Advanced Visible Light Mediated Dehydrogenation for Commercial N-Heterocycle Production
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways to construct nitrogen-containing heterocycles, which serve as critical scaffolds in countless bioactive molecules. A significant breakthrough in this domain is documented in patent CN113461617A, which discloses a method for preparing N-heterocycles through visible light mediated dehydrogenation. This technology represents a paradigm shift from traditional thermal methods to photochemical processes that operate under mild conditions. By leveraging the induction of visible light at room temperature, this approach circumvents the need for external heating sources, thereby reducing energy consumption and thermal stress on sensitive functional groups. For R&D directors and procurement managers alike, this innovation offers a compelling value proposition: the ability to synthesize complex structures like quinolines, indoles, and benzothiazoles with higher purity and lower environmental impact. As a reliable pharmaceutical intermediates supplier, understanding such technological advancements is crucial for maintaining a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of N-heterocyclic aromatic hydrocarbons has relied heavily on dehydration, ring-closing, and oxidative aromatization reactions using readily available nucleophilic reagents. However, these conventional pathways are fraught with significant chemical and economic inefficiencies that hinder large-scale adoption. Traditional methods typically necessitate the use of additional strong oxidants such as potassium permanganate or DDQ (2,3-Dichloro-5,6-dicyano-1,4-benzoquinone) to complete the final dehydroaromatization process. The reliance on such aggressive oxidizing agents often leads to poor compatibility with sensitive functional groups present in complex drug candidates, resulting in side reactions and reduced overall yields. Furthermore, the generation of substantial amounts of chemical byproducts creates a heavy burden on waste treatment facilities and increases the cost of goods sold. From a supply chain perspective, the handling and disposal of these hazardous oxidants introduce safety risks and regulatory compliance challenges that can delay project timelines and inflate operational expenditures.
The Novel Approach
In stark contrast to the harsh conditions of legacy methods, the novel approach described in the patent utilizes a visible light mediated dehydrogenation strategy that fundamentally alters the reaction landscape. The core of this innovation lies in the use of a novel four-coordination N,N-diaryl chelating borate compound as a photocatalyst. This specific catalyst design allows the reaction to proceed under the induction of visible light at room temperature, completely avoiding the need for heating. 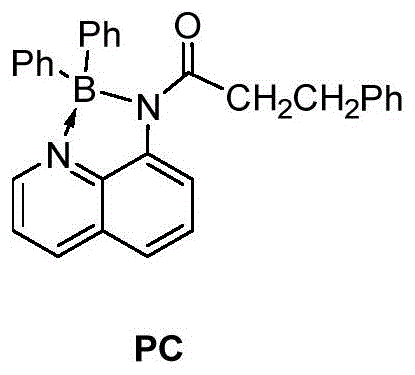 By replacing noble metal photocatalysts with this organic borate system, the method avoids the use of expensive and scarce resources like iridium or ruthenium. This substitution not only drives down raw material costs but also ensures that the reaction product is free from noble metal residues, a critical quality attribute for pharmaceutical intermediates. The use of oxygen or air as the oxidant further enhances the green chemistry profile, eliminating the need for stoichiometric metal oxidants and improving functional group tolerance.
By replacing noble metal photocatalysts with this organic borate system, the method avoids the use of expensive and scarce resources like iridium or ruthenium. This substitution not only drives down raw material costs but also ensures that the reaction product is free from noble metal residues, a critical quality attribute for pharmaceutical intermediates. The use of oxygen or air as the oxidant further enhances the green chemistry profile, eliminating the need for stoichiometric metal oxidants and improving functional group tolerance.
Mechanistic Insights into Visible Light Mediated Dehydrogenation
To fully appreciate the technical robustness of this method, one must delve into the mechanistic details of the photocatalytic cycle. The reaction initiates when the four-coordination N,N-diaryl chelating borate photocatalyst absorbs photons from the visible light source, typically blue LEDs with a wavelength around 455nm. This absorption promotes the catalyst to an excited state, enabling it to participate in single-electron transfer processes with the substrate. The aniline derivative and the aldehyde derivative undergo condensation to form an intermediate, which is then subjected to oxidative dehydrogenation facilitated by the excited photocatalyst and molecular oxygen. 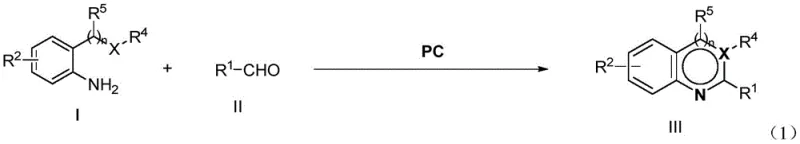 This mechanism ensures a smooth conversion to the aromatized N-heterocyclic compound without the formation of radical species that could degrade the product. The precision of this photochemical activation allows for exceptional control over the reaction pathway, minimizing the formation of impurities that are common in thermal oxidation processes. For R&D teams, this level of mechanistic clarity provides confidence in the reproducibility and reliability of the synthesis route.
This mechanism ensures a smooth conversion to the aromatized N-heterocyclic compound without the formation of radical species that could degrade the product. The precision of this photochemical activation allows for exceptional control over the reaction pathway, minimizing the formation of impurities that are common in thermal oxidation processes. For R&D teams, this level of mechanistic clarity provides confidence in the reproducibility and reliability of the synthesis route.
