Revolutionizing Axial Chiral Indolocarbazole Synthesis with Rh(II) Catalysis for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex chiral scaffolds, particularly those exhibiting axial chirality, which are pivotal in modern drug design and material science. Patent CN113087714B introduces a groundbreaking approach to synthesizing axial chiral aryl indole carbazole derivatives through a highly selective Rh(II)-catalyzed intermolecular N-H insertion reaction. This technology addresses the long-standing challenge of constructing C-N axial chirality with high enantioselectivity and yield, overcoming the limitations of traditional cross-coupling methods that often struggle with regioselectivity and harsh reaction conditions. The disclosed method utilizes specialized rhodium catalysts to facilitate the direct insertion of carbene intermediates into N-H bonds, providing a streamlined pathway to valuable indolocarbazole frameworks found in potent anticancer and antifungal agents.
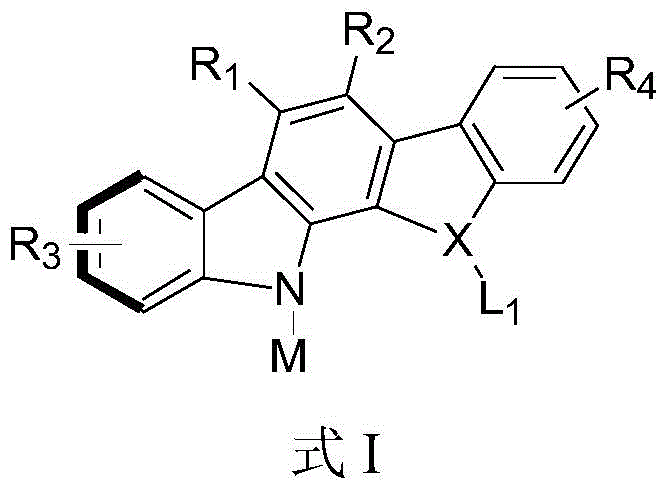
For R&D directors and process chemists, the significance of this invention lies in its ability to generate structurally diverse libraries of chiral molecules under mild conditions. The general formula I encompasses a wide range of substituents, allowing for extensive late-stage functionalization which is crucial for optimizing biological activity in drug discovery programs. By leveraging this patented technology, manufacturers can access high-purity intermediates that were previously difficult to synthesize with such precise stereochemical control, thereby accelerating the development of novel therapeutic candidates and advanced organic optoelectronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral biaryl and heteroaryl systems containing a C-N axis has relied heavily on transition metal-catalyzed cross-coupling reactions such as the Ullmann and Buchwald-Hartwig aminations. While these methods are versatile, they frequently suffer from significant drawbacks when applied to the synthesis of sterically hindered N-aryl indoles or carbazoles. The primary obstacle is the inherent instability of the C-N bond rotation barrier, which often necessitates extremely harsh reaction conditions, including high temperatures and strong bases, to achieve coupling. Furthermore, controlling the stereochemistry to obtain a single atropisomer is exceptionally challenging using these classical routes, often resulting in racemic mixtures that require difficult and costly resolution steps. The site selectivity is also a major concern, as competitive insertion into more reactive C-H bonds can lead to complex mixtures of regioisomers, drastically reducing the overall efficiency and purity of the desired product.
The Novel Approach
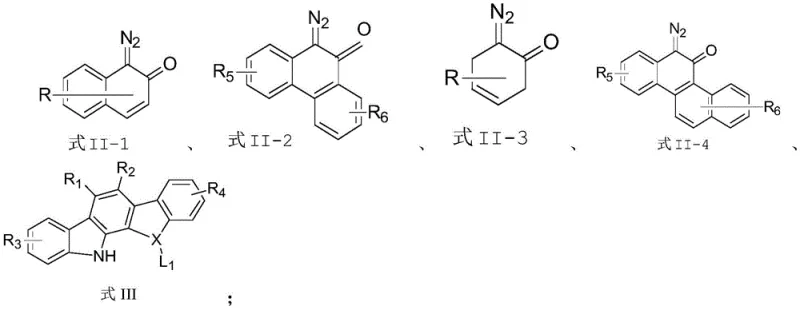
In sharp contrast, the methodology described in CN113087714B utilizes a carbene insertion strategy catalyzed by chiral dirhodium complexes, specifically Rh2(S-PTAD)4, to achieve unprecedented levels of stereocontrol. This novel approach bypasses the need for pre-activated halides and harsh coupling conditions by generating reactive carbene intermediates in situ from diazo compounds. These intermediates selectively insert into the N-H bond of the indolocarbazole scaffold, driven by the chiral environment of the rhodium catalyst. This results in the direct formation of the C-N axial bond with excellent enantioselectivity, often exceeding 98% ee, and high chemical yields. The reaction proceeds efficiently at room temperature in common solvents like chloroform, representing a paradigm shift towards greener and more efficient synthetic protocols for complex chiral architectures.
Mechanistic Insights into Rh(II)-Catalyzed N-H Insertion
The core of this technological advancement is the sophisticated catalytic cycle mediated by the dirhodium tetracarboxylate catalyst. Upon decomposition of the diazonaphthoquinone precursor, the rhodium catalyst generates a metal-carbenoid species that is highly electrophilic yet stereochemically defined by the bulky adamantyl and phthalimido ligands of the S-PTAD framework. This chiral carbenoid approaches the nucleophilic nitrogen of the indolocarbazole substrate within a tightly constrained chiral pocket. The steric bulk of the catalyst effectively differentiates between the two faces of the approaching nucleophile and dictates the rotational barrier of the forming C-N bond, locking the molecule into a specific atropisomeric configuration. This mechanism ensures that the newly formed axial chirality is established with high fidelity during the bond-forming event itself, rather than relying on thermodynamic equilibration or kinetic resolution post-synthesis.
