Revolutionizing Caprolactam Production: High-Selectivity Ionic Liquid Catalysis for Industrial Scale-Up
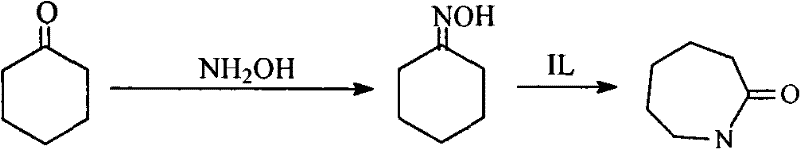
The chemical industry is currently witnessing a paradigm shift in the production of key polymer intermediates, driven by the urgent need for greener, more efficient synthetic routes. A pivotal development in this arena is documented in patent CN102001999B, which outlines a novel technique for the direct synthesis of caprolactam from cyclohexanone and hydroxylamine. This technology represents a significant departure from conventional multi-step processes, offering a streamlined one-pot solution that operates within a phosphate buffer solution system. For R&D directors and process engineers, the implications are profound, as this method not only simplifies the reaction pathway but also drastically improves selectivity metrics. By integrating an acidic ionic liquid catalyst, specifically N,N,N-trimethyl-N-sulfobutyl-ammonium hydrogen sulfate, the process achieves a caprolactam selectivity of up to 92.1%. This level of precision is critical for downstream applications in nylon-6 production, where impurity profiles can dictate the mechanical properties of the final polymer. The ability to conduct two distinct chemical transformations—oximation and Beckmann rearrangement—within a single reactor vessel under mild conditions marks a substantial advancement in process intensification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional industrial production of caprolactam has long been plagued by inherent inefficiencies and environmental burdens associated with the classic two-step method. In the conventional workflow, cyclohexanone is first converted to cyclohexanone oxime, which subsequently undergoes a Beckmann rearrangement in the presence of oleum or concentrated sulfuric acid to form caprolactam sulfate. This intermediate must then be neutralized with ammonia water, a step that inevitably generates massive quantities of low-value ammonium sulfate by-products. From a supply chain and operational perspective, this creates a logistical nightmare, requiring extensive waste treatment facilities and adding significant cost to the overall manufacturing equation. Furthermore, the use of strong mineral acids leads to severe equipment corrosion, necessitating the use of expensive corrosion-resistant materials and frequent maintenance schedules. The frequent transfer of materials between different reactors and containers increases the risk of contamination and exposure to hazardous substances, posing safety risks to personnel and complicating regulatory compliance regarding volatile organic compound emissions.
The Novel Approach
In stark contrast, the innovative methodology described in the patent data utilizes a phosphate buffer solution system that fundamentally alters the reaction landscape. By employing a staged feeding process within a single reactor, this approach eliminates the need for intermediate isolation and the harsh neutralization steps characteristic of older technologies. The use of room temperature ionic liquids as catalysts replaces the corrosive mineral acids, thereby preserving equipment integrity and extending the operational lifespan of the reactor infrastructure. This shift not only reduces the capital expenditure required for specialized corrosion-resistant machinery but also minimizes the footprint of the production facility by consolidating multiple unit operations into one. The mild reaction conditions, operating at normal pressure and temperatures ranging from 30 to 60°C, further contribute to energy efficiency by reducing the thermal load on the system. Consequently, this novel approach offers a robust pathway for cost reduction in polymer intermediate manufacturing, aligning perfectly with modern sustainability goals while maintaining high throughput capabilities.
Mechanistic Insights into Ionic Liquid-Catalyzed Beckmann Rearrangement
The core of this technological breakthrough lies in the unique catalytic behavior of the acidic ionic liquid, N,N,N-trimethyl-N-sulfobutyl-ammonium hydrogen sulfate, within the phosphate buffer medium. Unlike traditional solid acid catalysts that often suffer from deactivation or leaching issues, this ionic liquid provides a homogeneous catalytic environment that facilitates the Beckmann rearrangement with exceptional efficiency. The mechanism involves the activation of the cyclohexanone oxime intermediate through protonation by the acidic protons of the ionic liquid, lowering the activation energy required for the rearrangement to caprolactam. The phosphate buffer plays a crucial dual role: it maintains the pH stability necessary for the initial oximation of cyclohexanone with hydroxylamine sulfate, and it provides a compatible medium for the subsequent ionic liquid-catalyzed rearrangement. This compatibility ensures that the reaction proceeds smoothly without the formation of complex salt precipitates that could foul the reactor or complicate product separation. The result is a clean reaction profile where the primary by-product is unreacted cyclohexanone oxime, which can be easily recycled back into the process, thereby maximizing atom economy.
Controlling the impurity spectrum is paramount for producing high-purity caprolactam suitable for fiber-grade nylon-6 applications. The selective nature of this ionic liquid catalyst minimizes side reactions that typically lead to the formation of heavy ends or colored impurities. In comparative studies referenced in the patent data, traditional solid acid catalysts yielded selectivity levels below 53%, accompanied by significant amounts of benzenesulfonic acid by-products. In contrast, the buffered ionic liquid system achieves selectivity rates exceeding 92%, demonstrating superior control over the reaction trajectory. This high selectivity translates directly to reduced downstream purification costs, as fewer distillation columns or crystallization steps are required to meet stringent quality specifications. For procurement managers, this means a more reliable supply of consistent quality material, reducing the risk of batch rejections and ensuring continuity in the production of downstream polymers. The stability of the ionic liquid also suggests potential for reuse, further enhancing the economic viability of the process over long-term operation cycles.
