Advanced Synthetic Route for Racemic Clopidogrel: Enhancing Cost Efficiency and Supply Stability
Advanced Synthetic Route for Racemic Clopidogrel: Enhancing Cost Efficiency and Supply Stability
The global demand for antithrombotic agents remains robust, with clopidogrel standing as a cornerstone therapy for preventing cardiovascular events. However, the manufacturing landscape for this critical active pharmaceutical ingredient (API) has long been challenged by complex synthetic routes that rely on expensive, unstable, or difficult-to-handle precursors. A significant technological breakthrough is detailed in patent CN111606923B, which discloses a novel, three-step synthetic methodology for producing racemic clopidogrel. This innovative approach fundamentally reimagines the construction of the thienopyridine core by starting from readily available methyl benzoylformate rather than pre-formed cyclic amines. By leveraging a palladium-catalyzed ortho-chlorination strategy followed by reductive amination and a final acid-catalyzed cyclization, this process offers a compelling alternative for manufacturers seeking to optimize their supply chains. The technical elegance of this route lies not only in its chemical efficiency but also in its ability to bypass the logistical bottlenecks associated with traditional raw materials, positioning it as a highly attractive option for any reliable pharmaceutical intermediates supplier aiming to secure long-term production continuity.
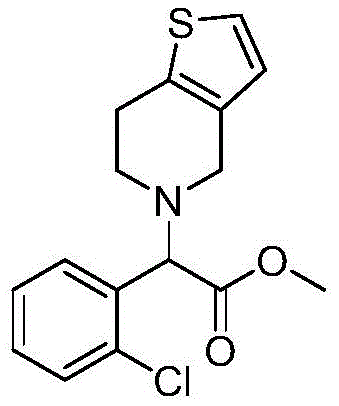
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of clopidogrel has been dominated by routes that introduce the thienopyridine moiety early in the sequence or rely on the coupling of complex fragments. Prominent prior art, such as the processes described in Sanofi patents US4529596 and US5036156, often necessitates the use of alpha-halogenated o-chlorophenyl methyl acetate reacting with specific thiophene derivatives. A major drawback of these legacy methods is the dependency on tetrahydrothieno[3,2-c]pyridine or its hydrochloride salt as a starting material. This cyclic amine is not only costly due to its own multi-step synthesis but also presents significant handling challenges; it is a low-melting solid that can be hygroscopic and difficult to purify on a multi-ton scale. Furthermore, alternative routes disclosed in patents like CN101845050 require the introduction of a benzenesulfonyl protecting group using benzenesulfonyl chloride. This reagent is notoriously unstable upon exposure to moisture, releasing irritating hydrogen chloride gas and posing severe occupational health and safety risks in a plant environment. These conventional pathways inherently inflate the cost of goods sold (COGS) and introduce unnecessary volatility into the supply chain, making cost reduction in API manufacturing a persistent struggle for procurement teams.
The Novel Approach
In stark contrast to the fragmented and reagent-heavy strategies of the past, the methodology outlined in CN111606923B constructs the molecule through a logical, linear assembly that maximizes atom economy and minimizes hazard. The process initiates with the direct functionalization of methyl benzoylformate, a stable and inexpensive commodity chemical. Instead of purchasing a pre-made cyclic amine, the synthesis builds the necessary carbon-nitrogen framework in situ. The second step employs 2-thiopheneethylamine, a vastly more accessible and affordable building block compared to tetrahydrothienopyridine derivatives. By deferring the formation of the final bicyclic ring system until the last step via a cyclization with paraformaldehyde, the process avoids the storage and stability issues associated with sensitive intermediates. This strategic shift allows for better control over impurity profiles at each stage, as the intermediates are distinct and separable. For a procurement manager, this translates to a dramatic simplification of the raw material portfolio, reducing dependency on niche suppliers and mitigating the risk of supply disruptions caused by the scarcity of specialized cyclic amines.
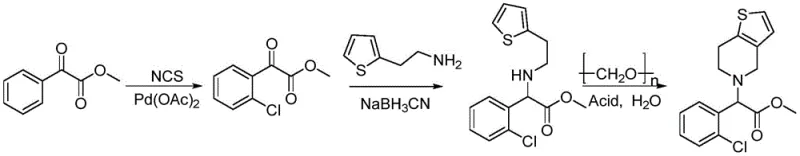
Mechanistic Insights into Pd-Catalyzed Ortho-Chlorination and Cyclization
The cornerstone of this synthetic innovation is the first step: a palladium-catalyzed directed ortho-chlorination. In this transformation, methyl benzoylformate serves as the substrate, reacting with N-chlorosuccinimide (NCS) as the chlorinating source. The reaction is mediated by palladium acetate (Pd(OAc)2) at a loading of 10-20 mol%, working in synergy with a specialized ligand system. The patent specifies the use of polysubstituted trifluoromethylanilines, such as 3,5-bis(trifluoromethyl)aniline, which act as electron-deficient ligands to modulate the electronic properties of the palladium center. Trifluoroacetic acid (TFA) plays a dual role here; it acts as a solvent component and promotes the formation of an imine intermediate between the ketone of the benzoylformate and the aniline ligand. This transient imine species acts as a directing group, coordinating the palladium catalyst to the ortho-position of the aromatic ring, thereby facilitating the selective C-H activation and subsequent chlorination. This mechanism ensures high regioselectivity, yielding methyl o-chlorobenzoylformate with minimal formation of meta- or para-isomers, which is critical for maintaining the purity required for downstream pharmaceutical applications.
