Advanced Synthetic Route for Key Penem Antibiotic Intermediate 4AA via L-Threonine
The global demand for carbapenem and penem antibiotics continues to surge, driving the need for efficient, scalable synthesis of their key intermediates. Patent CN102827199A introduces a groundbreaking synthetic method for producing (3R,4R)-3-[(1R)-tert-butyldimethylsiloxyethyl]-4-acetoxy-2-azetidinone, commonly known as 4AA. Unlike traditional routes that rely on expensive or scarce starting materials, this innovation utilizes L-threonine as the primary chiral pool source. The process encompasses eight distinct chemical transformations, including diazotization, nucleophilic substitution, and a critical beta-lactam ring closure, ultimately delivering a product with exceptional liquid phase purity of 99.34%. This technical breakthrough represents a significant leap forward for manufacturers seeking a reliable pharmaceutical intermediate supplier capable of delivering high-quality building blocks for next-generation antibacterial drugs.
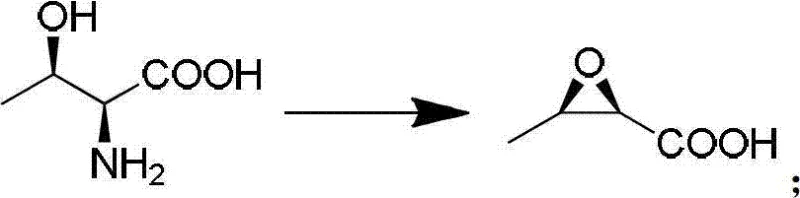
Historically, the manufacturing landscape for 4AA has been constrained by three primary synthetic pathways, each plagued by distinct logistical and economic inefficiencies. The first conventional method employs chiral 3-hydroxybutyric acid, a route characterized by notoriously low total yields and prohibitive costs that erode profit margins. The second approach utilizes chiral 1,3-butanediol; while technically viable, this raw material lacks robust domestic production capacity in many regions, forcing reliance on Japanese imports and creating fragile supply chains vulnerable to geopolitical shifts. The third pathway, starting from 6-APA, suffers from excessively high raw material costs, harsh reaction conditions that demand specialized equipment, and severe environmental pollution issues that complicate regulatory compliance. In stark contrast, the novel approach detailed in the patent leverages L-threonine, a bulk fermentation product that is inexpensive, widely available, and easy to handle. This shift not only simplifies the operational complexity but also aligns with green chemistry principles by reducing hazardous waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The legacy methods for synthesizing 4AA impose substantial burdens on both R&D budgets and supply chain stability. Routes dependent on 6-APA require aggressive reagents and generate significant toxic byproducts, necessitating costly waste treatment protocols that delay production timelines. Furthermore, the reliance on imported chiral 1,3-butanediol introduces unpredictable lead times and price volatility, making long-term procurement planning nearly impossible for large-scale API manufacturers. The low yields associated with the 3-hydroxybutyric acid route further exacerbate these issues, requiring larger reactor volumes and more raw material input to achieve the same output, thereby inflating the cost of goods sold. These cumulative factors render conventional methods increasingly obsolete in a competitive market that demands both economic efficiency and environmental stewardship.
The Novel Approach
The L-threonine-based synthesis route effectively dismantles these barriers by introducing a streamlined, eight-step sequence that operates under mild and controllable conditions. By initiating the synthesis with a readily available amino acid, the process bypasses the need for expensive chiral auxiliaries or imported diols. The reaction conditions are optimized for safety and scalability, with key steps such as diazotization occurring at manageable temperatures between -10°C and 0°C. Moreover, the protocol emphasizes solvent recovery, particularly for ethyl acetate and toluene, which drastically reduces material consumption and waste disposal costs. This holistic improvement in process design ensures that the manufacturing of high-purity pharmaceutical intermediates becomes not only technically feasible but also commercially sustainable for industrial applications.
Mechanistic Insights into Beta-Lactam Ring Cyclization
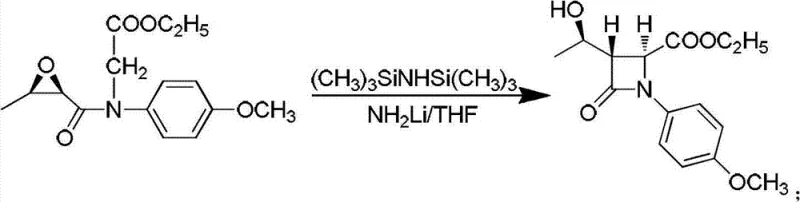
The cornerstone of this synthetic strategy lies in the formation of the four-membered beta-lactam ring, a structural motif essential for the biological activity of penem antibiotics. In Step 4 of the process, the condensation product (Intermediate III) undergoes a sophisticated cyclization reaction mediated by hexamethyldisilazane and lithium amide in toluene at -18°C. This specific combination of reagents facilitates an intramolecular nucleophilic attack that closes the ring with high stereoselectivity, preserving the crucial (3R,4R) configuration required for downstream antibiotic efficacy. The use of lithium amide as a strong base ensures complete deprotonation, while the low temperature suppresses side reactions such as epimerization or polymerization, which are common pitfalls in beta-lactam synthesis. This precise control over the reaction environment is critical for maintaining the optical purity of the intermediate.
