Advanced Synthesis of 6-Nitrosaccharin: A Scalable Route for High-Purity Herbicide Intermediates
Advanced Synthesis of 6-Nitrosaccharin: A Scalable Route for High-Purity Herbicide Intermediates
The global demand for high-performance herbicides such as iodosulfuron methyl sodium and foramsulfuron has necessitated a rigorous re-evaluation of their key precursor, 6-nitrosaccharin. Traditional manufacturing pathways have long been plagued by severe environmental liabilities and complex purification challenges. However, the technological landscape has shifted dramatically with the disclosure of patent CN101735167A, which introduces a groundbreaking preparation method that fundamentally alters the economic and ecological footprint of this critical intermediate. This novel approach replaces hazardous chromium-based oxidation with a sophisticated sequence of esterification, diazotization, and cyclization, offering a pathway that is not only chemically elegant but also commercially robust. For R&D directors and procurement strategists alike, understanding the nuances of this patent is essential for securing a competitive edge in the agrochemical supply chain. The method leverages readily available starting materials like 2-amino-4-nitrobenzoic acid, ensuring that production costs are significantly optimized without compromising on the stringent purity specifications required for downstream pharmaceutical and agricultural applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 6-nitrosaccharin relied heavily on the oxidation of 4-nitrotoluene-2-sulphonamide using sodium dichromate, a process disclosed in older literature such as U.S. Pat 3158233. This conventional route is fraught with substantial operational hazards and environmental drawbacks that render it increasingly obsolete in the modern regulatory landscape. The use of hexavalent chromium compounds introduces severe toxicity risks, necessitating expensive and complex waste treatment protocols to remove heavy metal residues from the final product. Furthermore, the reaction conditions are often violently exothermic and difficult to control on a multi-ton scale, leading to inconsistent batch quality and potential safety incidents. From a supply chain perspective, the reliance on such hazardous reagents creates vulnerability to regulatory crackdowns and fluctuating raw material availability. The generation of large volumes of chromium-containing sludge not only inflates disposal costs but also poses long-term liability issues for manufacturing facilities, making this traditional method economically unsustainable for forward-thinking enterprises seeking to minimize their carbon and toxic footprints.
The Novel Approach
In stark contrast to the archaic oxidation methods, the technology outlined in patent CN101735167A presents a refined, three-step synthetic strategy that prioritizes safety, efficiency, and environmental compatibility. By shifting the synthetic entry point to 2-amino-4-nitrobenzoic acid, the process bypasses the need for aggressive oxidants entirely, instead utilizing a controlled esterification followed by a precise diazotization sequence. This strategic pivot allows for much milder reaction conditions, typically operating between 0°C and 40°C, which drastically reduces energy consumption and thermal runaway risks. The elimination of heavy metal catalysts means that the resulting 6-nitrosaccharin is inherently cleaner, requiring less intensive downstream purification to meet the rigorous standards of agrochemical and pharmaceutical clients. Moreover, the raw material 2-amino-4-nitrobenzoic acid is widely sourced and cost-effective, providing a stable foundation for long-term production planning. This novel approach effectively decouples production scalability from environmental risk, offering a sustainable solution that aligns perfectly with the green chemistry initiatives driving the modern fine chemical industry.
Mechanistic Insights into Esterification and Diazotization-Chlorosulfonylation
The core chemical innovation of this patent lies in the meticulous orchestration of the diazotization and subsequent chlorosulfonylation steps, which transform the amino ester into the crucial sulfonyl chloride intermediate. The process begins with the acid-catalyzed esterification of 2-amino-4-nitrobenzoic acid in methanol, where catalysts such as thionyl chloride, sulfuric acid, or hydrogen chloride gas facilitate the formation of methyl 2-amino-4-nitrobenzoate with high conversion rates. Following isolation, this ester undergoes diazotization in an acidic medium, typically hydrochloric acid, at a tightly controlled temperature range of 10°C to 15°C to stabilize the diazonium salt. The true mechanistic brilliance is observed in the subsequent addition of a glacial acetic acid solution containing copper chloride and sulfur dioxide. This step effectively converts the diazonium species into methyl 2-chlorosulfonyl-4-nitrobenzoate through a radical-mediated substitution mechanism that is far more selective than traditional chlorosulfonation with chlorosulfonic acid. The use of copper chloride as a mediator ensures that the sulfonyl chloride group is introduced ortho to the ester functionality with minimal formation of regio-isomers or tar byproducts.
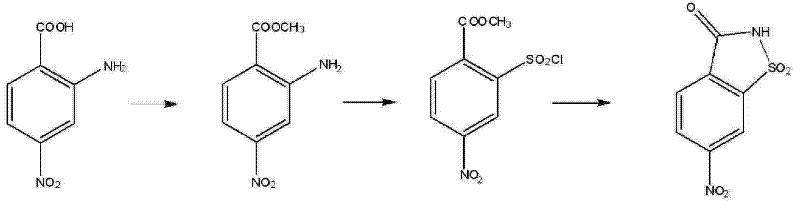
Following the formation of the chlorosulfonyl intermediate, the final cyclization step involves reacting this species with aqueous ammonia under mild thermal conditions of 20°C to 25°C. This ammonolysis reaction triggers an intramolecular nucleophilic attack where the nitrogen of the ammonia displaces the chlorine atom, simultaneously closing the sultam ring to form the saccharin backbone. The presence of the nitro group at the 6-position is preserved throughout this sequence, which is critical for the biological activity of the downstream herbicides. Impurity control is rigorously maintained by optimizing the pH during the workup; for instance, adjusting the solution to pH 1 with concentrated hydrochloric acid prior to crystallization ensures that unreacted amines and acidic byproducts remain in the aqueous phase while the neutral 6-nitrosaccharin precipitates as a high-purity solid. This mechanistic pathway not only delivers superior yields, reported up to 85% in the final step, but also ensures a consistent impurity profile that simplifies the quality control burden for manufacturers scaling this process from pilot plants to commercial reactors.
