Advanced Spiro[fluorene-9,9'-xanthene] Hole Transport Materials for High-Efficiency Perovskite Solar Cells
Advanced Spiro[fluorene-9,9'-xanthene] Hole Transport Materials for High-Efficiency Perovskite Solar Cells
The rapid evolution of perovskite solar cell (PSC) technology has created an urgent demand for hole transport materials (HTMs) that balance high efficiency with commercial viability. Patent CN112707882A introduces a groundbreaking series of spiro[fluorene-9,9'-xanthene] nuclear hole transport materials, specifically SFX-F, SFX-FM, and SFX-FP, which address the critical bottlenecks of cost and stability inherent in current market leaders. Unlike traditional architectures, these novel compounds utilize an asymmetric spiro core chemically modified with N-(4-methoxyphenyl)-9,9'-dimethyl-9H-fluorene-2-amine (FPA) groups to optimize energy levels and morphological stability. The strategic integration of the FPA unit into the xanthene moiety not only elevates the glass transition temperature but also enhances hole mobility, providing a robust foundation for next-generation photovoltaic devices. As a reliable electronic chemical supplier, understanding the structural nuances of these materials is essential for R&D teams aiming to surpass the 25% efficiency barrier while maintaining long-term operational stability.
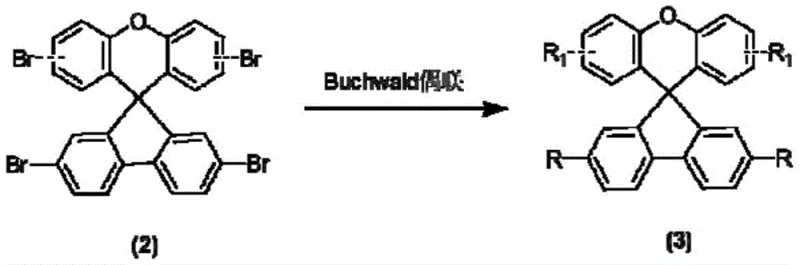
![General chemical structure of Spiro[fluorene-9,9'-xanthene] hole transport materials showing variable R and R1 groups](/insights/img/spiro-fluorene-xanthene-htm-synthesis-supplier-20260306134155-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The incumbent standard for high-efficiency perovskite solar cells, Spiro-OMeTAD, suffers from severe commercialization hurdles primarily driven by its convoluted synthetic pathway and exorbitant raw material costs. The synthesis of Spiro-OMeTAD typically requires multiple cryogenic lithiation steps using hazardous reagents such as n-butyllithium or tert-butyllithium at temperatures as low as -78°C, necessitating specialized infrastructure and rigorous safety protocols that drastically inflate manufacturing overheads. Furthermore, the purification of Spiro-OMeTAD is notoriously difficult due to the formation of closely related impurities that are hard to separate, often requiring repeated column chromatography or sublimation which lowers the overall yield and increases waste generation. From a supply chain perspective, the reliance on specific, high-purity spirobifluorene precursors creates a bottleneck, as the global supply of these intermediates is limited and subject to significant price volatility. Consequently, the high cost of goods sold (COGS) for Spiro-OMeTAD remains a prohibitive factor for the mass deployment of perovskite modules in utility-scale applications.
The Novel Approach
The innovative methodology disclosed in the patent circumvents these challenges by employing a thermodynamically favorable acid-catalyzed condensation to construct the spiro[fluorene-9,9'-xanthene] core, followed by a robust palladium-catalyzed cross-coupling reaction. This route eliminates the need for cryogenic conditions and pyrophoric organolithium reagents, allowing the reaction to proceed at moderate temperatures between 120°C and 170°C using methanesulfonic acid as a benign catalyst. The use of readily available phenol and bromophenol derivatives as starting materials significantly reduces raw material costs and simplifies the procurement process for large-scale manufacturing. Moreover, the modular nature of the Buchwald-Hartwig coupling step allows for precise tuning of the electronic properties by varying the aniline substituents, enabling the optimization of HOMO/LUMO energy levels without redesigning the entire synthetic sequence. This streamlined approach not only accelerates the time-to-market for new HTM variants but also ensures a more consistent impurity profile, which is critical for achieving reproducible device performance in commercial settings.
