Advanced Synthesis of Carboxyl-Substituted Resveratrol Analogs for Oncology and Cardiovascular Applications
The pharmaceutical industry continuously seeks structural modifications of natural products to enhance bioavailability and therapeutic potency, a challenge directly addressed by the technology disclosed in patent CN100368373C. This patent introduces a novel class of carboxyl-substituted resveratrol analogs, specifically designed to overcome the limitations of natural trans-resveratrol while retaining its potent anticancer and cardiovascular protective properties. The core innovation lies in the strategic introduction of a carboxylic acid group at the alpha-position of the stilbene double bond, creating compounds such as (E)-2-(3,5-dihydroxyphenyl)-3-(4-hydroxyphenyl)acrylic acid. These derivatives exhibit superior inhibition rates against lung carcinoma cells compared to the parent compound, as evidenced by IC50 values in the micromolar range. For R&D directors and procurement specialists, this represents a significant opportunity to access high-value intermediates for next-generation oncology and cardioprotective drug development. The synthesis route provided is not only chemically elegant but also industrially viable, utilizing standard organic transformations that can be readily scaled from laboratory benchtop to multi-ton commercial production facilities without requiring exotic catalysts or extreme pressure conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the supply of resveratrol and its derivatives has been heavily reliant on extraction from botanical sources such as grape skins, peanuts, or Polygonum cuspidatum, a method fraught with inherent supply chain vulnerabilities and quality inconsistencies. Natural extraction processes are subject to seasonal variations, geographical differences in plant chemistry, and the presence of complex impurity profiles that require extensive and costly downstream purification to meet pharmaceutical grade standards. Furthermore, the natural abundance of specific resveratrol analogs, particularly those with carboxyl substitutions, is virtually non-existent, making isolation impossible and necessitating total synthesis. Conventional synthetic routes often suffer from poor stereoselectivity, yielding mixtures of cis and trans isomers that complicate purification and reduce overall process efficiency. Additionally, many traditional methods rely on harsh reaction conditions or expensive transition metal catalysts that introduce heavy metal contamination risks, requiring additional remediation steps that drive up manufacturing costs and extend lead times for critical raw materials.
The Novel Approach
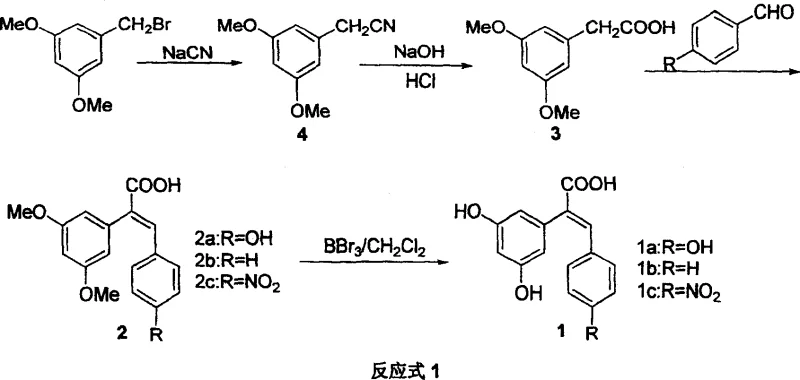
The methodology outlined in CN100368373C presents a robust alternative by employing a linear, four-step synthetic sequence that prioritizes high yield and operational simplicity. By starting from readily available 3,5-dimethoxybenzyl bromide, the process builds the carbon skeleton efficiently through a cyanation followed by hydrolysis, achieving exceptional conversion rates in the early stages. The key differentiator is the use of a Perkin condensation to construct the stilbene double bond, a classic reaction that is well-understood and easily controlled to favor the biologically active (E)-isomer. This approach eliminates the need for complex cross-coupling reactions that often require palladium catalysts, thereby simplifying the impurity profile and reducing the burden on quality control laboratories. The final demethylation step using boron tribromide is highly selective, ensuring that the sensitive carboxylic acid functionality remains intact while revealing the crucial phenolic hydroxyl groups required for biological activity. This streamlined workflow significantly reduces the number of unit operations compared to alternative convergent syntheses, translating directly into improved throughput and reduced solvent consumption for large-scale manufacturing.
