Advanced Composite Catalyst Technology for High-Purity MDA Manufacturing and Commercial Scale-Up
The global demand for high-performance polyurethanes and specialized pharmaceutical intermediates continues to drive innovation in the synthesis of diphenylmethane diamines (MDA). As a critical precursor for methylene diphenyl diisocyanate (MDI), the quality and cost-efficiency of MDA production are paramount for downstream manufacturers. A significant technological breakthrough in this sector is detailed in Chinese Patent CN108176392B, which introduces a novel composite catalyst capable of catalytically decomposing amine salts. This approach fundamentally shifts the paradigm from traditional alkali neutralization to a more sustainable catalytic decomposition process. By leveraging advanced nanomaterials and heterogeneous catalysis, this method addresses long-standing inefficiencies in MDA manufacturing, offering a pathway to superior product purity and reduced environmental impact. For industry leaders seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic advantages of this patent is crucial for strategic sourcing and process optimization.
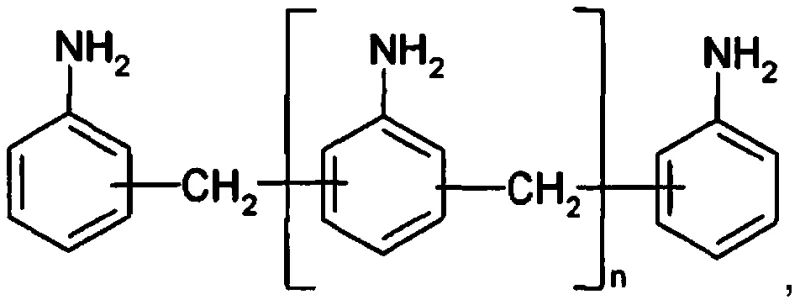
The structural versatility of MDA, as illustrated above, allows for the production of various isomers essential for tuning the properties of final polyurethane materials. However, accessing these structures efficiently has historically been hindered by cumbersome purification steps. The patent outlines a method where the amine hydrochloride salts, formed from the condensation of aniline and formaldehyde, are directly decomposed over a solid catalyst bed. This eliminates the need for liquid-liquid extraction involving massive volumes of brine, a common bottleneck in conventional facilities. The result is a streamlined workflow that aligns perfectly with the goals of modern commercial scale-up of complex pharmaceutical intermediates, ensuring that production capacity can be expanded without proportional increases in waste treatment infrastructure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional industrial processes for producing diphenylmethane diamines rely heavily on a neutralization step that has become increasingly problematic from both economic and environmental perspectives. In the standard workflow, the acidic reaction mixture containing aniline salts is treated with large quantities of caustic soda (sodium hydroxide) to liberate the free amine. This neutralization reaction typically occurs at elevated temperatures around 90°C to 100°C and generates stoichiometric amounts of sodium chloride salt. The presence of this inorganic salt creates severe challenges during phase separation, often leading to the formation of stable emulsions or impurity layers that are difficult to break. Consequently, the organic phase retains significant amounts of water and dissolved salts, necessitating energy-intensive distillation steps to dry the product. Furthermore, the disposal of the resulting saline wastewater places a heavy burden on environmental compliance systems, while the corrosive nature of the residual acidic salts poses risks to downstream equipment integrity if neutralization is incomplete.
The Novel Approach
In stark contrast, the technology disclosed in CN108176392B replaces the destructive neutralization step with a constructive catalytic decomposition. By passing the amine salt mixture through a fixed-bed reactor loaded with a specialized composite catalyst, the hydrochloride salts are thermally decomposed into free amines and hydrogen chloride gas. This gas-phase byproduct can be easily captured and recycled as hydrochloric acid for the initial salting reaction, effectively closing the acid loop. The absence of added alkali means no inorganic salts are generated within the reaction matrix, dramatically simplifying the subsequent phase separation. The organic phase emerging from this process contains significantly less water and virtually no suspended salt particles, which translates to reduced steam consumption during refining. This innovative route not only enhances the cost reduction in polyurethane precursor manufacturing but also delivers a product with a cleaner impurity profile, making it ideal for high-specification applications in the medical and automotive sectors.
Mechanistic Insights into Composite Catalyst Decomposition
The efficacy of this process hinges on the unique composition and preparation of the composite catalyst, which is engineered to maximize surface activity and thermal stability. The catalyst utilizes a nano-structured carrier, such as nano-SiO2 or multi-walled carbon nanotubes, which undergoes a rigorous dual-acid pretreatment. This pretreatment involves immersion in inorganic acid followed by reflux in a mixed acid system, a critical step that activates the carrier surface and enhances the adhesion of active metal species. The primary active components, typically selected from cerium oxide, tin oxide, or cuprous oxide, are dispersed as spherical nanoparticles on this activated support. These metal oxides possess strong redox properties and surface acidity that facilitate the cleavage of the amine-hydrogen chloride bond at relatively mild temperatures ranging from 60°C to 100°C. The secondary active components, such as chromium trioxide, further augment the catalyst's oxidative capability and structural integrity, preventing nanoparticle agglomeration during prolonged operation.
From a reaction engineering perspective, the mechanism involves the adsorption of the amine salt onto the acidic sites of the catalyst surface, followed by the thermal elimination of HCl. The porous three-dimensional network structure of the catalyst ensures high mass transfer efficiency, allowing the reactant molecules to access active sites deep within the particle matrix. This design effectively lowers the activation energy required for decomposition compared to thermal decomposition alone. Moreover, the specific arrangement of metal oxides creates a synergistic effect that suppresses side reactions, such as the formation of N-methylated byproducts or higher cyclic amines, which are common impurities in MDA synthesis. By maintaining a highly controlled surface environment, the catalyst ensures that the conversion rate remains consistently high, often exceeding 99%, while minimizing the degradation of the valuable diamine product. This level of control is essential for achieving the high-purity OLED material or pharmaceutical grade standards required by discerning international buyers.
