Advanced Catalytic Synthesis of Low-Impurity Diphenylmethane Diamines for Commercial Scale-Up
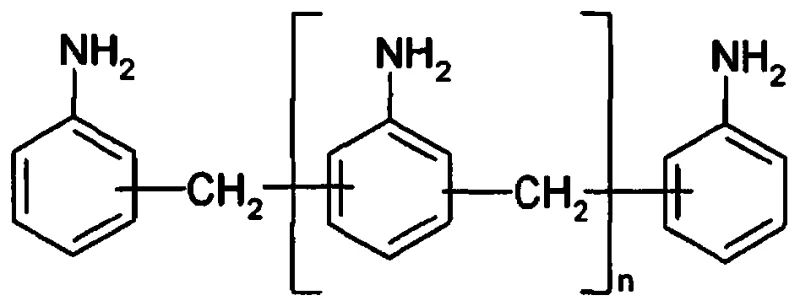
The global demand for high-performance polyurethane materials continues to drive the need for superior precursors, specifically diphenylmethane series diamines and polyamines (DAM). A significant technological breakthrough in this sector is detailed in patent CN107827756B, which introduces a novel preparation method capable of drastically reducing N-methyl impurity content. Traditional manufacturing routes often struggle with selectivity during the rearrangement phase, leading to byproducts that compromise the quality of downstream Methylene Diphenyl Diisocyanate (MDI). This patent addresses these challenges by employing a specialized fixed-bed reactor system loaded with advanced catalysts, such as vanadium phosphate oxide or rare earth solid solutions. By optimizing the transposition rearrangement stage, this method ensures that the N-methyl MDA content is suppressed to levels below 0.01 wt%, representing a substantial leap forward in product purity and process efficiency for industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of diphenylmethane diamines has relied heavily on the condensation of aniline with formaldehyde under acidic conditions, followed by a rearrangement step. While this general pathway is well-established, conventional implementations frequently suffer from poor selectivity during the critical transposition rearrangement phase. In standard processes, the reaction environment often promotes various side reactions, most notably the formation of N-methylated derivatives. These N-methyl impurities are notoriously difficult to remove through standard purification techniques like distillation due to their similar boiling points to the desired product. Consequently, manufacturers face significant challenges in meeting the stringent purity specifications required by top-tier polyurethane producers. The accumulation of these impurities not only degrades the quality of the final MDI but also complicates the phosgenation process, leading to operational inefficiencies and increased waste generation in large-scale facilities.
The Novel Approach
The innovative methodology described in the patent data fundamentally reengineers the rearrangement step by shifting from homogeneous or less selective conditions to a heterogeneous catalytic system within a fixed-bed reactor. Instead of relying solely on thermal energy or non-specific acid catalysis, this approach utilizes highly active and selective catalysts, including vanadium phosphate oxides and specific solid solutions like Nb2O5-La2O3 or Pr2O3-Ce2O3. This transition allows for precise control over the reaction pathway, effectively steering the conversion of polyaminobenzylaniline salts towards the desired diphenylmethane skeleton while inhibiting the methylation side reactions. The use of a fixed-bed configuration further enhances process stability and facilitates continuous operation, which is essential for modern chemical manufacturing. By decoupling the condensation and rearrangement stages and applying targeted catalysis in the latter, the process achieves a level of selectivity that conventional batch methods simply cannot match, resulting in a dramatically cleaner product profile.
Mechanistic Insights into Catalytic Transposition Rearrangement
The core of this technological advancement lies in the unique properties of the catalyst active components employed during the transposition rearrangement. The patent highlights the efficacy of vanadium phosphate oxides, which are known for their redox properties and acid-base bifunctionality, as well as rare earth oxide solid solutions. When supported on carriers like gamma-alumina or titanium dioxide, these active species create a specific surface environment that favors the migration of amino groups to the para-position of the aromatic ring without promoting N-methylation. The mechanism likely involves the activation of the benzylaniline intermediate through coordination with the metal centers of the catalyst, lowering the activation energy for the desired rearrangement while sterically or electronically hindering the attack of methylating agents. Furthermore, the solid solution nature of catalysts like Nb2O5-La2O3 ensures a uniform distribution of active sites, preventing localized hot spots that could trigger decomposition or unwanted side reactions. This precise mechanistic control is what enables the reduction of N-methyl impurities to trace levels, ensuring that the molecular architecture of the diamine remains intact and pure.
Impurity control in this context is not merely about removing contaminants post-reaction but preventing their formation at the source. The presence of N-methyl groups typically arises from the reaction of amine functionalities with formaldehyde or methanol (often present as a stabilizer in formaldehyde solutions) under acidic conditions. The novel catalyst system appears to modulate the acidity and the local concentration of reactive species within the pore structure of the fixed bed. By optimizing the residence time and temperature profile within the reactor—typically maintained between 65°C and 120°C—the process minimizes the exposure of the amine intermediates to conditions that favor methylation. Additionally, the robustness of these catalysts in strong acidic environments, such as those containing hydrochloric acid from the preceding condensation step, ensures long-term stability and consistent performance. This dual focus on enhancing desired reaction kinetics while suppressing parasitic pathways represents a sophisticated application of heterogeneous catalysis to solve a persistent problem in fine chemical synthesis.