Mechanistic Insights into Perkin Condensation and Demethylation
The heart of this synthetic strategy is the Perkin condensation reaction, which couples 3,5-dimethoxyphenylacetic acid with various para-substituted benzaldehydes to form the central stilbene framework. Mechanistically, this involves the generation of an enolate ion from the acetic acid derivative via deprotonation by potassium acetate in an acetic anhydride medium. This nucleophilic enolate attacks the carbonyl carbon of the benzaldehyde, followed by dehydration to establish the conjugated double bond system. The reaction conditions specified, heating to 40°C, are mild enough to prevent decomposition of sensitive functional groups yet sufficient to drive the equilibrium toward the product. The use of acetic anhydride serves a dual purpose as both solvent and dehydrating agent, pushing the reaction forward and minimizing the formation of side products. Following the condensation, the resulting dimethoxy-protected intermediate undergoes a critical demethylation step using boron tribromide (BBr3) in dichloromethane. This Lewis acid coordinates with the methoxy oxygen atoms, facilitating the cleavage of the methyl-oxygen bond to release methyl bromide and generate the free phenol. The reaction is typically conducted at room temperature over 24 to 36 hours, allowing for complete conversion without degrading the acrylic acid moiety, a testament to the chemoselectivity of the reagent system.
Understanding the impurity profile is crucial for regulatory compliance, and this route offers distinct advantages in controlling byproduct formation. The initial cyanation step proceeds with high specificity, minimizing the risk of dialkylation or elimination side reactions that could plague benzyl halide substitutions. During the hydrolysis of the nitrile to the carboxylic acid, the use of aqueous sodium hydroxide ensures complete conversion to the salt form, which is then precipitated upon acidification, effectively washing away neutral organic impurities. In the Perkin step, the primary impurity concern is the unreacted aldehyde or the self-condensation of the anhydride, both of which are easily removed during the recrystallization of the intermediate acid. The final demethylation generates boron-containing byproducts which are quenched into water-soluble boric acid species during the workup, allowing for easy separation from the organic product phase. This clean reaction profile ensures that the final API intermediate meets stringent purity specifications with minimal need for chromatographic purification, a key factor in reducing production costs.
How to Synthesize (E)-2-(3,5-dihydroxyphenyl)-3-(4-hydroxyphenyl)acrylic Acid Efficiently
The synthesis of these high-value resveratrol analogs follows a logical progression of functional group transformations that are amenable to standard chemical engineering practices. The process begins with the nucleophilic substitution of 3,5-dimethoxybenzyl bromide with sodium cyanide in an ethanol-water mixture, a reaction that exotherms moderately and requires careful temperature control around 65°C to maximize the 96% yield reported in the patent examples. Following isolation of the nitrile, hydrolysis is performed under basic reflux conditions, converting the cyano group into the corresponding carboxylate which is then acidified to precipitate the pure 3,5-dimethoxyphenylacetic acid. The subsequent Perkin condensation requires precise stoichiometry between the acid and the aldehyde component, typically using a slight excess of acetic anhydride to drive the reaction to completion within 2.5 hours. Finally, the deprotection step demands anhydrous conditions to prevent premature hydrolysis of the boron tribromide reagent, ensuring efficient demethylation to the final trihydroxy product. For detailed operational parameters and safety guidelines, please refer to the standardized protocol below.
- Perform nucleophilic substitution of 3,5-dimethoxybenzyl bromide with sodium cyanide to form the nitrile intermediate.
- Hydrolyze the nitrile intermediate using sodium hydroxide under reflux to obtain 3,5-dimethoxyphenylacetic acid.
- Conduct a Perkin condensation reaction between the acetic acid derivative and substituted benzaldehyde using acetic anhydride and potassium acetate.