How to Synthesize Caprolactam Efficiently
Implementing this advanced synthesis route requires precise adherence to the stoichiometric ratios and sequential addition protocols defined in the patent literature. The process begins with the preparation of a specific phosphate buffer solution, followed by the controlled addition of reactants to initiate oximation. Once the initial conversion is achieved, the ionic liquid catalyst is introduced to drive the rearrangement to completion. This structured approach ensures optimal yield and selectivity while maintaining safety standards. For detailed operational parameters, including exact molar ratios, temperature ramps, and mixing speeds, please refer to the standardized synthesis guide below which outlines the critical control points for successful scale-up.
- Prepare a phosphate buffer solution by mixing sodium hydroxide, sodium nitrate, phosphoric acid, and water to achieve a phosphate radical concentration of 3mol/L.
- Add hydroxylamine sulfate and cyclohexanone to the buffer solution at a molar ratio of 1: 2 and react at 30-50°C for 5-30 minutes under normal pressure.
- Directly add the ionic liquid catalyst N,N,N-trimethyl-N-sulfobutyl-ammonium hydrogen sulfate to the mixture and continue reacting for 10-30 minutes to obtain caprolactam.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this ionic liquid-based synthesis route offers transformative advantages that extend far beyond simple yield improvements. The elimination of corrosive mineral acids and the reduction of hazardous by-products fundamentally reshape the cost structure of caprolactam manufacturing. By removing the need for extensive neutralization and waste treatment associated with ammonium sulfate disposal, manufacturers can realize substantial cost savings in both raw material consumption and environmental compliance. The simplified equipment requirements mean that production facilities can be retrofitted or constructed with lower capital intensity, allowing for faster deployment of new capacity to meet market demand. Furthermore, the mild operating conditions reduce energy consumption, contributing to a lower carbon footprint which is increasingly becoming a criterion for supplier selection in global markets. These factors combine to create a more resilient and cost-effective supply chain capable of withstanding fluctuations in raw material prices and regulatory pressures.
- Cost Reduction in Manufacturing: The transition to a one-pot synthesis strategy significantly lowers operational expenditures by consolidating multiple reaction steps into a single vessel. This consolidation reduces the labor hours required for material handling and transfer, while also minimizing the loss of product during intermediate stages. The avoidance of expensive corrosion-resistant alloys for reactor construction, made possible by the non-corrosive nature of the ionic liquid and buffer system, results in drastic capital cost reductions. Additionally, the high selectivity of the process means that less raw material is wasted on by-products, effectively lowering the cost per kilogram of the final active ingredient. These efficiencies compound over time, providing a sustainable competitive advantage in pricing strategies for bulk chemical procurement.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical polymer intermediates is contingent upon the robustness of the manufacturing process. This technology enhances reliability by simplifying the process flow, thereby reducing the number of potential failure points such as pump transfers between reactors or complex filtration units. The ability to recycle unreacted oxime back into the process ensures that feedstock utilization is maximized, buffering the supply chain against volatility in cyclohexanone availability. Moreover, the stability of the ionic liquid catalyst suggests longer campaign lengths between maintenance shutdowns, ensuring consistent delivery schedules to downstream customers. This operational stability is crucial for just-in-time manufacturing environments where interruptions can cascade into significant production losses for end-users.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but the mild conditions of this synthesis route facilitate smoother scale-up from pilot to commercial production. The absence of high-pressure requirements and extreme temperatures reduces the engineering complexity associated with large-scale reactors. From an environmental standpoint, the process aligns with green chemistry principles by eliminating the generation of bulk inorganic salts like ammonium sulfate. This reduction in waste volume simplifies effluent treatment and lowers the regulatory burden on the manufacturing site. Compliance with increasingly stringent environmental regulations is easier to maintain, reducing the risk of fines or operational suspensions. This environmental stewardship enhances the brand value of the supplier, appealing to eco-conscious partners in the automotive and textile industries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalyzed synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for making informed decisions about process adoption and supplier qualification.
Q: What is the selectivity advantage of this ionic liquid method over traditional solid acid catalysts?
A: This patented process achieves a caprolactam selectivity of up to 92.1%, which is significantly higher than the less than 53% selectivity observed in traditional solid acid catalyst methods using oleum media.
Q: Does this process eliminate the production of ammonium sulfate by-products?
A: Yes, by utilizing a phosphate buffer system and avoiding sulfuric acid neutralization steps, the process avoids the generation of large amounts of low-value ammonium sulfate, aligning with green chemistry principles.
Q: What are the reaction conditions required for the Beckmann rearrangement step?
A: The rearrangement occurs under mild conditions at normal pressure and temperatures between 30-60°C, utilizing an acidic ionic liquid catalyst rather than harsh mineral acids.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Caprolactam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain competitiveness in the global fine chemicals market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts like the ionic liquid-catalyzed caprolactam synthesis can be successfully translated into industrial reality. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our capability to handle complex chemistries involving sensitive catalysts and buffer systems positions us as a strategic partner for companies seeking to optimize their polymer supply chains.
We invite you to collaborate with us to explore how this advanced synthesis route can benefit your specific application requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic benefits of switching to this greener technology. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to validate the quality and consistency of our production capabilities firsthand. Together, we can drive efficiency and sustainability in the production of high-performance polymer intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
