Advanced One-Step Dehydration Route for High-Purity Venlafaxine Impurity Manufacturing
The pharmaceutical industry's relentless pursuit of drug safety has elevated the importance of impurity profiling to a critical level, particularly for widely prescribed antidepressants like Venlafaxine hydrochloride. Patent CN114292198A introduces a groundbreaking preparation method for a specific Venlafaxine impurity, addressing a significant gap in the availability of high-quality reference standards for quality control laboratories. This innovation leverages a straightforward acid-catalyzed dehydration strategy to convert the precursor alcohol (Compound I) directly into the target alkene impurity (Compound II) with exceptional efficiency. By utilizing concentrated hydrochloric acid under controlled thermal conditions, the process eliminates the need for complex multi-step syntheses that have traditionally plagued impurity generation. This technical breakthrough not only streamlines the production of essential analytical standards but also offers a robust pathway for pharmaceutical manufacturers to enhance their quality assurance protocols regarding raw materials and finished dosage forms.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of specific drug-related impurities for regulatory compliance has been fraught with challenges, often involving laborious multi-step synthetic routes that suffer from poor atom economy and low overall yields. Traditional methods frequently rely on harsh reagents or transition metal catalysts that introduce difficult-to-remove contaminants, thereby complicating the purification process and inflating the cost of goods sold for reference standards. Furthermore, the lack of standardized, high-yield protocols for generating degradation products like the alkene derivative of Venlafaxine has forced quality control teams to rely on scarce or prohibitively expensive commercial sources. These conventional approaches often fail to provide the sufficient quantities of material required for comprehensive method validation and stability studies, creating bottlenecks in the drug development lifecycle and delaying time-to-market for generic formulations.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology disclosed in CN114292198A presents a remarkably elegant solution through a direct one-step dehydration reaction. The process transforms Compound I into Compound II by simply heating the substrate in an aqueous hydrochloric acid solution, leveraging the inherent reactivity of the benzylic alcohol moiety. This approach drastically reduces operational complexity by removing the need for intermediate isolation or specialized catalytic systems, allowing for a seamless transition from raw material to purified impurity. The visual representation of this transformation highlights the structural simplicity of the conversion, where the hydroxyl group is eliminated to form a stable carbon-carbon double bond conjugated with the aromatic system.
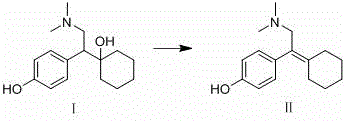
This novel route ensures high selectivity, minimizing the formation of side products and facilitating easier downstream purification via standard column chromatography. The ability to achieve such high conversion rates using commodity chemicals like hydrochloric acid represents a paradigm shift in how reference impurities are manufactured, offering a scalable and economically viable alternative for the global supply chain.
Mechanistic Insights into Acid-Catalyzed Dehydration
The core chemical transformation driving this synthesis is an acid-catalyzed elimination reaction, specifically following an E1 or E2 mechanism depending on the precise solvation and protonation dynamics within the concentrated acidic medium. The reaction initiates with the protonation of the hydroxyl group on the cyclohexyl ring of Compound I by the hydronium ions present in the 8mol/L to 12mol/L hydrochloric acid solution. This protonation converts the poor leaving group (-OH) into an excellent leaving group (water), which subsequently departs to generate a stabilized carbocation intermediate adjacent to the aromatic ring. The resonance stabilization provided by the phenyl group significantly lowers the activation energy for this step, facilitating the rapid formation of the cationic species even at moderate temperatures ranging from 80°C to 100°C.
Following the formation of the carbocation, a base present in the solution, such as a water molecule or chloride ion, abstracts a proton from the adjacent carbon atom, leading to the formation of the carbon-carbon double bond observed in Compound II. This elimination step is highly regioselective due to the thermodynamic stability of the resulting conjugated alkene system, which aligns the new pi-bond with the aromatic ring. The rigorous control of reaction parameters, including the mass-to-volume ratio of 1:5 to 1:15 and the specific reaction time window of 1.5 to 4 hours, ensures that the equilibrium favors the product while preventing potential over-reaction or polymerization. This mechanistic clarity allows process chemists to predict impurity profiles accurately and optimize the workup procedure, typically involving neutralization with saturated sodium bicarbonate and extraction with dichloromethane, to isolate the target molecule with high purity.
