming event, leading to products with superior optical purity without the need for downstream resolution.
Mechanistic Insights into Rh(II)-Catalyzed Asymmetric N-H Insertion
The core of this technological breakthrough lies in the unique mechanistic pathway facilitated by the chiral rhodium catalyst. The reaction initiates with the coordination of the diazo compound to the dirhodium center, followed by the extrusion of nitrogen gas to form a highly reactive rhodium-carbenoid species. This electrophilic carbenoid is then attacked by the nucleophilic nitrogen atom of the indolocarbazole substrate. The chiral environment provided by the bulky adamantyl and phthalimido ligands on the rhodium catalyst creates a sterically confined pocket that dictates the trajectory of the incoming nucleophile. This steric guidance is what translates point chirality from the catalyst into axial chirality in the final product, effectively locking the rotation around the newly formed C-N bond. The result is a highly stereoselective process that can achieve enantiomeric excess values exceeding 98% in many cases, as demonstrated by the synthesis of various derivatives.
Furthermore, the mechanism offers distinct advantages regarding impurity control and byproduct management. Traditional coupling methods often generate stoichiometric amounts of salt waste or require toxic tin or boron reagents. In this Rh(II)-catalyzed insertion, the only byproduct is nitrogen gas, which harmlessly vents from the reaction system. This "atom economical" aspect significantly simplifies the workup procedure and reduces the burden on wastewater treatment facilities. The high regioselectivity of the carbene insertion also minimizes the formation of positional isomers, which are notoriously difficult to separate in chiral chromatography. By suppressing side reactions such as C-H insertion or dimerization of the diazo compound through careful catalyst tuning, the process ensures a clean impurity profile, which is a critical parameter for regulatory approval in pharmaceutical manufacturing.
How to Synthesize Axial Chiral Aryl Indole Carbazole Derivatives Efficiently
The practical implementation of this synthesis route is designed for operational simplicity and robustness, making it highly attractive for scale-up operations. The standard protocol involves dissolving the diazonaphthoquinone derivative and the indolocarbazole substrate in a chlorinated solvent like chloroform, although toluene can also be employed depending on solubility requirements. The reaction is initiated by adding a catalytic amount of Rh2(S-PTAD)4, typically around 2 mol%, under an inert atmosphere to prevent catalyst deactivation. The mixture is then stirred at ambient temperature, eliminating the need for energy-intensive heating or cooling cycles. Detailed standardized synthesis steps, including specific molar ratios, quenching procedures, and purification methods, are outlined in the technical guide below.
- Prepare the reaction mixture by dissolving the diazonaphthoquinone derivative (Formula II) and the indolo[2,3-a]carbazole derivative (Formula III) in a suitable solvent such as chloroform or toluene.
- Add the chiral rhodium catalyst, specifically Rh2(S-PTAD)4, at a loading of approximately 2 mol% relative to the diazo compound under an inert atmosphere.
- Stir the reaction mixture at room temperature for 6 to 18 hours, then remove the solvent under reduced pressure to isolate the crude axial chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers substantial strategic benefits that translate directly into cost optimization and risk mitigation. The shift from multi-step, harsh coupling sequences to a direct catalytic insertion significantly streamlines the manufacturing process. By reducing the number of unit operations and eliminating the need for expensive pre-activated starting materials, the overall cost of goods sold (COGS) can be drastically reduced. The mild reaction conditions also mean that standard glass-lined or stainless steel reactors can be used without requiring specialized high-pressure or high-temperature equipment, lowering capital expenditure requirements for production facilities.
- Cost Reduction in Manufacturing: The elimination of stoichiometric coupling reagents and the use of low catalyst loadings contribute to significant raw material savings. Additionally, the high selectivity of the reaction reduces the need for complex and costly chiral separation processes, which are often the most expensive step in producing single-enantiomer APIs. The simplified workup procedure further lowers labor and utility costs associated with solvent recovery and waste disposal.
- Enhanced Supply Chain Reliability: The substrates required for this reaction, such as diazonaphthoquinones and indolocarbazoles, are becoming increasingly accessible through established supply chains. The robustness of the catalytic system ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed reactions or off-spec material. This reliability is crucial for maintaining continuous supply to downstream customers in the pharmaceutical and electronics sectors.
- Scalability and Environmental Compliance: The reaction's ability to proceed efficiently at room temperature and generate benign nitrogen gas as the only byproduct aligns perfectly with green chemistry principles. This facilitates easier regulatory approval and environmental compliance, avoiding the complexities associated with heavy metal waste streams common in traditional palladium or copper-catalyzed couplings. The process has been demonstrated to be scalable, providing a clear path from gram-scale laboratory synthesis to multi-kilogram commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this axial chiral synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What is the primary advantage of using Rh2(S-PTAD)4 over traditional coupling methods for C-N axis construction?
A: Unlike traditional Ullmann or Buchwald-Hartwig couplings which often require harsh conditions and struggle with atroposelectivity, the Rh2(S-PTAD)4 catalyzed N-H insertion operates under mild room temperature conditions and achieves excellent enantioselectivity (up to 99% ee) by leveraging carbene intermediate stereoselectivity.
Q: Can this synthesis method be applied to bioactive natural product derivatives?
A: Yes, the patent demonstrates successful late-stage functionalization of bioactive molecules such as Tjipanazole I, yielding axially chiral derivatives with high enantiomeric excess, proving its utility in drug discovery and modification.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction typically proceeds in chloroform or toluene at room temperature using a catalyst loading of 1-3 mol%, with reaction times ranging from 6 to 18 hours, making it highly energy-efficient compared to high-temperature coupling protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Aryl Indole Carbazole Derivative Supplier
As the demand for complex chiral intermediates continues to grow in the fields of oncology and organic electronics, having a manufacturing partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from pilot plant to full-scale manufacturing. Our stringent purity specifications and rigorous QC labs guarantee that every batch of axial chiral aryl indole carbazole derivatives meets the highest international standards, minimizing risks in your downstream synthesis.
We invite you to collaborate with us to leverage this cutting-edge Rh(II)-catalyzed technology for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project volume and purity requirements. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you accelerate your time to market with superior chiral building blocks.
