Advanced Diarylmethane URAT1 Inhibitors: Technical Breakthroughs and Commercial Scalability
Advanced Diarylmethane URAT1 Inhibitors: Technical Breakthroughs and Commercial Scalability
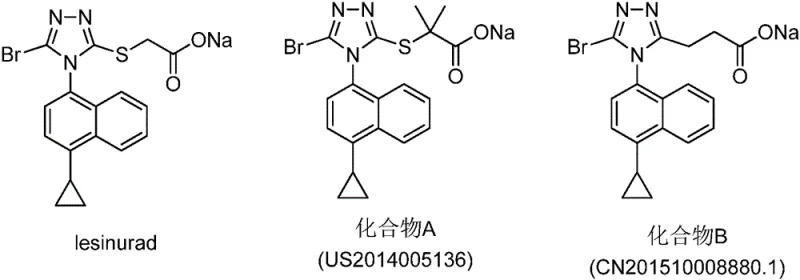
The pharmaceutical landscape for treating hyperuricemia and gout is undergoing a significant transformation driven by the need for safer and more effective Uric Acid Transporter 1 (URAT1) inhibitors. Patent CN107531649B introduces a novel class of carboxylic acid URAT1 inhibitors containing a diarylmethane structure, offering a robust alternative to existing therapies that often suffer from severe side effects or limited efficacy. This technology represents a critical advancement for R&D teams seeking to develop next-generation gout medications with improved safety profiles and enhanced pharmacokinetic properties. By leveraging a unique chemical scaffold that separates the triazole and naphthalene rings via a methylene or thioether linker, this invention addresses key structural limitations observed in earlier generations of URAT1 inhibitors. For global procurement and supply chain leaders, understanding the synthetic accessibility and scalability of these intermediates is paramount for securing a reliable pipeline of high-quality active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Current therapeutic options for managing gout, such as Lesinurad, have demonstrated clinical efficacy but are often accompanied by significant drawbacks that limit their long-term utility and patient compliance. Many existing URAT1 inhibitors feature structural motifs where the triazole and naphthalene rings are directly connected by covalent bonds, a design choice that can inadvertently lead to metabolic instability or off-target toxicity issues during prolonged treatment regimens. Furthermore, the synthesis of these conventional molecules frequently relies on complex coupling reactions that are difficult to control on a multi-ton scale, resulting in inconsistent impurity profiles and elevated production costs. The reliance on specific structural configurations also limits the ability to fine-tune physicochemical properties such as solubility and dissolution rates, which are critical for oral bioavailability. Consequently, there is a pressing demand within the industry for novel chemical entities that retain potent inhibitory activity while offering a more favorable safety and manufacturability profile.
The Novel Approach
The innovative strategy outlined in the patent data circumvents these historical challenges by introducing a flexible diarylmethane backbone that decouples the aromatic systems, thereby optimizing the spatial arrangement for URAT1 binding without compromising metabolic stability. This structural modification allows for extensive derivatization at multiple positions, enabling medicinal chemists to precisely modulate potency and selectivity through systematic structure-activity relationship studies. Unlike rigid prior art structures, this new scaffold accommodates various substituents such as halogens, alkyl groups, and cyano moieties, providing a versatile platform for developing a diverse library of drug candidates. From a manufacturing perspective, the synthetic routes designed for these compounds utilize robust reaction conditions that are amenable to process optimization and scale-up. This flexibility not only accelerates the drug discovery timeline but also ensures that the resulting intermediates can be produced consistently to meet stringent regulatory standards for purity and quality.
Mechanistic Insights into Diarylmethane-Based URAT1 Inhibition
The core innovation lies in the general formula (I) structure, which integrates a 1,2,4-triazole ring linked to a substituted naphthalene system through a methylene or thioether bridge, terminating in a carboxylic acid functional group. This specific arrangement is crucial for the molecule's ability to interact effectively with the uric acid transporter protein located in the renal proximal tubules. The carboxylic acid moiety plays a pivotal role in mimicking the uric acid substrate, facilitating competitive inhibition that prevents uric acid reabsorption and promotes its excretion in urine. Additionally, the presence of the triazole ring enhances the molecule's metabolic stability against oxidative degradation, a common failure point for many heterocyclic drugs. The versatility of the R groups allows for the fine-tuning of electronic and steric properties, ensuring that the inhibitor maintains high affinity for the target receptor even in the presence of physiological variations.