How to Synthesize Venlafaxine Impurity Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, operators must adhere to strict parameter controls regarding acid concentration and thermal input to maximize the yield which can reach up to 90 percent. The process begins by dissolving the starting material in the specified concentration of hydrochloric acid, followed by heating to the target temperature range where LC-MS monitoring confirms the complete consumption of the starting alcohol. Once the reaction is deemed complete, the mixture is cooled to ambient temperature and carefully neutralized to prevent acid-catalyzed degradation during the extraction phase. The detailed standardized synthesis steps, including specific workup procedures and purification protocols, are outlined in the guide below.
- Dissolve Compound I in an aqueous hydrochloric acid solution with a concentration between 8mol/L and 12mol/L, maintaining a mass-to-volume ratio of 1: 5 to 1:15.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for a duration of 1.5 to 4 hours to ensure complete dehydration.
- Cool the reaction to room temperature, neutralize the pH using saturated sodium bicarbonate, extract with DCM, and purify the organic phase via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented dehydration technology offers substantial strategic benefits that extend far beyond simple chemical synthesis. By replacing complex, multi-step impurity generation processes with a single-pot reaction using ubiquitous reagents, manufacturers can significantly reduce the operational overhead associated with sourcing specialized catalysts and managing hazardous waste streams. The reliance on hydrochloric acid, a globally available and inexpensive commodity chemical, insulates the production process from the volatility often seen in the pricing of rare earth metals or proprietary organocatalysts. This stability in raw material costs translates directly into more predictable budgeting for quality control departments and reduces the total cost of ownership for maintaining a comprehensive impurity library.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction of synthetic steps from multiple stages to a single dehydration event results in drastic cost savings. Without the need for costly ligand systems or extensive purification trains to remove metal residues, the overall manufacturing expense is significantly lowered. Furthermore, the high yield reported in the patent minimizes raw material waste, ensuring that every gram of starting material contributes efficiently to the final product inventory. This lean manufacturing approach allows suppliers to offer competitive pricing models for high-purity reference standards without compromising on margin or quality.
- Enhanced Supply Chain Reliability: Utilizing a process dependent on readily available hydrochloric acid and standard heating equipment mitigates the risk of supply disruptions caused by geopolitical issues affecting specialized reagent availability. The robustness of the reaction conditions, which tolerate a range of concentrations and temperatures, ensures consistent batch-to-batch reproducibility even when scaling up from grams to kilograms. This reliability is crucial for pharmaceutical companies that require a steady stream of reference materials for ongoing stability testing and regulatory filings, effectively reducing lead time for high-purity pharmaceutical intermediates needed for urgent quality investigations.
- Scalability and Environmental Compliance: The simplicity of the aqueous acidic medium facilitates easier waste treatment compared to organic solvent-heavy or metal-contaminated processes, aligning with increasingly stringent environmental regulations. The process generates primarily saline wastewater upon neutralization, which is far easier to treat than heavy metal effluents, thereby reducing the environmental compliance burden on manufacturing facilities. Additionally, the short reaction times of 1.5 to 4 hours allow for rapid turnover in reactor vessels, enhancing the throughput capacity of existing infrastructure and supporting the commercial scale-up of complex pharmaceutical intermediates to meet global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dehydration technology for Venlafaxine impurity production. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing quality control workflows.
Q: What is the primary advantage of the dehydration method described in CN114292198A?
A: The primary advantage is the simplicity and efficiency of the process, achieving yields up to 90% in a single step using common reagents like hydrochloric acid, which significantly simplifies quality control sample preparation.
Q: Why is the synthesis of specific Venlafaxine impurities critical for pharmaceutical manufacturers?
A: Synthesizing specific impurities is essential for establishing robust analytical methods and setting strict quality specifications, ensuring the safety and efficacy of the final Venlafaxine hydrochloride API and its formulations.
Q: Can this dehydration process be scaled for industrial reference standard production?
A: Yes, the process utilizes standard industrial reagents and moderate temperatures (80-100°C), making it highly suitable for batch production and commercial scale-up without requiring exotic catalysts or extreme conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Venlafaxine Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your pharmaceutical products depends on the availability of authentic and high-quality impurity standards. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether for early-stage R&D or full-scale QC operations. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay. By leveraging advanced synthetic strategies like the acid-catalyzed dehydration route, we provide a secure and efficient source for critical reference materials that support your regulatory compliance goals.
We invite you to contact our technical procurement team to discuss how our manufacturing capabilities can support your specific project needs. Request a Customized Cost-Saving Analysis today to understand how our optimized processes can reduce your overall expenditure on reference standards. We are ready to provide specific COA data and route feasibility assessments to demonstrate our commitment to being your trusted partner in pharmaceutical intermediate supply.
