Scalable Synthesis of Racemic Clopidogrel via Palladium-Catalyzed Ortho-Chlorination
The pharmaceutical industry continuously seeks robust and economically viable pathways for producing critical cardiovascular therapeutics. Patent CN111606923A introduces a transformative synthetic methodology for racemic clopidogrel, a pivotal antiplatelet agent widely used in preventing thrombotic events. This innovation addresses long-standing inefficiencies in prior art by leveraging a palladium-catalyzed direct ortho-chlorination strategy, bypassing the need for hazardous or prohibitively expensive precursors. By utilizing methyl benzoylformate as a stable and accessible starting material, the process establishes a new benchmark for efficiency in pharmaceutical intermediates manufacturing. The technical breakthrough lies not only in the novel catalytic system but also in the seamless integration of reductive amination and acid-catalyzed cyclization, offering a streamlined route that significantly enhances process controllability and safety profiles for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of clopidogrel has been plagued by reliance on unstable or costly reagents that complicate scale-up and increase operational risks. Traditional routes often necessitate the use of tetrahydrothieno[3,2-c]pyridine or its hydrochloride salt, which are characterized by low melting points and high market prices, thereby inflating the overall cost of goods sold. Furthermore, alternative methodologies involving benzenesulfonyl chloride introduce significant safety hazards due to the release of irritating hydrogen chloride gas upon moisture absorption, requiring stringent containment measures. These legacy processes often suffer from multi-step sequences with poor atom economy and difficult purification protocols, leading to inconsistent batch quality and extended production cycles. For procurement teams, these factors translate into volatile pricing and unreliable delivery schedules, creating bottlenecks in the manufacture of essential cardiovascular medications.
The Novel Approach
In stark contrast, the methodology disclosed in CN111606923A revolutionizes the synthesis by employing a direct C-H activation strategy that simplifies the molecular construction of the core scaffold. This approach utilizes methyl benzoylformate, a commercially abundant and stable ketone ester, which undergoes selective ortho-chlorination under mild conditions mediated by a palladium catalyst and a specialized trifluoromethylaniline ligand. The subsequent introduction of the thienopyridine moiety is achieved through a cost-effective reductive amination with 2-thiopheneethylamine, completely eliminating the need for expensive cyclic amine salts. This strategic shift not only drastically reduces raw material costs but also improves the environmental footprint by avoiding hazardous sulfonyl chlorides. The result is a robust, three-step sequence that delivers high-purity intermediates with excellent isolation properties, positioning this technology as a superior choice for cost reduction in API manufacturing.
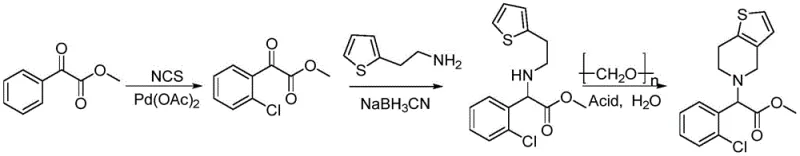
Mechanistic Insights into Pd-Catalyzed Ortho-Chlorination and Cyclization
The cornerstone of this synthetic advancement is the palladium-catalyzed ortho-chlorination of methyl benzoylformate, a reaction that exemplifies modern C-H functionalization chemistry. In this mechanism, the carbonyl group of the benzoylformate acts as a directing group, coordinating with the palladium acetate catalyst to facilitate the activation of the adjacent ortho C-H bond. The presence of N-chlorosuccinimide (NCS) serves as the chlorinating agent, while the bulky 3,5-bis(trifluoromethyl)aniline ligand plays a critical role in modulating the electronic environment of the metal center, enhancing both regioselectivity and turnover frequency. Trifluoroacetic acid is employed to promote the formation of an imine intermediate, further accelerating the chlorination kinetics. This precise control over the catalytic cycle ensures that the reaction proceeds with high fidelity at temperatures ranging from 30 to 120°C, minimizing side reactions and maximizing the yield of the crucial methyl o-chlorobenzoylformate intermediate.
Following the chlorination, the pathway proceeds through a reductive amination and a Pictet-Spengler-type cyclization, both of which are optimized for impurity control. The reductive amination utilizes sodium cyanoborohydride under mildly acidic conditions to couple the chlorinated ester with 2-thiopheneethylamine, forming a stable amino ester linkage without affecting the sensitive ester functionality. The final cyclization step employs paraformaldehyde as a formaldehyde source in an aqueous acidic medium, which hydrolyzes to generate the reactive electrophile in situ. This promotes the condensation with the secondary amine and subsequent ring closure to form the tetrahydrothieno[3,2-c]pyridine core. The use of aqueous acid not only drives the equilibrium towards the product but also facilitates the removal of basic impurities during workup. This mechanistic elegance ensures that the final high-purity racemic clopidogrel is obtained with minimal byproduct formation, simplifying downstream purification and ensuring compliance with stringent regulatory standards.
