Scalable Production of L-Selenomethylselenocysteine via Elemental Selenium Substitution
Scalable Production of L-Selenomethylselenocysteine via Elemental Selenium Substitution
The global demand for organic selenium supplements has surged due to increasing awareness of selenium's critical role in antioxidant defense, immune function, and cancer prevention. However, the manufacturing landscape for high-purity L-selenomethylselenocysteine (L-Se-MeSeCys) has long been constrained by hazardous reagents and inefficient processes. A groundbreaking approach detailed in patent CN114805155A introduces a transformative synthetic route that utilizes elemental selenium as the primary selenium source. This innovation addresses the longstanding pain points of toxicity, odor, and cost associated with traditional diselenide-based methods. By leveraging a nucleophilic substitution strategy on protected serine derivatives, this technology offers a robust pathway for producing this vital nutritional ingredient with superior safety profiles and operational simplicity.
For pharmaceutical and nutraceutical manufacturers, the shift towards greener, more economical synthesis routes is not merely a regulatory preference but a strategic imperative. The methodology disclosed in CN114805155A replaces volatile and malodorous dimethyl diselenide with stable, inexpensive selenium powder. This fundamental change in raw material selection cascades into significant advantages across the supply chain, from procurement savings to enhanced worker safety. As we delve deeper into the technical specifics, it becomes evident that this process represents a paradigm shift in the commercial production of organoselenium compounds, aligning perfectly with modern principles of sustainable chemistry and cost-effective manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of L-selenomethylselenocysteine has relied on three primary strategies, each fraught with significant industrial drawbacks. The first approach involves the 1,4-conjugated addition of methyl selenol to alpha-amino acrylic acid derivatives. This route is inherently inefficient, often yielding racemic mixtures that necessitate costly and yield-reducing chiral resolution steps to isolate the biologically active L-enantiomer. The second conventional method utilizes N-tert-butoxy acyl-L-serine but requires the formation of a beta-lactone intermediate followed by selenylation ring opening. This multi-step sequence suffers from difficult raw material preparation and the use of expensive reagents, leading to poor overall yields and inflated production costs. Perhaps most critically, the third common method employs N-acetyl-3-chloro-L-serine and sodium methylselenolate generated from dimethyl diselenide.
The reliance on dimethyl diselenide in traditional processes presents a severe operational bottleneck. This reagent is notorious for its extremely unpleasant, garlic-like odor, which poses significant challenges for containment and worker comfort in large-scale facilities. Furthermore, dimethyl diselenide is chemically unstable and difficult to store safely over long periods. The generation of the necessary nucleophile often involves the reduction of diselenides with sodium borohydride, a reaction characterized by violent exotherms that pose serious safety risks during scale-up. These factors combined—low yields, high costs, safety hazards, and environmental concerns—have created an urgent need for a superior manufacturing technology that can deliver high-purity L-selenomethylselenocysteine without compromising on safety or economics.
The Novel Approach
The innovative process described in patent CN114805155A circumvents these historical limitations by employing elemental selenium powder directly in conjunction with a Grignard reagent. This method initiates with the formation of a reactive seleno-Grignard species under mild low-temperature conditions, typically between -30°C and 0°C. This activated selenium species then undergoes a direct nucleophilic substitution reaction with a protected amino acid halide, specifically N-tert-butoxy acyl-3-iodo-L-serine methyl ester. The elegance of this approach lies in its simplicity and the use of the chiral pool; by starting with L-serine derivatives, the stereochemistry is preserved throughout the synthesis, eliminating the need for resolution.
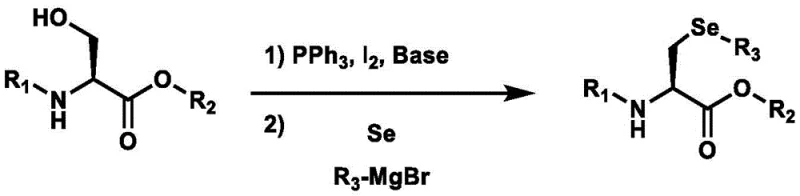
Furthermore, the process is designed as a streamlined one-pot procedure where the substitution is followed directly by acidic hydrolysis to remove protecting groups. This consolidation of steps significantly reduces solvent usage, processing time, and waste generation compared to multi-step traditional routes. The use of elemental selenium not only eradicates the foul odor associated with diselenides but also leverages a raw material that is abundant, cheap, and easy to handle. The reaction conditions are notably mild, avoiding the violent exotherms of borohydride reductions, thereby enhancing the inherent safety of the manufacturing process. This novel approach effectively decouples high-quality production from high operational risk, offering a commercially viable solution for the growing nutraceutical market.
Mechanistic Insights into Selenium-Grignard Nucleophilic Substitution
The core chemical transformation in this patented method relies on the generation of a highly nucleophilic selenolate anion via a Grignard-type activation of elemental selenium. When elemental selenium powder reacts with a Grignard reagent such as methylmagnesium bromide (MeMgBr), it forms a magnesium selenolate species (R-Se-MgBr). This species acts as a potent nucleophile, capable of attacking electrophilic carbon centers with high efficiency. In the context of this synthesis, the electrophile is the beta-carbon of a protected serine derivative that has been converted into a good leaving group, typically an iodide. The reaction proceeds via an SN2 mechanism, where the selenolate attacks the carbon bearing the iodine atom, displacing the iodide ion and forming the carbon-selenium bond essential for the target molecule.