How to Synthesize Diphenylmethane Diamine Efficiently
Implementing this advanced synthesis route requires a systematic approach that integrates the initial condensation of aniline and formaldehyde with the subsequent catalytic rearrangement. The process begins with the formation of polyaminobenzylaniline salts in a condensation reactor, where parameters such as acid concentration and molar ratios are tightly controlled. Following this, the crude mixture is transferred to the fixed-bed reactor containing the specialized catalyst, where the critical transformation occurs. The detailed operational parameters, including specific flow rates, temperature gradients, and catalyst loading procedures, are essential for replicating the high selectivity reported in the patent documentation. For R&D teams looking to adopt this technology, understanding the interplay between the condensation effluent and the fixed-bed catalyst is paramount to achieving the target purity specifications. The standardized synthesis steps outlined below provide a foundational framework for scaling this efficient production method.
- Perform condensation of aniline and formaldehyde in the presence of an acidic catalyst to form polyaminobenzylaniline salts.
- Pass the reaction mixture through a fixed-bed reactor containing vanadium phosphate oxide or rare earth solid solution catalysts for transposition rearrangement.
- Neutralize the mixture with alkali, separate organic phases, wash thoroughly, and distill to isolate high-purity diamines and polyamines.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this low-impurity synthesis method offers tangible strategic benefits beyond mere technical specification compliance. The primary advantage lies in the significant enhancement of product quality consistency, which directly translates to reduced risk in the supply of critical polyurethane intermediates. By minimizing N-methyl impurities at the source, manufacturers can eliminate or drastically reduce the need for complex and energy-intensive downstream purification steps, such as multiple distillation columns or extensive washing cycles. This simplification of the production workflow leads to substantial cost reductions in manufacturing operations, as less energy is consumed and fewer resources are wasted on separating difficult byproducts. Furthermore, the use of a fixed-bed reactor system supports continuous processing, which inherently improves throughput and equipment utilization rates compared to traditional batch operations. This shift towards continuous manufacturing aligns with modern industry trends for efficiency and reliability, ensuring a more stable supply of high-purity materials for downstream customers.
- Cost Reduction in Manufacturing: The elimination of expensive and complex purification stages required to remove N-methyl impurities results in direct operational savings. By preventing the formation of these byproducts through superior catalytic selectivity, the process reduces the load on distillation units and lowers utility consumption associated with heating and cooling. Additionally, the extended lifespan and stability of the heterogeneous catalysts reduce the frequency of catalyst replacement and regeneration, further contributing to lower overall production costs. This economic efficiency allows suppliers to offer competitive pricing while maintaining high margins, creating a win-win scenario for both producers and buyers in the value chain.
- Enhanced Supply Chain Reliability: The robustness of the fixed-bed catalytic process ensures a consistent and uninterrupted flow of high-quality diamines. Unlike batch processes that may suffer from variability between runs, the continuous nature of this technology provides a steady output that helps stabilize inventory levels and meet just-in-time delivery requirements. The ability to produce material with consistently low impurity profiles reduces the likelihood of batch rejections or customer complaints, thereby strengthening trust and long-term partnerships between suppliers and major polyurethane manufacturers. This reliability is crucial for maintaining the continuity of production lines in industries where downtime is extremely costly.
- Scalability and Environmental Compliance: The technology is inherently scalable, moving seamlessly from pilot studies to full commercial production without the nonlinear challenges often faced in batch scale-up. The improved selectivity also means less waste generation, as fewer byproducts are formed that require disposal or treatment. This aligns with increasingly stringent environmental regulations regarding chemical manufacturing emissions and effluent quality. By adopting a cleaner production method, companies can demonstrate a commitment to sustainability and regulatory compliance, which is becoming a key differentiator in global supply chains and a prerequisite for doing business with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this low-impurity diamine synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and technical stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing frameworks or for sourcing high-specification intermediates from qualified suppliers. The answers reflect the core innovations in catalyst design and process engineering that define this advanced method.
Q: How does the novel catalyst reduce N-methyl impurities in DAM?
A: The use of specific active components like vanadium phosphate oxide or Nb2O5-La2O3 solid solutions in a fixed-bed reactor significantly improves the selectivity of the transposition rearrangement stage, suppressing side reactions that form N-methyl byproducts.
Q: What is the achievable purity level for N-methyl MDA using this method?
A: According to the patented technology, the content of N-methyl MDA impurities can be reduced to below 0.01 wt%, which is critical for producing high-quality MDI for polyurethane applications.
Q: Why is low N-methyl content critical for MDI production?
A: High levels of N-methyl impurities in the diamine precursor negatively impact the subsequent phosgenation reaction, leading to inferior quality monomeric and polymeric MDI, which affects the final performance of polyurethane foams and materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenylmethane Diamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of raw materials dictates the performance of the final polymer. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated catalytic technologies described in patents like CN107827756B are translated into reliable industrial reality. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of diphenylmethane diamine meets the exacting standards required for high-end polyurethane applications. We understand the critical nature of N-methyl impurity control and have optimized our processes to deliver products that facilitate superior MDI synthesis for our global clientele.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that demonstrates how switching to our high-purity intermediates can optimize your overall production economics. Please contact us today to request specific COA data and route feasibility assessments, and let us show you how our commitment to innovation and quality can support your supply chain goals.