Following the chlorination, the synthesis proceeds through a reductive amination and a final Pictet-Spengler-type cyclization. In the second step, the newly installed chloro-ketone reacts with 2-thiopheneethylamine in the presence of sodium cyanoborohydride (NaBH3CN) and acetic acid. This reductive amination efficiently links the thiophene side chain to the chiral center precursor without affecting the ester functionality. The final step is particularly elegant from a process chemistry perspective: the linear amine intermediate is treated with paraformaldehyde in an acidic aqueous solution (e.g., dilute HCl). Under heated conditions (70-90°C), the paraformaldehyde depolymerizes to generate formaldehyde in situ, which reacts with the secondary amine to form an iminium ion. This electrophilic species then undergoes an intramolecular electrophilic aromatic substitution on the thiophene ring, closing the six-membered piperidine ring to form the thienopyridine core of racemic clopidogrel. This acid-catalyzed cyclization in water is not only environmentally friendlier than organic solvent-heavy alternatives but also drives the reaction to completion through the thermodynamic stability of the newly formed aromatic-fused system.
How to Synthesize Racemic Clopidogrel Efficiently
The execution of this synthetic route requires precise control over reaction parameters to maximize the reported overall yield of approximately 50%. The process begins with the careful preparation of the chlorination mixture, ensuring the molar ratio of NCS to substrate is maintained between 1:1 and 3:1 to prevent over-chlorination. Following the isolation of the chloro-intermediate, the reductive amination is conducted at ambient temperatures (15-35°C), which significantly reduces energy consumption compared to cryogenic methods often used in similar transformations. The final cyclization step utilizes common industrial paraformaldehyde and dilute mineral acids, avoiding the need for exotic reagents. For R&D teams looking to implement this technology, the detailed standardized synthesis steps are provided below to ensure reproducibility and adherence to the patented conditions.
- Perform ortho-chlorination of methyl benzoylformate using NCS and Pd(OAc)2 with a trifluoromethylaniline ligand to obtain methyl o-chlorobenzoylformate.
- Conduct reductive amination between the chlorinated intermediate and 2-thiopheneethylamine using sodium cyanoborohydride.
- Execute cyclization with paraformaldehyde in an acidic aqueous solution to form the thienopyridine ring system of racemic clopidogrel.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits that extend far beyond the laboratory bench. For organizations focused on cost reduction in pharmaceutical intermediates manufacturing, the substitution of high-cost cyclic amines with commodity chemicals represents a direct and substantial decrease in raw material expenditure. The elimination of benzenesulfonyl chloride further removes the costs associated with specialized corrosion-resistant equipment and hazardous waste disposal, streamlining the operational overhead. Moreover, the use of stable, non-hygroscopic starting materials simplifies warehouse management and reduces the risk of material degradation during storage, ensuring a consistent quality of input for production batches. This stability is a key factor in enhancing supply chain reliability, as it decouples production schedules from the volatile availability of niche reagents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of expensive, multi-step precursors like tetrahydrothieno[3,2-c]pyridine hydrochloride with inexpensive, bulk-available 2-thiopheneethylamine and paraformaldehyde. By constructing the complex heterocyclic core in the final step, the process avoids the value loss associated with carrying expensive structural motifs through multiple low-yielding transformations. Additionally, the mild reaction conditions, particularly the room temperature reductive amination, lower the utility costs related to heating and cooling, contributing to a leaner manufacturing budget without compromising on the stringent purity specifications required for API intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on chemically stable and widely sourced raw materials. Unlike the moisture-sensitive reagents used in conventional routes, methyl benzoylformate and N-chlorosuccinimide possess excellent shelf lives and are produced by multiple global vendors, mitigating the risk of single-source dependency. The robustness of the intermediates allows for flexible inventory management, enabling manufacturers to stockpile key precursors during favorable market conditions. This flexibility ensures continuous production capability even during periods of global logistics disruption, providing a strategic advantage for reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex heterocycles, utilizing solvents like dichloroethane and water which are manageable in large-scale reactors. The final cyclization step occurring in an acidic aqueous medium simplifies the workup procedure, potentially reducing the volume of organic solvent waste generated per kilogram of product. The avoidance of gaseous HCl generation (from sulfonyl chlorides) and the use of solid paraformaldehyde instead of gaseous formaldehyde solutions greatly improves the environmental, health, and safety (EHS) profile of the plant. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the burden on waste treatment facilities, supporting sustainable long-term operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the technology for potential partners and licensees.
Q: What are the primary cost drivers eliminated in this new clopidogrel synthesis route?
A: This route eliminates the need for expensive and unstable tetrahydrothieno[3,2-c]pyridine hydrochloride or benzenesulfonyl chloride, replacing them with commodity chemicals like 2-thiopheneethylamine and paraformaldehyde.
Q: How does the Pd-catalyzed chlorination step improve process safety?
A: By utilizing N-chlorosuccinimide (NCS) under mild thermal conditions (30-120°C) with a directing ligand, the process avoids the use of hazardous gaseous chlorine or highly corrosive sulfuryl chloride typically found in traditional electrophilic aromatic substitutions.
Q: Is this synthetic method suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights mild reaction conditions, easy-to-control parameters, and an overall yield approaching 50%, making it highly viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Racemic Clopidogrel Supplier
The synthetic pathway detailed in CN111606923B represents a paradigm shift in the production of cardiovascular drug intermediates, offering a blend of chemical ingenuity and economic pragmatism. At NINGBO INNO PHARMCHEM, we recognize the transformative potential of such technologies and have positioned ourselves at the forefront of their industrial application. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of racemic clopidogrel or its key intermediates meets the exacting standards of the global pharmaceutical industry.
We invite forward-thinking pharmaceutical companies and generic drug manufacturers to collaborate with us to leverage this cost-effective synthesis. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in Pd-catalyzed transformations and heterocycle synthesis can drive value and security into your supply chain.