How to Synthesize 6-Nitrosaccharin Efficiently
Implementing this synthesis route requires precise adherence to the thermal and stoichiometric parameters defined in the patent to maximize yield and purity. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic cooling beyond standard industrial chillers. Operators must focus on the careful addition rates of reagents, particularly during the exothermic esterification and the sensitive diazotization phases, to prevent localized hot spots that could degrade the diazonium intermediate. The post-treatment procedures, involving solvent recovery and pH-controlled crystallization, are integral to achieving the reported HPLC purities of over 95%. For technical teams looking to adopt this methodology, the following guide outlines the standardized operational sequence derived directly from the patent examples, ensuring a reproducible and safe manufacturing protocol.
- Perform reflux esterification of 2-amino-4-nitrobenzoic acid in methanol using an acidic catalyst like thionyl chloride to obtain methyl 2-amino-4-nitrobenzoate.
- Conduct diazotization at 0-20°C followed by reaction with copper chloride and sulfur dioxide in acetic acid to generate methyl 2-chlorosulfonyl-4-nitrobenzoate.
- React the chlorosulfonyl intermediate with aqueous ammonia at 20-25°C to induce cyclization and form the final 6-nitrosaccharin product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route represents a strategic opportunity to de-risk the supply of critical agrochemical intermediates while driving down total landed costs. The shift away from chromium-based chemistry eliminates the need for costly heavy metal remediation and hazardous waste disposal services, which traditionally constitute a significant portion of the operational expenditure in fine chemical manufacturing. Furthermore, the reliance on commodity chemicals like methanol, hydrochloric acid, and 2-amino-4-nitrobenzoic acid insulates the production process from the volatility associated with specialized oxidants. This raw material flexibility ensures a more resilient supply chain capable of withstanding market fluctuations and logistical disruptions. By partnering with suppliers who utilize this advanced methodology, buyers can secure a more consistent flow of high-quality intermediates, reducing the risk of production stoppages due to quality failures or regulatory non-compliance.
- Cost Reduction in Manufacturing: The elimination of sodium dichromate and the associated waste treatment infrastructure leads to substantial cost savings in both raw material procurement and environmental compliance. The simplified workflow reduces labor hours and energy consumption, as the reactions proceed efficiently at near-ambient temperatures without the need for extreme heating or cooling. Additionally, the high yields achieved in each step minimize material loss, ensuring that the overall mass balance is optimized for maximum economic efficiency. These factors combine to lower the unit cost of production, allowing for more competitive pricing structures in the final herbicide markets without sacrificing margin.
- Enhanced Supply Chain Reliability: Sourcing 2-amino-4-nitrobenzoic acid is significantly more straightforward than procuring specialized chromium reagents, as it is a bulk chemical produced by numerous global vendors. This abundance reduces lead times and mitigates the risk of single-source dependency. The robustness of the synthetic route means that production campaigns can be extended with fewer interruptions for maintenance or cleaning, ensuring a steady output of 6-nitrosaccharin to meet seasonal demand spikes in the agricultural sector. Suppliers utilizing this method can offer more reliable delivery schedules, which is critical for just-in-time manufacturing models employed by major agrochemical corporations.
- Scalability and Environmental Compliance: The process is inherently scalable, having been validated from gram-scale laboratory experiments to multi-kilogram pilot runs with consistent results. The absence of toxic heavy metals simplifies the regulatory approval process for new manufacturing sites and facilitates easier expansion into regions with strict environmental laws. Waste streams are primarily organic and acidic, which are easier to treat and neutralize compared to heavy metal sludge. This environmental friendliness enhances the corporate social responsibility profile of the supply chain, aligning with the sustainability goals of end-user companies and reducing the likelihood of regulatory fines or shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 6-nitrosaccharin using this advanced methodology. These insights are derived from the specific experimental data and claims within the patent documentation, providing clarity on process capabilities and product specifications. Understanding these details is vital for technical evaluators assessing the feasibility of integrating this intermediate into their existing formulation pipelines.
Q: What are the primary advantages of this new synthesis method over traditional chromium oxidation?
A: The patented method eliminates the use of toxic sodium dichromate, significantly reducing environmental pollution and heavy metal waste treatment costs while utilizing cheaper, widely available raw materials like 2-amino-4-nitrobenzoic acid.
Q: How does the process ensure high purity for agrochemical applications?
A: By strictly controlling the diazotization temperature between 10°C and 15°C and using hydrochloric acid as the medium, the process minimizes side reactions, achieving yields up to 97% HPLC purity in the final step.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method features simple operational steps, mild reaction conditions (0-40°C), and avoids hazardous oxidants, making it highly scalable and compliant with modern industrial safety and environmental standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Nitrosaccharin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to cleaner, more efficient synthetic routes is paramount for the future of the agrochemical industry. Our technical team has extensively analyzed the methodology described in patent CN101735167A and possesses the expertise to implement this process at a commercial scale with precision and reliability. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and speed. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of 6-nitrosaccharin meets the exacting standards required for herbicide synthesis. We are committed to delivering a product that not only performs exceptionally in downstream reactions but also aligns with your sustainability and cost-efficiency targets.
We invite you to collaborate with us to optimize your supply chain for 6-nitrosaccharin and related intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to reach out to us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. Let us be your partner in driving innovation and efficiency in your chemical manufacturing operations.
