Revolutionizing Vitamin A Production: Advanced C-14 Aldehyde Synthesis and Commercial Scalability
Revolutionizing Vitamin A Production: Advanced C-14 Aldehyde Synthesis and Commercial Scalability
The global demand for Vitamin A and its derivatives continues to drive innovation in fine chemical manufacturing, particularly for key intermediates that dictate the efficiency of the entire value chain. Patent CN102190565B introduces a transformative methodology for synthesizing 2-methyl-4-(2,6,6-trimethyl-1-cyclohexen-1-yl)-2-butenal, widely known as C-14 aldehyde. This critical intermediate serves as the foundational building block for the industrial production of retinyl acetate and other vital retinoids. The disclosed technology leverages a sophisticated Wittig-Horner condensation strategy followed by a controlled acid-catalyzed hydrolysis, offering a distinct departure from legacy synthetic pathways. By optimizing reaction conditions between -40°C and 30°C and utilizing accessible precursors like beta-cyclocitral, this invention addresses long-standing challenges in yield optimization and impurity control. For R&D directors and procurement specialists, understanding this patent is crucial for evaluating next-generation supply partners capable of delivering high-purity pharmaceutical intermediates with enhanced cost structures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
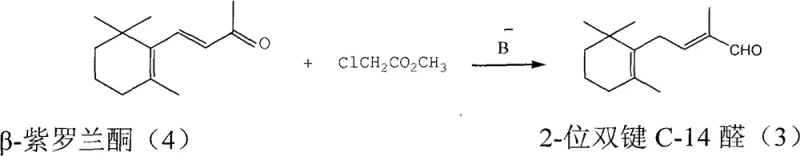
Historically, the industrial preparation of C-14 aldehyde has relied on chemically intensive and environmentally burdensome processes that pose significant risks to operational continuity. One prevalent legacy method involves the Darzens condensation of beta-ionone with methyl chloroacetate, a reaction that necessitates the use of excessive amounts of alkali and chloroacetate to drive conversion, leading to complex waste streams and difficult downstream processing. Another traditional pathway utilizes methyl sulfonium salts to generate terminal epoxides, which are subsequently opened using magnesium bromide; however, this route requires expensive methyl iodide and generates hazardous dimethyl sulfide byproducts alongside dangerous DMSO-sodium salt residues. These conventional approaches, as illustrated in older synthetic schemes involving multi-step functional group manipulations, often suffer from poor atom economy and require rigorous safety protocols to handle volatile sulfur compounds and strong bases. Furthermore, the classical Roche route involving Grignard reactions, hydrogenation, and multiple halogenation steps creates a lengthy linear sequence that amplifies material loss at every stage, making it economically unviable for modern cost-sensitive manufacturing environments.
The Novel Approach
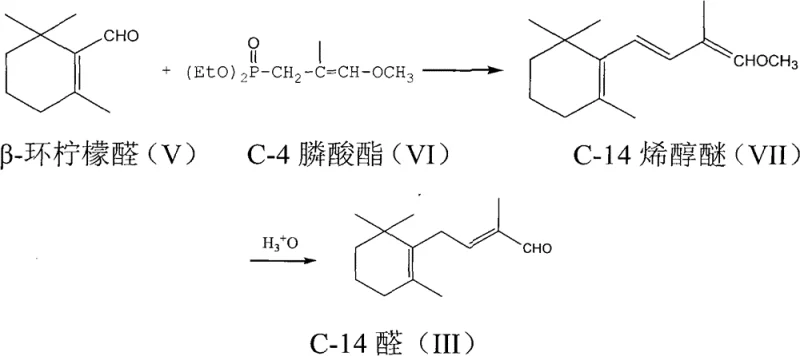
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in the patent streamlines the synthesis into a highly efficient sequence centered around a Wittig-Horner condensation. This method initiates with the rearrangement and dissociation of a C-4 phosphonate ester in the presence of a base within an ether or dipolar aprotic solvent, generating a reactive carbanion species under mild thermal conditions ranging from -40°C to 30°C. Upon the addition of beta-cyclocitral, the system undergoes condensation to form a C-14 enol ether intermediate with high selectivity, bypassing the need for hazardous sulfur reagents or excessive halogenated solvents. The subsequent hydrolysis of this enol ether using an acid catalyst not only reveals the aldehyde functionality but also facilitates a spontaneous migration of the double bond to the thermodynamically stable 2-position. This telescoped logic reduces the total number of unit operations, minimizes solvent consumption, and significantly lowers the E-factor of the process, representing a paradigm shift towards greener and more economical cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Wittig-Horner Condensation and Hydrolytic Isomerization