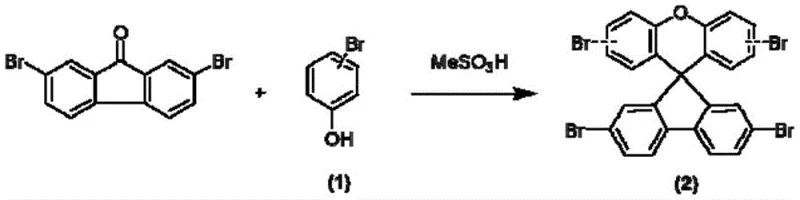
Mechanistic Insights into Acid-Catalyzed Cyclization and Pd-Coupling
The formation of the critical spiro[fluorene-9,9'-xanthene] scaffold relies on an electrophilic aromatic substitution mechanism facilitated by methanesulfonic acid (MeSO3H), which acts as both a solvent and a proton source to activate the carbonyl group of 2,7-dibromofluorenone. Under heating conditions of 120-170°C, the acid protonates the ketone oxygen, increasing the electrophilicity of the central carbon and facilitating a nucleophilic attack by the electron-rich phenol ring. This cyclization step is highly regioselective, favoring the ortho-position relative to the hydroxyl group due to intramolecular hydrogen bonding stabilization in the transition state, which ensures the formation of the desired spiro-junction with minimal isomeric byproducts. The subsequent dehydration leads to the stable spiro-core intermediate (Formula 2), where the orthogonal arrangement of the fluorene and xanthene units inhibits molecular aggregation, thereby enhancing the amorphous character and film-forming properties of the final material. This mechanistic pathway is superior to Friedel-Crafts alkylation using Lewis acids like AlCl3, as it avoids the generation of stoichiometric amounts of corrosive waste and simplifies the downstream workup procedure.
Following the core synthesis, the functionalization is achieved through a palladium-catalyzed Buchwald-Hartwig amination, a powerful tool for constructing C-N bonds under mild conditions. In this catalytic cycle, the Pd(0) species undergoes oxidative addition into the carbon-bromine bond of the spiro-core intermediate, forming a reactive aryl-palladium complex that is susceptible to nucleophilic attack by the amine donor. The presence of bulky phosphine ligands, such as tri-tert-butylphosphine, stabilizes the active Pd(0) species and accelerates the reductive elimination step, which releases the final coupled product and regenerates the catalyst. The use of strong bases like potassium tert-butoxide ensures the deprotonation of the amine to generate a more nucleophilic amido species, driving the reaction to completion even with sterically hindered substrates. Crucially, this coupling method offers excellent tolerance to functional groups, allowing the preservation of the methoxy and methyl substituents that are vital for the hole-transporting capability, while minimizing the formation of homocoupling impurities that often plague nickel-catalyzed alternatives.
How to Synthesize Spiro[fluorene-9,9'-xanthene] Derivatives Efficiently
The synthesis of these high-performance hole transport materials is designed for scalability, utilizing a two-stage process that begins with the construction of the brominated spiro-core followed by the attachment of the charge-transporting arms. The initial condensation reaction requires careful temperature control to prevent polymerization of the phenol starting material, while the subsequent coupling step demands anhydrous conditions to maintain catalyst activity. Operators should note that the stoichiometry of the base and ligand is critical; an excess of base ensures complete deprotonation of the amine, while the ligand-to-palladium ratio must be optimized to prevent catalyst precipitation during the extended reflux period. For detailed standardized operating procedures regarding reagent grades, specific addition rates, and purification protocols, please refer to the technical guide below.
- Condense 2,7-dibromofluorenone with phenol derivatives using methanesulfonic acid at 120-170°C to form the brominated spiro-core intermediate.
- Perform Buchwald-Hartwig coupling between the spiro-core intermediate and N-(4-methoxyphenyl)-9,9-dimethyl-9H-fluoren-2-amine using a Pd catalyst.
- Purify the final crude product via column chromatography using petroleum ether and ethyl acetate to obtain high-purity hole transport material.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this new class of spiro[fluorene-9,9'-xanthene] materials offers substantial strategic benefits centered around cost reduction and supply security. The elimination of cryogenic processing steps translates directly into lower capital expenditure (CAPEX) for manufacturing facilities, as standard stainless steel reactors can be used instead of specialized glass-lined vessels equipped for extreme low-temperature operations. Furthermore, the replacement of expensive organolithium reagents with commodity chemicals like methanesulfonic acid and phenols drastically reduces the variable cost per kilogram, making the final HTM price point much more competitive against the entrenched Spiro-OMeTAD standard. This cost efficiency is compounded by the higher yields observed in the acid-catalyzed cyclization step, which minimizes raw material waste and maximizes the throughput of the production line. By adopting this synthetic route, manufacturers can achieve a more predictable cost structure, shielding themselves from the volatility associated with specialty reagent markets.