- Execute demethylation using boron tribromide (BBr3) in dichloromethane to reveal the final polyphenolic structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits in terms of cost stability and supply security. The reliance on commodity chemicals such as sodium cyanide, acetic anhydride, and substituted benzaldehydes means that raw material sourcing is not dependent on volatile agricultural markets or single-source botanical suppliers. The high yields observed in the initial steps, particularly the near-quantitative conversion in the cyanation phase, drastically reduce the amount of starting material required per kilogram of final product, leading to substantial cost savings in raw material procurement. Furthermore, the elimination of precious metal catalysts removes a significant cost center associated with both the purchase of catalysts and the validation of heavy metal clearance, simplifying the regulatory dossier for downstream drug manufacturers. The robustness of the crystallization steps described in the patent allows for high recovery rates of intermediates, minimizing waste disposal costs and enhancing the overall atom economy of the process.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by utilizing high-yielding reactions that minimize raw material waste, particularly in the early stages where yields exceed 90%. By avoiding expensive transition metal catalysts and complex purification techniques like column chromatography, the overall cost of goods sold is drastically reduced compared to alternative cross-coupling methodologies. The use of common solvents like ethanol, methanol, and dichloromethane further ensures that solvent recovery and recycling can be implemented easily, lowering operational expenditures.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as all key reagents are bulk petrochemical derivatives available from multiple global suppliers, mitigating the risk of supply disruptions. The synthetic route is insensitive to minor fluctuations in reaction conditions, ensuring consistent batch quality and reducing the likelihood of failed batches that could delay delivery schedules. This reliability allows for accurate forecasting and inventory planning, essential for maintaining continuous production lines in the pharmaceutical sector.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate at atmospheric pressure, making the transfer from pilot plant to commercial scale straightforward without requiring specialized high-pressure equipment. The waste streams generated are primarily aqueous salts and organic solvents which can be treated using standard effluent treatment protocols, ensuring compliance with environmental regulations. The high selectivity of the demethylation step reduces the formation of hazardous byproducts, contributing to a greener manufacturing profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these carboxyl-substituted resveratrol analogs. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is critical for evaluating the feasibility of integrating these intermediates into your specific drug development pipelines.
Q: What are the primary advantages of this synthetic route over natural extraction?
A: Unlike natural extraction which suffers from seasonal variability and low concentrations in plant sources, this synthetic pathway described in CN100368373C offers consistent batch-to-batch reproducibility and significantly higher overall yields, particularly in the initial cyanation step which achieves up to 96% conversion.
Q: How is the stereochemistry controlled during the Perkin reaction?
A: The process utilizes specific reaction conditions, including heating to 40°C in acetic anhydride with potassium acetate, which favors the formation of the thermodynamically stable (E)-isomer (trans-configuration), ensuring the biological activity matches that of natural trans-resveratrol derivatives.
Q: Is the demethylation step scalable for industrial production?
A: Yes, the demethylation using BBr3 in dichloromethane at room temperature is a robust method widely used in fine chemical manufacturing. The patent demonstrates yields between 77% and 87% across different substituents, indicating a reliable process suitable for commercial scale-up with proper safety controls for handling boron tribromide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carboxyl-Substituted Resveratrol Analog Supplier
NINGBO INNO PHARMCHEM stands ready to support your R&D and commercialization efforts with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including the safe management of reagents like boron tribromide and sodium cyanide, ensuring that every batch meets stringent purity specifications. We understand that consistency is paramount in pharmaceutical manufacturing, which is why our rigorous QC labs employ advanced analytical techniques to verify the stereochemistry and impurity profile of every lot before shipment. By partnering with us, you gain access to a supply chain that is both resilient and cost-optimized, allowing you to focus on clinical development while we manage the complexities of chemical manufacturing.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are available to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your project timelines. Let us help you secure a reliable supply of these high-purity pharmaceutical intermediates and drive your oncology or cardiovascular programs forward with confidence.
