Advanced Manufacturing of Vitamin A Intermediate C-14 Aldehyde via Novel Wittig-Horner Condensation
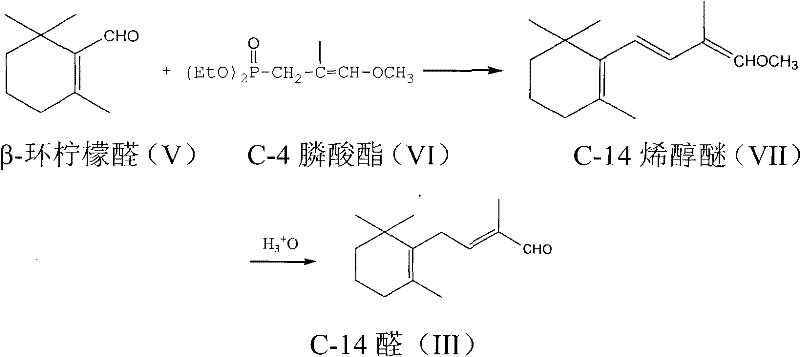
The pharmaceutical and nutraceutical industries continuously seek robust synthetic pathways for essential vitamins, and Patent CN102190565A presents a significant breakthrough in the manufacturing of Vitamin A intermediates. This intellectual property discloses a highly efficient method for preparing 2-methyl-4-(2,6,6-trimethyl-1-cyclohexen-1-yl)-2-butenal, commonly known as C-14 aldehyde or tetradecanal, which serves as a critical building block in the C14+C6 synthesis route for Vitamin A acetate. The innovation lies in a streamlined two-step sequence that begins with a Wittig-Horner condensation between beta-cyclocitral and a specific C-4 phosphonate ester, followed by a controlled acid-catalyzed hydrolysis. This approach fundamentally shifts the paradigm from older, more hazardous methodologies to a safer, more cost-effective process that leverages widely available starting materials. By optimizing reaction conditions to operate within a moderate temperature range of -40°C to 30°C and utilizing standard ether or dipolar aprotic solvents, the technology offers a compelling solution for manufacturers aiming to enhance their supply chain reliability for high-purity vitamin intermediates while minimizing environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of C-14 aldehyde has been plagued by significant technical and economic hurdles that hinder efficient large-scale production. Traditional Method A, often relying on Darzens condensation as illustrated in prior art, necessitates the use of greatly excessive amounts of methyl chloroacetate and strong alkaline condensing agents, leading to substantial waste generation and difficult downstream processing. Alternatively, Method B involves the formation of terminal epoxides using methylsulfonium salts and expensive methyl iodide, followed by ring-opening with magnesium bromide. This older pathway is particularly problematic because it relies on hazardous reagents like dimethyl sulfide and dangerous strong bases such as DMSO sodium salts, which pose severe safety risks and complicate regulatory compliance for modern chemical plants. Furthermore, these conventional routes often struggle with selectivity issues, requiring complex purification steps to separate unwanted isomers, thereby driving up the overall cost of goods sold and extending lead times for procurement teams seeking reliable sources of this key intermediate.
The Novel Approach
In stark contrast to these legacy processes, the novel methodology described in the patent introduces a sophisticated yet operationally simple strategy that bypasses the need for hazardous epoxidation or excessive halogenated reagents. The core of this innovation is the direct coupling of beta-cyclocitral with a C-4 phosphonate derivative through a Wittig-Horner condensation reaction, which proceeds with high efficiency under inert gas protection. This reaction pathway allows for precise control over the formation of the carbon-carbon double bond, yielding a C-14 enol ether intermediate that can be subsequently hydrolyzed to the target aldehyde. The elegance of this route is further enhanced by its compatibility with standard industrial solvents like tetrahydrofuran and dimethyl sulfoxide, eliminating the need for specialized or exotic reaction media. By adopting this modern synthetic logic, manufacturers can achieve a drastic simplification of the process flow, resulting in a more sustainable production cycle that aligns perfectly with the growing demand for green chemistry practices in the fine chemical sector.
