Advanced Manufacturing of 6-Nitrosaccharin: Technical Breakthroughs and Commercial Scalability
Advanced Manufacturing of 6-Nitrosaccharin: Technical Breakthroughs and Commercial Scalability
The global demand for high-performance herbicides such as iodosulfuron methyl sodium and foramsulfuron has necessitated a rigorous re-evaluation of their key precursor, 6-nitrosaccharin. Patent CN101735167B introduces a transformative preparation method that fundamentally alters the economic and environmental landscape of producing this critical agrochemical intermediate. Unlike legacy processes that rely on hazardous oxidation steps, this novel approach utilizes a streamlined sequence of esterification, diazotization, and cyclization starting from 2-amino-4-nitrobenzoic acid. For R&D directors and procurement strategists, this patent represents a pivotal shift towards greener chemistry, offering a pathway that not only mitigates regulatory risks associated with heavy metal waste but also leverages inexpensive, commercially abundant feedstocks to drive down the overall cost of goods sold. The technical robustness of this method ensures consistent quality, making it an ideal candidate for reliable agrochemical intermediate supplier partnerships aiming for long-term stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 6-nitrosaccharin has been plagued by significant environmental and operational hurdles, primarily exemplified by the method disclosed in U.S. Pat 3158233. This conventional route relies on the oxidation of 4-nitrotoluene-2-sulfonamide using sodium dichromate, a hexavalent chromium compound known for its extreme toxicity and carcinogenic properties. The reaction conditions are notoriously violent and difficult to control, often leading to safety incidents and requiring specialized, corrosion-resistant equipment that drives up capital expenditure. Furthermore, the generation of chromium-containing wastewater imposes a massive burden on downstream waste treatment facilities, forcing manufacturers to incur substantial costs for neutralization and disposal to meet increasingly stringent environmental regulations. These factors collectively render the traditional oxidation pathway economically inefficient and environmentally unsustainable for modern large-scale production, creating a pressing need for alternative synthetic strategies that can bypass these inherent liabilities.
The Novel Approach
In stark contrast, the methodology detailed in CN101735167B circumvents these issues by adopting a constructive synthetic strategy rather than a destructive oxidation one. By initiating the synthesis with the esterification of 2-amino-4-nitrobenzoic acid, the process establishes a stable foundation using a raw material that is widely sourced and cost-effective. The subsequent transformation involves a sophisticated diazotization followed by a chlorosulfonylation step mediated by a copper chloride-sulfur dioxide system, which operates under mild acidic conditions. This approach completely eliminates the need for toxic chromium oxidants, thereby removing the associated environmental hazards and simplifying the purification workflow. The final cyclization with ammonia water proceeds smoothly at moderate temperatures, yielding the target molecule with high purity. This paradigm shift not only enhances operator safety but also aligns perfectly with the principles of green chemistry, offering a sustainable solution for cost reduction in agrochemical intermediate manufacturing.
Mechanistic Insights into Esterification and Diazotization-Chlorosulfonylation
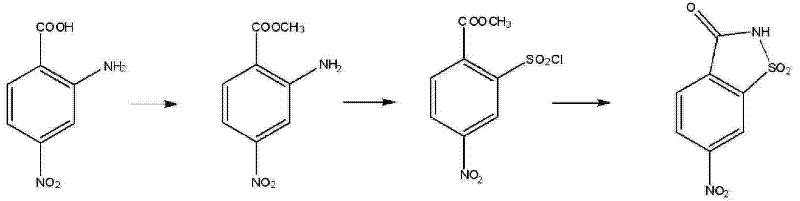
The core of this technical breakthrough lies in the precise orchestration of functional group transformations, beginning with the acid-catalyzed esterification of the carboxylic acid moiety. In this step, 2-amino-4-nitrobenzoic acid reacts with excess methanol in the presence of an acidic catalyst, with thionyl chloride proving to be the superior choice over sulfuric acid or hydrogen chloride gas due to its dual role as a dehydrating agent and catalyst. This reaction proceeds under reflux for approximately 8 to 12 hours, driving the equilibrium towards the formation of 2-amino-4-nitro methyl benzoate with impressive yields reaching up to 90%. The protection of the carboxylic acid as a methyl ester is crucial, as it prevents unwanted side reactions during the subsequent harsh acidic conditions of the diazotization phase, ensuring that the molecular integrity is maintained throughout the synthetic sequence.
Following esterification, the mechanism advances to a complex diazotization-chlorosulfonylation cascade that constructs the saccharin core. The amino group is first converted into a diazonium salt using sodium nitrite in a hydrochloric acid medium at a tightly controlled temperature range of 10°C to 15°C. Immediately thereafter, the introduction of a glacial acetic acid solution containing saturated cupric chloride and sulfur dioxide facilitates the replacement of the diazo group with a chlorosulfonyl functionality. This specific reagent system is critical for generating the 2-chlorosulfonyl-4-nitrobenzoic acid methyl ester intermediate, which serves as the direct precursor for ring closure. The final step involves nucleophilic attack by ammonia on the sulfonyl chloride group, triggering an intramolecular cyclization that forms the sultam ring structure of 6-nitrosaccharin. This mechanistic pathway is highly selective, minimizing the formation of by-products and ensuring a final product purity that meets rigorous pharmaceutical and agrochemical standards.