How to Synthesize Racemic Clopidogrel Efficiently
The implementation of this synthetic route requires careful attention to reaction parameters to maximize the reported yields of 65%, 82%, and 93% across the three respective steps. The process begins with the preparation of the chlorinated intermediate, followed by the coupling with the thiophene amine, and concludes with the acid-mediated ring closure. Each stage benefits from standard laboratory equipment and does not require exotic high-pressure reactors, making it highly adaptable for pilot and production scales. For detailed operational parameters, including specific solvent ratios, temperature ramps, and workup procedures, operators should refer to the standardized protocol below which encapsulates the critical process controls identified in the patent literature.
- Perform ortho-chlorination of methyl benzoylformate using NCS and Pd(OAc)2 with a trifluoromethylaniline ligand.
- Conduct reductive amination with 2-thiopheneethylamine using sodium cyanoborohydride to form the amino ester intermediate.
- Execute cyclization with paraformaldehyde in acidic aqueous solution to yield racemic clopidogrel.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages that directly address the pain points of procurement managers and supply chain directors. By substituting expensive and unstable precursors with commodity chemicals like methyl benzoylformate and 2-thiopheneethylamine, the process fundamentally alters the cost structure of production. The elimination of hazardous reagents such as benzenesulfonyl chloride reduces the need for specialized waste treatment and safety infrastructure, leading to substantial operational expenditure savings. Furthermore, the mild reaction conditions and high selectivity minimize the generation of complex impurity profiles, which streamlines quality control and reduces the time required for batch release. These factors collectively contribute to a more resilient and cost-efficient supply chain for pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the replacement of high-cost starting materials with readily available bulk chemicals. Traditional routes relying on tetrahydrothieno[3,2-c]pyridine hydrochloride incur significant raw material expenses due to the complexity of synthesizing the cyclic amine salt. By constructing the ring system in situ from linear precursors, this method bypasses those costs entirely. Additionally, the high yield of the final cyclization step (up to 93%) ensures that material throughput is maximized, reducing the cost per kilogram of the final active ingredient. The avoidance of expensive transition metal scavengers, thanks to the efficient catalytic system, further contributes to the overall economic viability of the process.
- Enhanced Supply Chain Reliability: Supply continuity is critical for life-saving medications, and this route enhances reliability by utilizing raw materials with broad global availability. Methyl benzoylformate and 2-thiopheneethylamine are produced by multiple chemical manufacturers worldwide, mitigating the risk of single-source dependency that often plagues specialized intermediates. The stability of these starting materials also allows for longer storage times and easier logistics management, reducing the risk of spoilage during transit. Consequently, manufacturers can maintain leaner inventory levels while ensuring consistent production schedules, effectively reducing lead time for high-purity pharmaceutical intermediates and safeguarding against market volatility.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, featuring reaction conditions that are easily managed in large-scale reactors. The use of aqueous acid in the final step simplifies waste streams compared to organic-solvent-heavy alternatives, aligning with modern green chemistry principles. The ability to isolate intermediates with high purity at each stage allows for flexible manufacturing strategies, such as campaign production of the chlorinated ester for other applications. This scalability, combined with reduced hazardous waste generation, ensures that production facilities can meet increasingly stringent environmental regulations without compromising output capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of racemic clopidogrel via this novel pathway. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on yield expectations, raw material sourcing, and process safety. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this technology for their own manufacturing portfolios.
Q: What are the key advantages of this Pd-catalyzed route over traditional methods?
A: This method avoids the use of expensive and unstable tetrahydrothieno[3,2-c]pyridine hydrochloride or benzenesulfonyl chloride, utilizing cheaper starting materials like methyl benzoylformate and 2-thiopheneethylamine for significant cost reduction.
Q: What is the overall yield of the synthetic process described in CN111606923A?
A: The patent reports an overall yield of approximately 50%, with individual step yields reaching up to 65% for chlorination, 82% for amination, and 93% for cyclization.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild and easy to control (30-120°C), and the intermediates can be effectively separated, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Racemic Clopidogrel Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for cardiovascular therapeutics like racemic clopidogrel. Our team of expert chemists has extensively evaluated the Pd-catalyzed ortho-chlorination pathway and confirmed its potential for robust industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the highest international standards for pharmaceutical intermediates.
We invite global partners to collaborate with us to leverage this advanced synthetic technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your production costs and secure a reliable supply of high-quality clopidogrel intermediates for the global market.