Controlling the reaction environment is critical to maintaining the integrity of the chiral center and preventing side reactions. The patent specifies the use of polar aprotic solvents like N,N-dimethylformamide (DMF) or tetrahydrofuran (THF), which facilitate the solubility of the intermediates and stabilize the ionic species involved. The temperature control, initially kept low during the formation of the seleno-Grignard reagent and then allowed to warm to room temperature during substitution, ensures that the reaction kinetics are optimized for substitution rather than elimination or decomposition. Following the bond formation, the protecting groups (such as Boc and methyl ester) are removed via acidic hydrolysis using hydrochloric acid. This step cleaves the ester and carbamate functionalities, revealing the free amine and carboxylic acid of the final L-selenomethylselenocysteine product. The final pH adjustment to alkaline conditions (pH 9-10) using ammonia facilitates the crystallization of the zwitterionic product, ensuring high purity and recovery.
How to Synthesize L-Selenomethylselenocysteine Efficiently
The practical execution of this synthesis involves a carefully sequenced addition of reagents to maximize yield and safety. The process begins with the activation of selenium powder, followed by the introduction of the amino acid substrate, and concludes with a workup that isolates the pure crystalline product. The detailed standardized synthetic steps for implementing this patented technology are outlined below.
- Preparation of Seleno-Grignard Reagent: React elemental selenium powder with a Grignard reagent (e.g., MeMgBr) at low temperatures (-30°C to 0°C) under inert atmosphere.
- Nucleophilic Substitution: Introduce the protected amino acid halide (e.g., N-Boc-3-iodo-L-alanine methyl ester) to the reaction mixture to effect substitution.
- Hydrolysis and Isolation: Perform acidic hydrolysis (e.g., HCl reflux) to remove protecting groups, followed by pH adjustment and recrystallization to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the CN114805155A process translates into tangible strategic benefits that extend far beyond simple chemical yield. The primary advantage lies in the radical simplification of the raw material portfolio. By replacing specialized, hazardous, and expensive reagents like dimethyl diselenide and sodium borohydride with commodity chemicals like elemental selenium and Grignard reagents, manufacturers can significantly reduce their exposure to volatile raw material markets. Elemental selenium is a widely available industrial commodity with a stable supply chain, ensuring continuity of production even during global disruptions. This shift mitigates the risk of production stoppages caused by the scarcity of niche reagents, providing a more resilient foundation for long-term manufacturing planning.
- Cost Reduction in Manufacturing: The economic impact of this new route is profound, driven primarily by the elimination of costly chiral resolution steps and expensive reagents. Traditional methods often suffer from theoretical maximum yields of 50% due to racemization, requiring double the starting material to achieve the same output of L-enantiomer. In contrast, this chiral pool approach preserves stereochemistry from the outset, theoretically doubling the efficiency of raw material utilization. Additionally, the removal of malodorous diselenides eliminates the need for specialized scrubbing systems and extensive personal protective equipment (PPE) protocols, leading to substantial operational expenditure (OpEx) savings. The one-pot nature of the reaction further reduces solvent consumption and energy usage by consolidating multiple unit operations into a single vessel.
- Enhanced Supply Chain Reliability: Supply chain stability is bolstered by the use of robust, shelf-stable starting materials. Unlike dimethyl diselenide, which degrades and poses storage challenges, elemental selenium powder can be stored indefinitely without special precautions. This stability allows for bulk purchasing strategies that capitalize on economies of scale, reducing the frequency of orders and logistics overhead. Furthermore, the mild reaction conditions (-30°C to room temperature) reduce the dependency on extreme cooling or heating infrastructure, making the process adaptable to a wider range of manufacturing facilities. This flexibility ensures that production can be scaled up or shifted between sites with minimal capital investment in specialized reactor modifications.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a cleaner profile that aligns with increasingly stringent global regulations. The absence of toxic heavy metal catalysts and the reduction of hazardous waste streams simplify wastewater treatment and disposal procedures. The replacement of violent exothermic reductions with controlled Grignard formations enhances process safety, lowering insurance premiums and reducing the risk of catastrophic incidents. The streamlined workflow results in less waste generation per kilogram of product, supporting corporate sustainability goals and reducing the carbon footprint of the manufacturing operation. These factors collectively position the manufacturer as a responsible supplier, a key differentiator when bidding for contracts with major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selenium substitution technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when adopting this route.
Q: Why is elemental selenium preferred over dimethyl diselenide in this synthesis?
A: Elemental selenium is significantly cheaper, odorless, and more stable than dimethyl diselenide, which is known for its terrible smell and instability. This switch drastically improves workplace safety and reduces raw material costs.
Q: How does this method ensure chiral purity?
A: The process utilizes chiral pool synthesis starting from L-serine derivatives. By avoiding racemization-prone steps like conjugate addition to achiral acrylic acids, the method maintains high optical purity (>99% L-configuration) without needing complex resolution.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the reaction conditions are mild (-30°C to room temperature) and avoid violent exothermic reactions associated with sodium borohydride reductions. The one-pot nature of the substitution and hydrolysis simplifies equipment requirements and waste handling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Selenomethylselenocysteine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven scale-up capabilities. Our team has extensively analyzed the methodology presented in CN114805155A and possesses the requisite experience to translate this laboratory-scale innovation into robust commercial reality. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel selenium chemistry are fully realized at an industrial level. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of L-selenomethylselenocysteine meets the highest standards for nutritional and pharmaceutical applications.
We invite forward-thinking organizations to collaborate with us to optimize their supply chains for selenium-based ingredients. By leveraging our technical proficiency, you can secure a reliable source of high-purity intermediates while achieving significant cost efficiencies. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this green synthesis method can enhance your product portfolio and bottom line.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