Furthermore, the impurity profile of this reaction is remarkably clean due to the high specificity of the carbene insertion. Unlike radical-based couplings that can generate numerous side products through homocoupling or hydrogen abstraction, this ionic-like insertion pathway minimizes byproduct formation. The use of mild conditions also preserves sensitive functional groups on the substrate, such as esters, halogens, or hydroxyl groups, which might otherwise decompose under the rigorous conditions of traditional coupling. This functional group tolerance is critical for pharmaceutical applications where complex molecular architectures must be maintained. The result is a process that not only delivers high enantiomeric excess but also simplifies downstream purification, significantly reducing the burden on process development teams aiming for commercial viability.
How to Synthesize Axial Chiral Aryl Indole Carbazole Efficiently
The synthesis protocol outlined in the patent provides a robust and reproducible method for generating these high-value chiral intermediates. The process involves the cross-coupling of a diazonaphthoquinone derivative with an indolo[2,3-a]carbazole derivative in the presence of a chiral rhodium catalyst. The reaction is typically conducted in chloroform at room temperature, requiring only a catalytic loading of 2 mol% Rh2(S-PTAD)4. This simplicity belies the sophistication of the transformation, which achieves gram-scale synthesis with high conversion rates. For detailed operational parameters and safety guidelines regarding the handling of diazo compounds, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by dissolving diazonaphthoquinone derivatives and indolo[2,3-a]carbazole derivatives in chloroform or toluene.
- Add the chiral catalyst Rh2(S-PTAD)4 (2 mol%) to the solution under an inert atmosphere at room temperature.
- Stir the reaction for 6 to 18 hours, then remove the solvent to isolate the high-purity chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this Rh(II)-catalyzed synthesis offers substantial strategic advantages over legacy manufacturing routes. The ability to produce complex chiral intermediates in fewer steps with higher purity directly translates to reduced manufacturing costs and improved supply security. By eliminating the need for expensive palladium catalysts and harsh reagents associated with traditional cross-coupling, the overall cost of goods sold (COGS) can be significantly optimized. Moreover, the mild reaction conditions reduce energy consumption and minimize the generation of hazardous waste, aligning with increasingly stringent environmental regulations and sustainability goals that global supply chains must adhere to.
- Cost Reduction in Manufacturing: The elimination of expensive transition metals like palladium and the avoidance of high-temperature processing significantly lowers the operational expenditure associated with production. The high enantioselectivity achieved directly means that there is no need for costly chiral resolution steps or recycling of unwanted enantiomers, which traditionally consume a large portion of the budget in chiral synthesis. Additionally, the high atom economy of the insertion reaction ensures that raw materials are utilized more efficiently, further driving down the cost per kilogram of the final active pharmaceutical ingredient or intermediate.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system allows for consistent batch-to-batch reproducibility, which is a critical factor for maintaining a stable supply of key intermediates. The use of readily available solvents like chloroform and the stability of the catalyst under ambient conditions simplify logistics and storage requirements. This reliability reduces the risk of production delays caused by sensitive reaction parameters, ensuring that downstream customers receive their materials on schedule. The scalability demonstrated in the patent, moving from milligram to gram scales without loss of selectivity, indicates a clear path to multi-kilogram commercial production.
- Scalability and Environmental Compliance: The process operates at room temperature, which drastically reduces the energy footprint compared to thermal coupling reactions that require heating to over 100°C. Lower energy usage not only cuts costs but also simplifies the engineering requirements for large-scale reactors. Furthermore, the high selectivity reduces the formation of toxic byproducts, easing the burden on waste treatment facilities and ensuring compliance with green chemistry principles. This makes the technology highly attractive for manufacturers looking to future-proof their operations against tightening environmental regulations while maintaining high throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this axial chiral synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of this method in industrial settings.
Q: What is the primary advantage of this Rh(II) catalyzed method over traditional Ullmann coupling?
A: Unlike traditional Ullmann or Buchwald-Hartwig couplings which often require harsh conditions and yield racemic mixtures, this Rh(II) catalyzed N-H insertion operates at room temperature with excellent enantioselectivity (up to 99% ee), directly constructing the C-N axial chirality without pre-activation.
Q: Can this synthesis method be applied to complex bioactive molecules?
A: Yes, the patent demonstrates successful late-stage functionalization of complex bioactive scaffolds such as Tipranazole I derivatives, achieving high yields and enantioselectivity, proving its utility for drug development.
Q: What are the typical reaction conditions for this synthesis?
A: The reaction typically proceeds in chloroform or toluene at room temperature using 2 mol% of Rh2(S-PTAD)4 catalyst, with reaction times ranging from 6 to 18 hours depending on the substrate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Aryl Indole Carbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Rh(II)-catalyzed technology in advancing the synthesis of complex pharmaceutical intermediates and functional materials. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market readiness is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements, guaranteeing that every batch meets the highest international standards for quality and consistency.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific project requirements. Whether you are developing novel anticancer agents based on the indolocarbazole scaffold or advanced OLED materials, our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global market.