The core chemical innovation lies in the precise control of the Wittig-Horner reaction dynamics and the subsequent acid-catalyzed transformation. Initially, the C-4 phosphonate ester is treated with a strong base such as potassium tert-butoxide or sodium ethylate, which abstracts an acidic proton to generate a stabilized phosphonate carbanion. This nucleophile attacks the carbonyl carbon of beta-cyclocitral, forming a betaine intermediate that collapses to eliminate a phosphate species and establish the new carbon-carbon double bond, yielding the C-14 enol ether. Crucially, the reaction temperature is maintained between -40°C and 30°C to prevent side reactions and ensure the formation of the desired stereoisomer. Following isolation, the enol ether is subjected to hydrolysis using acids like p-toluenesulfonic acid or trifluoroacetic acid in a homogeneous solvent system containing water. During this hydrolytic cleavage of the enol ether moiety, the resulting allylic alcohol intermediate undergoes a rapid dehydration and double bond migration. This mechanistic feature is vital because it converts any minor 3-position double bond isomers directly into the target 2-position conjugated aldehyde system, effectively self-correcting stereochemical imperfections generated in the earlier condensation step.
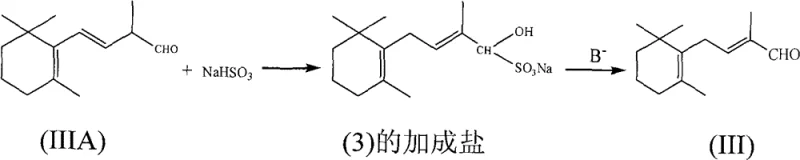
Furthermore, the patent discloses a sophisticated purification mechanism that leverages the chemistry of bisulfite adducts to achieve exceptional purity levels required for vitamin synthesis. The crude C-14 aldehyde, which may contain trace organic impurities and residual 3-position isomers, is treated with an aqueous solution of sodium bisulfite to form a water-soluble addition salt. This step selectively sequesters the aldehyde from non-carbonyl contaminants, which are removed via organic solvent extraction. Upon treating the aqueous layer with a base such as sodium carbonate, the bisulfite adduct dissociates to regenerate the free aldehyde. Remarkably, this basic dissociation environment also catalyzes the isomerization of any remaining 3-position double bonds to the more stable 2-position configuration. This dual-function purification step ensures that the final product meets stringent quality specifications without requiring energy-intensive distillation or chromatography, thereby enhancing the overall process robustness and reliability for a reliable pharma intermediates supplier.
How to Synthesize C-14 Aldehyde Efficiently
The synthesis of this high-value vitamin intermediate is designed for scalability, utilizing standard reactor configurations found in multipurpose fine chemical facilities. The process begins with the generation of the phosphonate anion under inert atmosphere, followed by the controlled addition of the cyclic aldehyde to manage exotherms and maintain stereocontrol. After the condensation is complete, the reaction mixture is quenched, and the organic phase containing the enol ether is separated and concentrated. The subsequent hydrolysis step is performed at near-ambient temperatures (10-35°C), minimizing energy input for heating or cooling. Finally, the innovative bisulfite purification protocol is applied to the crude aldehyde slurry to deliver the final specification material. For detailed operational parameters, stoichiometry, and safety guidelines, please refer to the standardized synthesis protocol below.
- Perform rearrangement dissociation of C-4 phosphonate with base in ether or dipolar aprotic solvent at -40 to 30°C.
