Advanced Manufacturing of Dexlansoprazole Intermediates for Global Pharmaceutical Supply Chains
Advanced Manufacturing of Dexlansoprazole Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical landscape for gastroesophageal reflux disease (GERD) treatment continues to evolve, with dexlansoprazole standing out as a superior proton pump inhibitor due to its dual-release pharmacokinetic profile. Patent CN102108077B introduces a transformative synthetic methodology that addresses long-standing challenges in the production of this high-value active pharmaceutical ingredient (API). Unlike traditional routes that struggle with impurity management and optical purity, this novel approach utilizes a strategic late-stage substitution of trifluoroethanol on a chiral chloro-pyridyl intermediate. This technical breakthrough not only simplifies the operational workflow but also ensures a higher degree of stereochemical integrity, which is critical for meeting the stringent regulatory standards required for global generic and branded drug markets.
For R&D directors and process chemists, the significance of this patent lies in its ability to decouple the asymmetric oxidation step from the etherification step. Conventional wisdom dictated installing the trifluoroethoxy group early in the synthesis, but this often led to uncontrollable over-oxidation. By reversing the order of operations, the inventors have created a pathway that is inherently more robust and less prone to generating hard-to-remove byproducts. This shift represents a paradigm change in how we approach the synthesis of complex benzimidazole derivatives, offering a clearer path from bench-scale discovery to commercial manufacturing without the baggage of legacy purification bottlenecks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of dexlansoprazole has relied heavily on either resolution of racemic mixtures or direct asymmetric oxidation of the fully substituted trifluoroethoxy precursor. The resolution methods, while chemically straightforward, suffer from a theoretical maximum yield of only 50%, making them economically inefficient and wasteful for large-scale production. Furthermore, the direct asymmetric oxidation of the trifluoroethoxy-containing thioether is plagued by the formation of sulfone analogues. These sulfone impurities possess physicochemical properties remarkably similar to the desired sulfoxide, rendering standard recrystallization techniques ineffective and necessitating the use of preparative chromatography, which is cost-prohibitive and difficult to scale.
Additionally, the presence of the electron-withdrawing trifluoroethoxy group during the oxidation phase can complicate the reaction kinetics, often requiring precise control of transition metal catalysts like vanadium to suppress N-oxide formation. Even with these measures, the removal of trace metal residues and sulfone byproducts adds multiple unit operations to the process flow. This complexity increases the overall cycle time, reduces the overall throughput, and introduces significant variability in batch-to-batch consistency, posing a serious risk to supply chain reliability for downstream formulation manufacturers who require consistent quality attributes.
The Novel Approach
The methodology disclosed in CN102108077B elegantly circumvents these issues by utilizing a chloro-substituted pyridine intermediate as the substrate for the critical asymmetric oxidation step. By performing the oxidation on the simpler chloro-thioether scaffold, the reaction proceeds with higher selectivity and significantly lower formation of sulfone impurities. Once the chiral sulfoxide center is established with high optical purity, the chlorine atom is subsequently displaced by trifluoroethanol under alkaline conditions. This sequence ensures that the delicate sulfoxide moiety is not subjected to harsh oxidative conditions in the presence of the trifluoroethoxy group, thereby preserving the stereochemical integrity of the molecule.
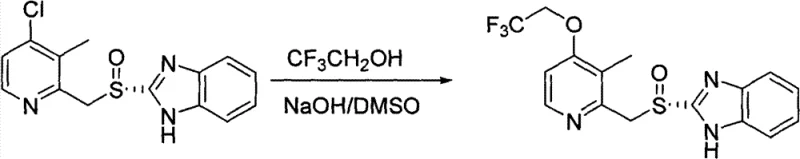
This strategic reordering of synthetic steps results in a much cleaner crude product profile, allowing for purification via simple crystallization rather than chromatography. The use of readily available reagents like sodium hydroxide and dimethyl sulfoxide for the substitution step further enhances the economic viability of the process. For procurement managers, this translates to a reduction in raw material costs and solvent consumption, while for supply chain heads, it means a more predictable and scalable manufacturing timeline. The ability to produce high-purity dexlansoprazole without the need for complex chiral resolution or extensive chromatographic purification marks a significant advancement in process chemistry efficiency.
Mechanistic Insights into Asymmetric Oxidation and Nucleophilic Substitution
The core of this synthetic strategy relies on a highly enantioselective oxidation mediated by a titanium-tartrate complex, followed by a nucleophilic aromatic substitution. In the first stage, the achiral sulfide precursor is treated with titanium tetraisopropoxide and a chiral ligand, typically diethyl L-(+)-tartrate, to form a chiral catalyst complex in situ. Upon the addition of an oxidant such as cumene hydroperoxide at low temperatures, oxygen is transferred to the sulfur atom with high facial selectivity. The steric environment created by the tartrate ligand dictates the approach of the oxidant, ensuring the formation of the desired (R)-enantiomer of the sulfoxide with minimal formation of the (S)-enantiomer or the over-oxidized sulfone.