Mechanistic Insights into Wittig-Horner Condensation and Isomerization
The mechanistic foundation of this synthesis rests on the precise generation of a reactive carbanion species from the C-4 phosphonate precursor, which acts as the nucleophile in the condensation step. Under the influence of strong organic bases such as potassium tert-butoxide or sodium ethoxide, the phosphonate undergoes a rearrangement dissociation reaction to form the corresponding carbanion, a critical intermediate that must be stabilized within the reaction matrix at temperatures preferably between -20°C and 10°C. Once formed, this carbanion attacks the carbonyl group of the beta-cyclocitral, initiating the Wittig-Horner coupling that constructs the extended carbon skeleton required for the vitamin A side chain. The reaction kinetics are carefully managed to ensure complete dissociation of the phosphonate before the addition of the aldehyde, a procedural nuance that maximizes yield and minimizes the formation of side products. This level of mechanistic control is essential for R&D directors focused on impurity profiling, as it ensures that the resulting C-14 enol ether possesses the correct stereochemistry necessary for the subsequent conversion to the biologically active all-trans vitamin A structure.
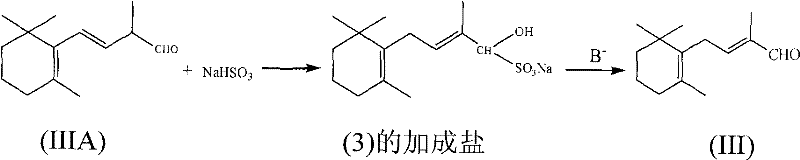
Beyond the initial coupling, the patent reveals a fascinating and highly valuable purification mechanism that simultaneously achieves chemical purification and stereochemical correction. During the final stage, the crude C-14 aldehyde, which may contain a mixture of 2-position and 3-position double bond isomers, is treated with an aqueous solution of sodium bisulfite. This treatment forms a water-soluble bisulfite adduct, effectively separating the aldehyde from non-carbonyl organic impurities which remain in the organic phase. More importantly, the formation of this adduct appears to provide the thermodynamic driving force required to shift the less stable 3-position double bond isomer into the desired 2-position configuration. Upon subsequent treatment with a base like sodium carbonate, the pure 2-position double bond C-14 aldehyde is regenerated. This dual-function step eliminates the need for energy-intensive distillation or chromatographic separation to remove isomers, representing a significant advancement in process chemistry that directly translates to higher overall yields and reduced production costs.
How to Synthesize C-14 Aldehyde Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly regarding temperature control and stoichiometry, to ensure optimal conversion rates and product quality. The process begins with the preparation of the phosphonate carbanion in a dry, inert atmosphere, followed by the controlled addition of beta-cyclocitral to maintain the exothermic nature of the condensation within safe limits. Following the isolation of the enol ether intermediate, the hydrolysis step utilizes mild acid catalysts such as p-toluenesulfonic acid or trifluoroacetic acid in a homogeneous solvent system to cleave the enol ether linkage. The detailed standardized synthetic steps for implementing this protocol in a pilot or commercial plant setting are outlined below.
- Perform a rearrangement dissociation of C-4 phosphonate with a strong base in an ether or dipolar aprotic solvent at temperatures between -40°C and 30°C.
- Add beta-cyclocitral to the reaction mixture to undergo Wittig-Horner condensation, yielding the C-14 enol ether intermediate.