- Cost Reduction in Manufacturing: The synthetic pathway leverages thermally driven acid catalysis rather than energy-intensive cryogenic lithiation, effectively removing the need for expensive cooling infrastructure and hazardous pyrophoric reagents. This shift significantly lowers the operational expenditure (OPEX) by reducing energy consumption for refrigeration and minimizing the costs associated with the safe handling and disposal of dangerous chemicals. Additionally, the use of palladium catalysts, while precious, is highly efficient with turnover numbers that justify the investment, especially when compared to the stoichiometric quantities of lithium reagents required in conventional methods. The overall simplification of the workflow reduces labor hours and quality control testing time, contributing to a leaner and more cost-effective manufacturing process.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including 2,7-dibromofluorenone and various substituted phenols, are commodity chemicals available from multiple global suppliers, ensuring a robust and diversified supply chain. This contrasts sharply with the proprietary or limited-source precursors often required for legacy HTMs, which can lead to single points of failure and delivery delays. The robustness of the reaction conditions also means that production is less susceptible to disruptions caused by minor variations in utility supplies, such as cooling water temperature fluctuations. Consequently, manufacturers can maintain higher inventory levels and shorter lead times, providing a reliable buffer against market shocks and ensuring continuous availability for downstream solar cell producers.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram pilot runs without the need for complex engineering modifications. The absence of halogenated solvents in the initial cyclization step and the ability to recover and recycle the toluene solvent in the coupling step align with green chemistry principles, reducing the environmental footprint of the manufacturing operation. Waste streams are primarily organic and easier to treat compared to the heavy metal and lithium salt wastes generated by traditional routes, simplifying compliance with increasingly stringent environmental regulations. This environmental compatibility not only reduces disposal costs but also enhances the corporate sustainability profile of the final photovoltaic product, a key selling point in the renewable energy sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these novel hole transport materials in perovskite solar cell production lines. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making. Understanding these specifics helps R&D and procurement teams evaluate the feasibility of integrating SFX-F, SFX-FM, or SFX-FP into their existing device architectures.
Q: How does the thermal stability of SFX-FM compare to conventional Spiro-OMeTAD?
A: The SFX-FM material exhibits a significantly higher thermal decomposition temperature of 445°C and a glass transition temperature of 140°C, surpassing the thermal limits of standard Spiro-OMeTAD, which enhances device longevity under operational heat stress.
Q: What represents the primary cost advantage in this synthetic route?
A: The primary cost advantage stems from replacing cryogenic lithiation steps with a thermal acid-catalyzed condensation, eliminating the need for expensive pyrophoric reagents like tert-butyllithium and reducing energy consumption for cooling.
Q: Can these materials be scaled for industrial perovskite module production?
A: Yes, the synthesis utilizes robust reaction conditions (80-120°C) and commercially available starting materials like phenols and fluorenones, making the process highly amenable to multi-kilogram scale-up without specialized low-temperature infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro[fluorene-9,9'-xanthene] Supplier
As the global demand for high-efficiency perovskite solar cells accelerates, securing a stable supply of advanced hole transport materials is paramount for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this technological shift, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver these complex spiro-core intermediates with unmatched consistency. Our state-of-the-art facilities are equipped to handle the specific thermal and catalytic requirements of the Buchwald-Hartwig coupling and acid-cyclization steps, ensuring that every batch meets stringent purity specifications required for high-performance optoelectronic applications. With rigorous QC labs dedicated to analyzing trace impurities and verifying thermal stability parameters, we guarantee that our materials provide the reliable foundation your solar modules need to achieve record-breaking efficiencies.
We invite forward-thinking partners to collaborate with us to optimize their supply chains and reduce manufacturing costs without compromising on performance. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our spiro[fluorene-9,9'-xanthene] derivatives can serve as a superior, cost-effective alternative to traditional hole transport materials in your next-generation photovoltaic projects.