How to Synthesize 6-Nitrosaccharin Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters defined in the patent to maximize yield and purity. The process is divided into three distinct operational stages: the initial esterification to activate the substrate, the critical diazotization and sulfonylation sequence to install the reactive handle, and the final ammonolysis to close the ring. Each stage demands specific attention to temperature control and reagent stoichiometry; for instance, maintaining the diazotization temperature below 20°C is vital to prevent the decomposition of the unstable diazonium intermediate, while the final cyclization benefits from a slightly warmer range of 20°C to 25°C to ensure complete conversion without degrading the nitro group. Detailed standard operating procedures regarding reagent addition rates, workup protocols involving ethyl acetate extraction, and crystallization conditions are essential for reproducibility. The standardized synthesis steps outlined below provide a comprehensive guide for scaling this technology from the laboratory bench to pilot plant operations.
- Perform reflux esterification of 2-amino-4-nitrobenzoic acid in methanol using thionyl chloride as a catalyst to obtain 2-amino-4-nitro methyl benzoate.
- Conduct diazotization at 10-15°C with sodium nitrite and hydrochloric acid, followed by reaction with a copper chloride-sulfur dioxide acetic acid solution to form the chlorosulfonyl intermediate.
- React the chlorosulfonyl intermediate with aqueous ammonia at 20-25°C to induce cyclization and precipitate high-purity 6-nitrosaccharin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers profound strategic advantages that extend far beyond simple chemical yield. The most significant impact is observed in the drastic simplification of the supply chain risk profile; by shifting away from sodium dichromate, manufacturers eliminate the need for handling Class 1 carcinogens, thereby reducing insurance premiums, regulatory compliance costs, and the potential for supply disruptions caused by environmental shutdowns. Furthermore, the reliance on 2-amino-4-nitrobenzoic acid as the starting material leverages a commodity chemical market that is stable and competitive, insulating the production process from the volatility often seen with specialized oxidants. This transition enables a more resilient sourcing strategy, ensuring continuous production capability even in fluctuating market conditions.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive heavy metal waste treatment protocols and the utilization of low-cost, high-volume raw materials. By removing the sodium dichromate oxidation step, the facility saves significantly on the capital and operational expenditures associated with hazardous waste disposal and effluent treatment plants. Additionally, the high efficiency of the thionyl chloride catalyzed esterification reduces solvent consumption and energy usage during the reflux process, contributing to a leaner manufacturing cost structure. The overall process design minimizes unit operations, allowing for faster batch turnover and higher asset utilization rates without compromising on product quality.
- Enhanced Supply Chain Reliability: The switch to a non-toxic synthetic pathway dramatically improves the reliability of the supply chain by removing regulatory bottlenecks. Facilities no longer face the stringent permitting delays or transportation restrictions associated with shipping and storing large quantities of hexavalent chromium compounds. The raw material, 2-amino-4-nitrobenzoic acid, is produced by multiple global suppliers, ensuring a diversified sourcing base that mitigates the risk of single-supplier dependency. This robustness allows for more accurate lead time forecasting and strengthens the ability to meet just-in-time delivery commitments for downstream herbicide manufacturers.
- Scalability and Environmental Compliance: From a scalability perspective, the mild reaction conditions ranging from 0°C to 40°C make this process exceptionally easy to scale from kilogram to multi-tonne production without requiring exotic high-pressure or high-temperature reactors. The absence of heavy metal contaminants simplifies the purification process, often allowing for straightforward crystallization techniques that are easily automated. Environmentally, the process generates benign by-products that are easier to treat, aligning with corporate sustainability goals and facilitating smoother audits from international clients who demand green manufacturing credentials. This compliance advantage future-proofs the production line against tightening global environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 6-nitrosaccharin synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on yield optimization, safety protocols, and scalability factors. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements based on this superior manufacturing route.
Q: What are the primary advantages of this new synthesis route over traditional methods?
A: The patented method eliminates the use of toxic sodium dichromate oxidants found in conventional processes, significantly reducing environmental pollution and waste treatment costs while utilizing cheaper, widely available raw materials like 2-amino-4-nitrobenzoic acid.
Q: What are the critical temperature controls required for high yield?
A: Precise temperature control is essential; diazotization should be maintained between 10°C and 15°C, while the final cyclization with ammonia achieves optimal yield (85%) at 20°C to 25°C.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates under mild conditions (0-40°C) without high-pressure requirements, uses common solvents like methanol and ethyl acetate, and avoids hazardous heavy metal catalysts, making it highly scalable and safe for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Nitrosaccharin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just a technical upgrade but a strategic imperative for the modern chemical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature are faithfully translated into robust industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 6-nitrosaccharin meets the exacting standards required for herbicide synthesis. Our commitment to quality assurance means that our clients receive a product with a consistent impurity profile, minimizing variability in their downstream formulations.
We invite global partners to collaborate with us to leverage this advanced manufacturing technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this dichromate-free route for your operations. We encourage you to contact us today to discuss your volume requirements,索取 specific COA data for our current inventory, and review our comprehensive route feasibility assessments. Let us be your partner in achieving a more sustainable and cost-effective supply of high-purity 6-nitrosaccharin.