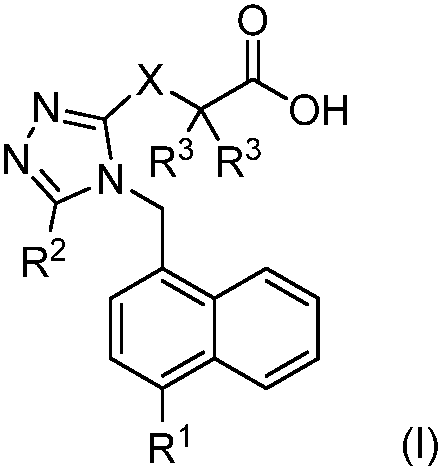
Impurity control is a critical aspect of the mechanistic design, as the synthetic pathway is engineered to minimize the formation of genotoxic impurities or difficult-to-remove byproducts. The stepwise construction of the triazole ring via cyclization of hydrazine derivatives ensures high regioselectivity, reducing the burden on downstream purification processes. Furthermore, the final hydrolysis steps are optimized to prevent racemization or decomposition of the sensitive carboxylic acid group, ensuring that the final active pharmaceutical ingredient meets rigorous chiral purity specifications. This attention to chemical detail translates directly into reduced risk during clinical development, as cleaner drug substances are less likely to exhibit unpredictable toxicological profiles. For quality assurance teams, this inherent process robustness provides a significant advantage in maintaining batch-to-batch consistency and complying with international pharmacopoeia standards.
How to Synthesize Diarylmethane URAT1 Inhibitors Efficiently
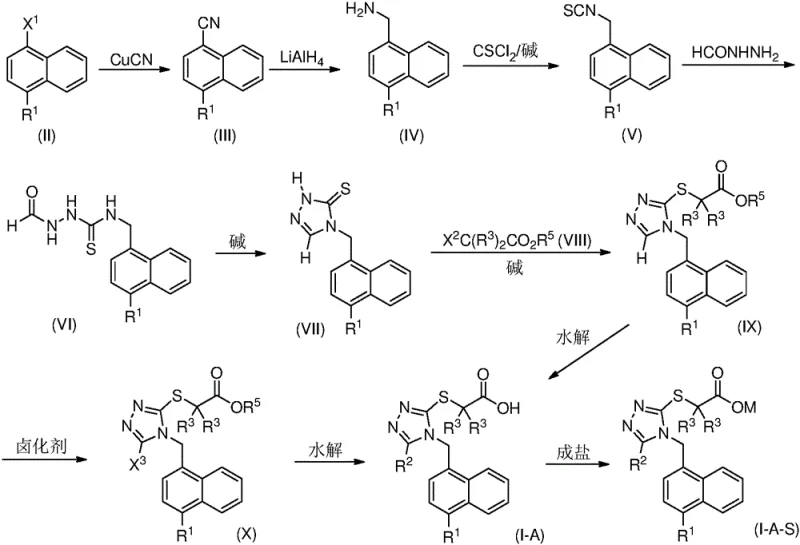
The synthesis of these high-value intermediates follows a logical and scalable sequence that begins with the functionalization of a commercially available naphthalene derivative. The process involves key transformations such as cyanation, reduction, and heterocyclic ring closure, all of which are performed using standard industrial reagents and equipment. The initial steps focus on installing the necessary amine functionality on the naphthalene core, which serves as the anchor for the subsequent triazole formation. This approach avoids the use of precious metal catalysts that often complicate supply chains and increase raw material costs. Instead, the route relies on abundant chemicals like copper cyanide and lithium aluminum hydride, which are readily accessible from global chemical suppliers. The final stages involve alkylation and hydrolysis to reveal the active carboxylic acid, completing the molecular architecture required for potent biological activity.