Beyond the cyclization event, the entire pathway is engineered to minimize impurity profiles through careful selection of protecting groups and oxidation states. The initial conversion of L-threonine to the epoxy intermediate involves a diazotization followed by an intramolecular nucleophilic substitution, which sets the initial stereochemistry. Subsequent steps involve strategic protection of the hydroxyl group using tert-butyldimethylsilyl chloride and acetylation, which shield sensitive functionalities during the rigorous oxidative decarboxylation step. The final deprotection via ozonolysis cleanly removes the p-methoxyphenyl group without damaging the delicate beta-lactam core. This meticulous orchestration of functional group transformations ensures that the final 4AA product meets the stringent purity specifications of 99.34% HPLC purity, demonstrating superior impurity control compared to older methodologies.
How to Synthesize 4AA Efficiently
The synthesis of 4AA via this patented route is a multi-stage process that requires precise temperature control and stoichiometric management to maximize yield and purity. The procedure begins with the conversion of L-threonine into an epoxy intermediate, followed by coupling with p-methoxyanilinoethyl acetate to build the carbon skeleton. The subsequent cyclization, protection, hydrolysis, and oxidative decarboxylation steps transform this skeleton into the final beta-lactam structure. Each stage has been optimized in the patent examples to demonstrate reproducibility on a 50L pilot scale, providing a clear roadmap for technology transfer. For detailed operational parameters, reagent quantities, and workup procedures, please refer to the standardized synthesis guide below.
- Diazotization of L-Threonine with sodium nitrite and hydrochloric acid at -5°C to form the epoxy intermediate.
- Condensation of the epoxy intermediate with p-methoxyanilinoethyl acetate followed by cyclization using hexamethyldisilazane and lithium amide.
- Protection, hydrolysis, oxidative decarboxylation, and final ozonolysis deprotection to yield high-purity 4AA.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this L-threonine-based route offers transformative benefits that extend far beyond simple chemical yield. The primary advantage lies in the fundamental shift from scarce, imported specialty chemicals to abundant, fermentation-derived bulk commodities. This transition significantly mitigates supply risk, ensuring continuity of supply even during global logistics disruptions. Furthermore, the process design incorporates solvent recovery systems that allow for the reuse of organic solvents like toluene and ethyl acetate, leading to substantial cost savings in raw material procurement. The mild reaction conditions also reduce energy consumption and equipment wear, contributing to a lower overall cost of manufacturing while enhancing the sustainability profile of the production facility.
- Cost Reduction in Manufacturing: The elimination of expensive chiral starting materials like 6-APA or imported 1,3-butanediol results in a drastic reduction in direct material costs. By utilizing L-threonine, a commodity chemical produced at massive scale globally, manufacturers can stabilize their input costs and avoid the price volatility associated with niche fine chemicals. Additionally, the high yields reported in the patent examples, such as 96.5% for Intermediate II and 93.1% for the crude 4AA step, mean that less raw material is wasted per kilogram of final product. This efficiency translates directly into improved gross margins and a more competitive pricing structure for the final API.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for critical chiral intermediates is a major vulnerability in pharmaceutical supply chains. This new method diversifies the supply base by leveraging L-threonine, which is manufactured by numerous producers worldwide, thereby reducing the risk of shortages. The robustness of the synthesis, demonstrated by its successful execution in 50L reactors with consistent results, indicates high process reliability. This stability allows supply chain planners to forecast production schedules with greater confidence, reducing lead time for high-purity antibiotics and ensuring that downstream API manufacturing lines remain operational without interruption.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, featuring simple unit operations such as filtration, extraction, and distillation that are easily adapted to large-scale reactors. The ability to recover and recycle solvents not only lowers costs but also significantly reduces the volume of hazardous waste requiring disposal, simplifying environmental compliance. The use of milder reagents and lower temperatures compared to the 6-APA route minimizes the generation of toxic byproducts, aligning with increasingly strict environmental regulations. This makes the commercial scale-up of complex beta-lactams safer, cleaner, and more economically viable for long-term production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on purity, scalability, and raw material sourcing. Understanding these details is essential for R&D teams evaluating technology transfer and procurement teams assessing vendor capabilities.
Q: What are the advantages of using L-Threonine over 6-APA for 4AA synthesis?
A: L-Threonine is a readily available, cost-effective bulk amino acid, whereas 6-APA routes suffer from high raw material costs and harsh reaction conditions. The L-Threonine route offers milder conditions and better suitability for large-scale industrial production.
Q: What is the achieved purity of 4AA using this novel method?
A: The patented process achieves a liquid phase purity of up to 99.34% for the final 4AA product, meeting stringent specifications required for downstream antibiotic synthesis.
Q: Is this process suitable for commercial scale-up?
A: Yes, the process is designed for industrial production with simple operations, recyclable solvents, and the use of domestically available raw materials, eliminating supply chain bottlenecks associated with imported chiral diols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4AA Supplier
As the pharmaceutical industry moves towards more sustainable and cost-effective manufacturing paradigms, the ability to execute complex synthetic routes like the L-threonine to 4AA conversion becomes a key differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise temperature controls and inert atmosphere conditions required for beta-lactam synthesis, ensuring that every batch meets stringent purity specifications. With rigorous QC labs and a commitment to process optimization, we deliver high-purity pharmaceutical intermediates that empower our partners to bring life-saving antibiotics to market faster and more efficiently.
We invite you to collaborate with us to unlock the full potential of this advanced synthetic technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance your supply chain resilience and drive down your manufacturing costs.