Impurity control is another critical aspect where this photocatalytic method excels over traditional oxidation techniques. In conventional methods using strong oxidants, over-oxidation is a frequent issue, leading to the formation of N-oxides or ring-opened byproducts that are difficult to separate. The visible light mediated process, however, operates under kinetic control where the energy input is precisely tuned to the absorption band of the catalyst. This specificity prevents the activation of unwanted side reactions, thereby preserving the integrity of sensitive substituents on the aromatic rings. The absence of transition metals in the catalyst system means there is no risk of metal leaching into the final product, which simplifies the purification workflow significantly. Instead of requiring specialized scavenging resins or complex extraction protocols to remove trace metals, the workup can often be achieved through standard filtration and chromatography. This streamlined purification process directly translates to higher recovery rates and reduced solvent consumption, aligning with the goals of cost reduction in fine chemical manufacturing.
How to Synthesize Quinazoline Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and purity. The general procedure involves mixing the aniline derivative, the benzaldehyde derivative, and the photocatalyst in a suitable polar aprotic solvent such as N-methylpyrrolidone or acetonitrile. The reaction mixture is then stirred uniformly to ensure homogeneous distribution of the catalyst before being exposed to the light source. It is essential to maintain an aerobic environment, either by flushing with oxygen or simply operating under air, as molecular oxygen serves as the terminal electron acceptor in the catalytic cycle.  The reaction is typically conducted at a mild temperature range of 25-30°C for approximately 10 hours under irradiation from 10W blue LEDs. Detailed standardized synthesis steps see the guide below.
The reaction is typically conducted at a mild temperature range of 25-30°C for approximately 10 hours under irradiation from 10W blue LEDs. Detailed standardized synthesis steps see the guide below.
- Mix aniline derivative, benzaldehyde derivative, and the novel borate photocatalyst (1 mol%) in a solvent such as N-methylpyrrolidone or acetonitrile.
- Maintain the reaction mixture under an aerobic environment (air or oxygen) and irradiate with 455nm blue LEDs at 25-30°C for 10 hours.
- Upon completion, filter the mixture, remove solvent via rotary evaporation, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this visible light mediated technology offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the dependence on noble metal catalysts, manufacturers are no longer subject to the volatile pricing and geopolitical supply risks associated with metals like palladium, platinum, or iridium. This shift to an organic borate-based system stabilizes the cost structure of the synthesis, allowing for more accurate long-term budgeting and pricing contracts. Furthermore, the use of air or oxygen as the oxidant removes the need to procure, store, and handle hazardous chemical oxidants, reducing inventory costs and safety compliance burdens. These factors collectively contribute to substantial cost savings in the overall manufacturing process, making the final intermediates more competitive in the global market.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal photocatalysts represents a direct reduction in bill of materials costs. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, while the simplified workup lowers solvent and labor expenses. The absence of metal residues also negates the need for costly metal scavenging steps, further enhancing the economic efficiency of the production line.
- Enhanced Supply Chain Reliability: Relying on readily available organic starting materials and air as a reagent significantly de-risks the supply chain. There is no dependency on single-source suppliers for specialized metal catalysts, ensuring continuity of supply even during market disruptions. The robustness of the reaction conditions also means that production can be maintained across different manufacturing sites with minimal re-validation efforts.
- Scalability and Environmental Compliance: The use of LED light sources and room temperature conditions makes this process highly amenable to scale-up, including continuous flow technologies. The green chemistry profile, characterized by reduced waste and safer reagents, facilitates easier regulatory approval and aligns with corporate sustainability goals, reducing the environmental footprint of chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light mediated dehydrogenation technology. These insights are derived directly from the patent data and are intended to clarify the feasibility and advantages of adopting this method for your specific project needs. Understanding these details is essential for making informed decisions about process development and vendor selection.
Q: What are the advantages of using the borate photocatalyst over noble metal catalysts?
A: The novel four-coordination N,N-diaryl chelating borate compound eliminates the need for expensive noble metals like iridium or ruthenium. This significantly reduces raw material costs and removes the risk of toxic metal residue in the final bioactive molecule, simplifying downstream purification.
Q: Does this method require harsh oxidants like DDQ or potassium permanganate?
A: No, this method utilizes molecular oxygen or air as the terminal oxidant. This avoids the use of strong chemical oxidants which often cause poor functional group compatibility and generate hazardous waste, thereby improving the environmental profile of the synthesis.
Q: Is this photocatalytic process scalable for industrial production?
A: Yes, the reaction operates at room temperature (25-30°C) using standard blue LED arrays. This mild condition facilitates heat management during scale-up and allows for continuous flow processing, making it highly suitable for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photocatalytic technologies in modern drug synthesis. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-equipped to adapt the visible light mediated dehydrogenation process described in CN113461617A to meet your specific volume requirements while maintaining stringent purity specifications. We operate rigorous QC labs that ensure every batch of high-purity N-heterocycle meets the highest international standards, providing you with the confidence needed to advance your drug candidates through clinical trials and into the market.
We invite you to leverage our expertise to optimize your supply chain and reduce your time to market. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals. Partner with us to secure a reliable supply of complex pharmaceutical intermediates produced with the latest in green chemistry innovation.