Following the successful establishment of the chiral center, the second mechanistic phase involves the displacement of the 4-chloro group on the pyridine ring. The electron-deficient nature of the pyridine ring, activated by the adjacent nitrogen atom and the newly formed sulfoxide group, facilitates a nucleophilic attack by the trifluoroethoxide anion. Under basic conditions provided by sodium hydroxide in a polar aprotic solvent like DMSO, the trifluoroethanol is deprotonated to form a potent nucleophile. This species attacks the C-4 position of the pyridine ring, displacing the chloride leaving group through an addition-elimination mechanism. This step is crucial as it installs the pharmacophore responsible for the drug's metabolic stability and acid suppression efficacy without disturbing the sensitive chiral sulfoxide bond.
How to Synthesize Dexlansoprazole Efficiently
The synthesis of dexlansoprazole via this novel route requires careful attention to temperature control and reagent stoichiometry to maximize yield and optical purity. The process begins with the preparation of the chiral sulfoxide intermediate, where maintaining low temperatures during the addition of the oxidant is critical to preventing racemization and over-oxidation. Once the intermediate is isolated, the subsequent substitution reaction requires anhydrous conditions and a strong base to drive the equilibrium towards the product. Detailed standard operating procedures for each unit operation, including quenching, extraction, and crystallization, are essential for reproducibility.
- Perform asymmetric oxidation on 2-[(4-chloro-3-methyl-2-pyridyl)methyl]thio]benzimidazole using titanium tetraisopropoxide, diethyl tartrate, and cumene hydroperoxide to generate the chiral sulfoxide intermediate.
- React the resulting chiral chloro-sulfoxide intermediate with trifluoroethanol in the presence of a strong base like sodium hydroxide in a polar aprotic solvent such as DMSO.
- Isolate the final dexlansoprazole product through aqueous workup and crystallization, avoiding complex chromatographic purification steps required by conventional methods.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for organizations seeking to optimize their API sourcing strategies. The elimination of chromatographic purification steps is perhaps the most significant cost driver, as column chromatography is notoriously expensive in terms of silica gel consumption, solvent usage, and labor hours. By replacing this with crystallization, the process becomes amenable to continuous manufacturing and large-batch processing, drastically reducing the cost of goods sold (COGS). This efficiency gain allows suppliers to offer more competitive pricing without compromising on the quality or purity of the final active ingredient.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive transition metal catalysts like vanadium, which are often required in conventional asymmetric oxidation methods to suppress impurities. Furthermore, the avoidance of column chromatography removes a major bottleneck in production capacity, leading to substantial savings in operational expenditures. The use of commodity chemicals such as sodium hydroxide and common solvents like toluene and DMSO ensures that raw material costs remain stable and predictable, shielding the supply chain from volatility associated with specialty reagents.
- Enhanced Supply Chain Reliability: By simplifying the synthetic route and reducing the number of purification steps, the overall lead time for production is significantly shortened. This agility allows manufacturers to respond more rapidly to fluctuations in market demand, ensuring a steady supply of dexlansoprazole intermediates for downstream formulation. The robustness of the chloro-intermediate oxidation step also means fewer failed batches and less waste, contributing to a more reliable and consistent supply stream for pharmaceutical partners who cannot afford interruptions in their production schedules.
- Scalability and Environmental Compliance: The process is inherently greener due to the reduced solvent intensity and the absence of heavy metal catalysts that require complex removal and disposal protocols. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The ability to scale this reaction from kilogram to multi-ton quantities without losing efficiency makes it an ideal candidate for commercial manufacturing, providing a secure foundation for long-term supply agreements and reducing the risk of technology transfer failures between pilot and production plants.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel dexlansoprazole synthesis method. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's potential impact on your supply chain. Understanding these nuances is essential for making informed decisions about vendor qualification and process adoption.
Q: How does this new method improve impurity profiles compared to traditional asymmetric oxidation?
A: By delaying the introduction of the trifluoroethoxy group until after the oxidation step, this method significantly reduces the formation of sulfone analogues, which are notoriously difficult to separate from the target sulfoxide in conventional routes.
Q: Is column chromatography required for purification in this process?
A: No, the process is designed to allow for purification via crystallization and standard extraction techniques, eliminating the need for expensive and non-scalable column chromatography often needed to remove sulfone impurities in prior art.
Q: What are the scalability advantages of using the chloro-intermediate route?
A: The chloro-intermediate is more stable and easier to handle during the critical oxidation step, allowing for better control over stereoselectivity and yield without the steric and electronic complications introduced by the bulky trifluoroethoxy group.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexlansoprazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert process chemists has extensively evaluated the route described in CN102108077B and possesses the technical capability to implement this efficient strategy at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this innovative technology for your dexlansoprazole projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can drive value and efficiency for your organization.