- Add beta-cyclocitral to conduct Wittig-Horner condensation, yielding C-14 enol ether.
- Hydrolyze the enol ether with acid catalyst and water at 10-35°C, followed by bisulfite purification to obtain C-14 aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented methodology offers profound advantages in terms of cost structure and supply chain resilience. By eliminating the reliance on volatile sulfur compounds like dimethyl sulfide and expensive alkylating agents such as methyl iodide, manufacturers can drastically reduce raw material procurement costs and mitigate the risks associated with handling hazardous chemicals. The simplified two-step reaction sequence from beta-cyclocitral to the final aldehyde reduces the total processing time and equipment occupancy, allowing for higher throughput in existing production assets. Moreover, the use of common solvents like tetrahydrofuran and toluene, along with readily available inorganic bases, ensures that the supply chain is not vulnerable to bottlenecks associated with specialty reagents. This operational simplicity translates directly into substantial cost savings and improved margin potential for downstream vitamin producers.
- Cost Reduction in Manufacturing: The elimination of multi-step sequences involving Grignard reagents and halogenation significantly lowers the consumption of utilities and labor hours per kilogram of product. By avoiding the use of expensive methyl sulfonium salts and the associated waste treatment costs for sulfur-containing byproducts, the overall variable cost of goods sold is optimized. The high selectivity of the Wittig-Horner reaction minimizes the formation of difficult-to-separate impurities, reducing the load on purification units and increasing the effective yield of the process without requiring capital-intensive new equipment.
- Enhanced Supply Chain Reliability: The primary starting materials, beta-cyclocitral and phosphonate esters, are commoditized chemicals with stable global supply networks, reducing the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, ensures consistent batch-to-batch quality even when scaling up from pilot to commercial production. This reliability is critical for maintaining continuous inventory levels for high-purity pharmaceutical intermediates, ensuring that downstream customers face no disruption in their own vitamin A manufacturing schedules.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional Darzens or epoxide routes, simplifying effluent treatment and lowering environmental compliance costs. The absence of heavy metals and persistent organic pollutants aligns with increasingly strict global environmental regulations, facilitating easier permitting for commercial scale-up of complex pharmaceutical intermediates. Additionally, the mild reaction conditions reduce the energy footprint of the plant, contributing to sustainability goals while maintaining high production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on reaction mechanisms, purification strategies, and scalability factors. Understanding these details is essential for technical teams evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: How does the new Wittig-Horner route improve upon traditional Darzens condensation methods?
A: The novel route eliminates the need for excessive methyl chloroacetate and harsh alkaline conditions associated with Darzens condensation. It utilizes milder temperatures (-40 to 30°C) and avoids hazardous reagents like dimethyl sulfide, significantly enhancing operational safety and environmental compliance.
Q: What is the mechanism behind the high purity achieved in the final C-14 aldehyde product?
A: The process employs a unique purification strategy where the crude aldehyde forms a sodium bisulfite adduct. Subsequent dissociation with base not only isolates the aldehyde but also catalyzes the isomerization of the less stable 3-position double bond to the thermodynamically favored 2-position, ensuring high stereochemical purity.
Q: Are the raw materials for this synthesis readily available for large-scale production?
A: Yes, the key starting materials, beta-cyclocitral and C-4 phosphonate, are commercially accessible or can be synthesized via established literature methods. This availability supports robust supply chain continuity and reduces dependency on exotic or expensive reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C-14 Aldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of essential vitamins and nutraceuticals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated Wittig-Horner chemistry described in CN102190565B can be translated into robust industrial reality. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced GC-MS and HPLC capabilities to guarantee that every batch of C-14 aldehyde meets the exacting standards required for vitamin A synthesis. Our commitment to process excellence means we can deliver this key intermediate with consistent quality, supporting your long-term production goals.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized manufacturing protocols, we can help you reduce total landed costs while securing a stable supply of this vital building block. Please contact our technical procurement team to request specific COA data, route feasibility assessments, and samples for your qualification process. Let us partner with you to enhance the efficiency and sustainability of your vitamin A value chain.