- Functionalize the naphthalene core via bromination and cyanation, followed by reduction to the aminomethyl intermediate.
- React the amine with thiophosgene and formylhydrazine to construct the 1,2,4-triazole-3-thione ring system.
- Alkylate the triazole nitrogen with a haloacetate ester, followed by halogenation and final hydrolysis to yield the target carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic technology offers substantial strategic benefits regarding cost efficiency and supply continuity. The elimination of complex transition metal catalysis steps significantly reduces the dependency on volatile precious metal markets, leading to more predictable raw material pricing and lower overall production costs. Moreover, the use of common solvents and reagents simplifies the sourcing process, allowing manufacturers to qualify multiple vendors for critical inputs and mitigate the risk of single-source supply disruptions. The robust nature of the reaction conditions also means that the process can be transferred between different manufacturing sites with minimal re-validation, enhancing operational flexibility. These factors collectively contribute to a more resilient supply chain capable of withstanding global market fluctuations and regulatory changes.
- Cost Reduction in Manufacturing: The streamlined synthetic route eliminates the need for expensive chromatographic purifications often required by less selective reactions, thereby reducing solvent consumption and waste disposal costs. By optimizing reaction yields and minimizing the formation of side products, the process achieves a higher mass efficiency, which directly translates to lower cost of goods sold. The ability to crystallize intermediates at various stages further enhances purity without resorting to costly preparative HPLC techniques. This economic efficiency makes the technology highly attractive for generic drug manufacturers seeking to enter the market with competitive pricing strategies while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than specialized custom synthons ensures that production schedules are not held hostage by the lead times of niche suppliers. This accessibility allows for larger batch sizes and longer campaign runs, which improves inventory management and reduces the frequency of changeover operations. Additionally, the stability of the intermediates allows for safe storage and transportation, facilitating a distributed manufacturing model where different steps can be performed at geographically diverse locations. This decentralization capability is crucial for mitigating regional risks and ensuring uninterrupted supply to global pharmaceutical partners.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing aqueous workups and recyclable solvents wherever possible to minimize the environmental footprint. The absence of heavy metal residues simplifies the waste treatment process, ensuring compliance with increasingly stringent environmental regulations regarding effluent discharge. Scalability is further supported by the exothermic nature of key reactions, which can be safely managed using standard cooling systems available in most multipurpose chemical plants. This alignment with sustainability goals not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel URAT1 inhibitors. These insights are derived directly from the patent specifications and are intended to clarify the feasibility of integrating this technology into existing drug development pipelines. Understanding these details is essential for stakeholders evaluating the potential for licensing or contract manufacturing partnerships. The answers reflect a commitment to transparency and technical accuracy, ensuring that all parties have a clear understanding of the capabilities and limitations of the described chemistry.
Q: What structural features enhance URAT1 inhibition in these compounds?
A: The patent highlights that the diarylmethane structure combined with a carboxylic acid moiety and a triazole ring significantly improves inhibitory activity compared to direct covalent links found in prior art like Lesinurad.
Q: Are the synthesis routes suitable for large-scale manufacturing?
A: Yes, the described pathways utilize commercially available starting materials and standard reagents like CuCN and LiAlH4, avoiding exotic catalysts that hinder scalability.
Q: How does this technology address solubility issues common in gout medications?
A: The introduction of the carboxylic acid group allows for the formation of pharmaceutically acceptable salts, such as sodium salts, which typically exhibit improved aqueous solubility and bioavailability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diarylmethane URAT1 Inhibitor Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise and infrastructure required to bring complex synthetic routes like this URAT1 inhibitor series to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from laboratory bench to industrial reactor. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation. This commitment to quality guarantees that every batch of intermediate we supply meets the exacting standards required for pharmaceutical registration and clinical trials.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements with a Customized Cost-Saving Analysis. Whether you need specific COA data for regulatory filings or detailed route feasibility assessments for process optimization, our experts are ready to provide the data-driven insights you need. Contact us today to secure a reliable supply of high-purity URAT1 inhibitors and accelerate your path to market with a partner dedicated to excellence and innovation.