- Hydrolyze the C-14 enol ether using an acid catalyst and water, followed by purification via sodium bisulfite adduct formation to isolate pure C-14 aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers tangible benefits that extend far beyond simple chemical yield improvements. The primary advantage lies in the substantial cost reduction in vitamin A intermediate manufacturing achieved by eliminating the reliance on expensive and volatile raw materials like methyl iodide and chloroacetate. By switching to beta-cyclocitral and phosphonate esters, which are produced on a massive scale for the fragrance and flavor industries, companies can secure a more stable and predictable pricing structure for their key inputs. Furthermore, the removal of hazardous reagents simplifies the handling requirements and reduces the need for specialized containment equipment, thereby lowering capital expenditure requirements for facility upgrades. This shift not only improves the bottom line but also mitigates supply chain risks associated with the availability of niche chemical reagents, ensuring continuous production even during periods of market volatility.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of costly purification steps and the reduction of waste disposal fees associated with halogenated byproducts. Since the bisulfite purification method effectively removes impurities and corrects isomers in a single operation, the need for multiple distillation runs or chromatographic columns is removed, leading to significant savings in energy consumption and solvent usage. Additionally, the higher atom economy of the Wittig-Horner reaction compared to the Darzens condensation means that a greater proportion of the starting material mass ends up in the final product, directly improving the material cost efficiency per kilogram of C-14 aldehyde produced.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved because the key starting material, beta-cyclocitral, is a commodity chemical with a robust global supply network, unlike the specialized sulfonium salts required in older methods. This abundance ensures that manufacturers are not held hostage by single-source suppliers for critical reagents, allowing for greater flexibility in vendor selection and negotiation. The simplified process flow also means that production cycles are shorter, enabling facilities to respond more rapidly to fluctuations in market demand for vitamin A and its derivatives, thus reducing the risk of stockouts and ensuring timely delivery to downstream pharmaceutical customers.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this route is superior because it avoids the generation of sulfur-containing waste streams and reduces the overall volume of organic solvents required per unit of product. The mild reaction conditions, operating near ambient temperatures rather than extreme heat or cryogenic cold, reduce the energy load on plant utilities and lower the carbon footprint of the manufacturing process. These factors make the technology highly scalable, allowing for seamless transition from laboratory bench scale to multi-ton commercial production without the need for complex engineering modifications, ensuring that the supply of high-purity intermediates can grow in lockstep with the expanding global nutraceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this C-14 aldehyde synthesis technology. These answers are derived directly from the experimental data and process descriptions found within the patent documentation, providing clarity on reaction conditions, purification mechanisms, and scalability potential. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this C-14 aldehyde synthesis method over traditional Darzens condensation?
A: Unlike the traditional Darzens condensation which requires a large excess of methyl chloroacetate and harsh alkaline conditions, this novel Wittig-Horner route utilizes readily available beta-cyclocitral and operates under milder temperature conditions ranging from -40°C to 30°C, significantly simplifying the operational complexity and reducing waste generation.
Q: How does the purification process ensure the correct double bond isomerization?
A: The process employs a unique purification step where the crude aldehyde reacts with sodium bisulfite to form an adduct. This step not only removes organic impurities but also drives the thermodynamic isomerization of the less stable 3-position double bond to the desired 2-position double bond, ensuring high stereochemical purity without requiring complex chromatographic separation.
Q: Is this synthetic route suitable for large-scale commercial production of vitamin A intermediates?
A: Yes, the route is highly amenable to scale-up because it avoids the use of expensive and hazardous reagents like methyl iodide or dangerous strong bases such as DMSO sodium salts found in older epoxide methods. The use of standard solvents like tetrahydrofuran and common acid catalysts allows for straightforward implementation in existing chemical manufacturing infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C-14 Aldehyde Supplier
As the global demand for high-quality vitamins continues to rise, partnering with a manufacturer who understands the intricacies of advanced intermediate synthesis is paramount for success. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in complex organic synthesis to deliver C-14 aldehyde and other critical vitamin precursors with unmatched consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of multinational corporations. We operate state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of C-14 aldehyde meets the exacting standards required for the synthesis of pharmaceutical-grade Vitamin A.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through the adoption of superior synthetic technologies. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating exactly how our efficient processes can improve your margins. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will secure your position as a leader in the competitive vitamin and nutraceutical marketplace.