How to Synthesize Diphenylmethane Diamine Efficiently
Implementing this catalytic technology requires precise adherence to the preparation and operational parameters defined in the patent to ensure optimal performance and safety. The synthesis begins with the meticulous preparation of the catalyst, followed by the integration of the catalytic reactor into the existing condensation workflow. Operators must monitor temperature and pressure gradients carefully to maintain the catalyst in its active state while maximizing the recovery of HCl gas. The following guide outlines the critical stages for establishing this efficient production line, focusing on the transition from batch neutralization to continuous catalytic processing.
- Prepare the composite catalyst by acid-treating a nano-SiO2 or carbon nanotube carrier, followed by impregnation with cerium, tin, and chromium salts.
- React aniline with formaldehyde under acidic conditions to form the corresponding amine hydrochloride salt mixture.
- Pass the amine salt mixture through a fixed-bed reactor containing the composite catalyst at 60-100°C to decompose the salt into free amine and recoverable HCl gas.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic decomposition technology offers transformative benefits that extend far beyond simple yield improvements. The most immediate impact is seen in the raw material cost structure, where the complete elimination of caustic soda removes a significant variable cost component from the manufacturing budget. Since the process does not introduce large volumes of aqueous alkali, the downstream separation units operate with much higher efficiency, reducing the load on wastewater treatment facilities and lowering utility costs associated with steam and cooling water. This operational leanness translates into a more resilient supply chain, as the production process becomes less dependent on the fluctuating prices and availability of bulk chemicals like sodium hydroxide. Additionally, the reduction in equipment fouling due to salt deposition extends the campaign life of reactors and distillation columns, decreasing maintenance downtime and ensuring more consistent delivery schedules for customers.
- Cost Reduction in Manufacturing: The economic argument for this technology is robust, driven primarily by the removal of caustic soda from the bill of materials. In traditional processes, the cost of neutralizing agents and the subsequent handling of saline waste can account for a substantial portion of operating expenses. By switching to a catalytic route, manufacturers eliminate these recurring costs entirely. Furthermore, the energy intensity of the process is significantly lowered because the organic product stream contains far less water, requiring less thermal energy for drying and distillation. The ability to recycle the generated HCl gas back into the front end of the process further compounds these savings by reducing the need for fresh acid purchases. These cumulative efficiencies result in a markedly lower cost of goods sold, providing a competitive edge in price-sensitive markets without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexities of waste management and equipment maintenance inherent in older technologies. The catalytic process mitigates these risks by simplifying the workflow and reducing the generation of hazardous waste streams. With fewer unit operations dedicated to washing and salt removal, the overall footprint of the plant is optimized, allowing for greater throughput within existing infrastructure. The robustness of the composite catalyst, designed to withstand thermal stress and resist deactivation, ensures long run-times between regeneration cycles. This reliability allows supply chain planners to forecast production volumes with greater confidence, reducing the likelihood of stockouts or delays caused by unplanned shutdowns. Consequently, partners can rely on a steadier flow of high-purity pharmaceutical intermediates to meet their own production targets.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial effluent tighten globally, the environmental profile of a manufacturing process becomes a critical factor in vendor selection. This catalytic method inherently produces significantly less wastewater compared to neutralization-based routes, as there is no saline brine to dispose of. The reduction in liquid waste volume eases the burden on effluent treatment plants and lowers the risk of non-compliance penalties. Moreover, the process is highly scalable; the use of fixed-bed reactors allows for linear capacity expansion by simply adding more reactor trains or increasing catalyst volume, without the need for disproportionately larger separation tanks. This scalability supports the commercial scale-up of complex polymer additives and intermediates, enabling manufacturers to respond agilely to market demand surges while maintaining a sustainable operational footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic MDA synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of how this method compares to incumbent technologies. Understanding these details is vital for technical teams evaluating the feasibility of adopting this greener and more efficient manufacturing route.
Q: How does the catalytic decomposition method improve MDA purity compared to traditional neutralization?
A: By avoiding the addition of caustic soda (NaOH), this method prevents the formation of inorganic salt byproducts (NaCl) that often create emulsions and impurity layers during phase separation. The catalytic route yields an organic phase with significantly lower water content and fewer entrained impurities, resulting in higher purity MDA suitable for sensitive downstream applications.
Q: What are the primary cost-saving mechanisms in this patented process?
A: The process eliminates the recurring cost of purchasing large quantities of caustic soda for neutralization. Furthermore, because no aqueous caustic solution is added, the water load in the organic phase is drastically reduced, leading to substantial energy savings during the subsequent distillation and drying steps. Maintenance costs are also lowered due to reduced salt scaling in downstream equipment.
Q: Can the HCl byproduct be recycled in this manufacturing process?
A: Yes, a key advantage of this technology is the generation of HCl gas as a clean byproduct of the amine salt decomposition. This gas can be absorbed in process water to regenerate high-concentration hydrochloric acid, which can then be recycled back to the front-end aniline salting step, creating a closed-loop system that minimizes waste discharge.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenylmethane Diamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic processes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust industrial realities. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical instrumentation. Whether you require custom synthesis of specialized MDA isomers or large-volume supply for polyurethane production, our infrastructure is designed to accommodate your specific needs with precision and reliability.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your application. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and expert insight. Let us collaborate to optimize your supply chain and drive innovation in your product development pipeline.
